Which Statement Best Describes Metallic Bonding
Ever wondered why that spoon you're stirring your tea with feels solid and shiny, or why a nail can be hammered into wood without crumbling into dust? The answer, my curious friends, lies in a rather fascinating type of chemical bond called metallic bonding. It might sound like something straight out of a science lab, but understanding it is actually quite a blast and explains so much about the world around us!
Think about it: metals are everywhere! From the intricate circuitry in your phone to the sturdy framework of buildings, metals are the backbone of our modern lives. Metallic bonding is the secret sauce that gives these materials their unique and incredibly useful properties. Its purpose? To hold metal atoms together in a way that creates a stable, yet flexible, structure. The primary benefit is the creation of materials that are strong, conductive (both heat and electricity!), and malleable – meaning you can shape them without breaking them. Imagine if your pots and pans were brittle and couldn't conduct heat; cooking would be a very different, and likely frustrating, experience!
In education, exploring metallic bonding often involves hands-on activities. Students might look at different metal samples, observe their luster, and test their conductivity. Demonstrations showing how wires carry electricity or how heat travels through a metal rod are classic examples. In our daily lives, we see its effects constantly. The aluminum foil in your kitchen is a perfect example of its malleability. The copper wiring in your home showcases its excellent electrical conductivity. Even the chrome on your car's bumper, with its characteristic shine, is a testament to metallic bonding's ability to create reflective surfaces.
So, how can you, as a curious individual, delve a little deeper into this world of metallic bonds without needing a full chemistry set? It's simpler than you might think! Next time you're handling a metal object, take a moment to really observe it. Notice its texture, its weight, and how it reflects light. You could even do a simple conductivity test (with adult supervision, of course!) using a battery, a small light bulb, and different metal objects – you'll quickly see which ones allow the electricity to flow. Think about the different types of metals you encounter and try to relate their uses to the properties we've discussed. Is that flimsy aluminum can shaped the way it is because of how it's bonded? Absolutely!
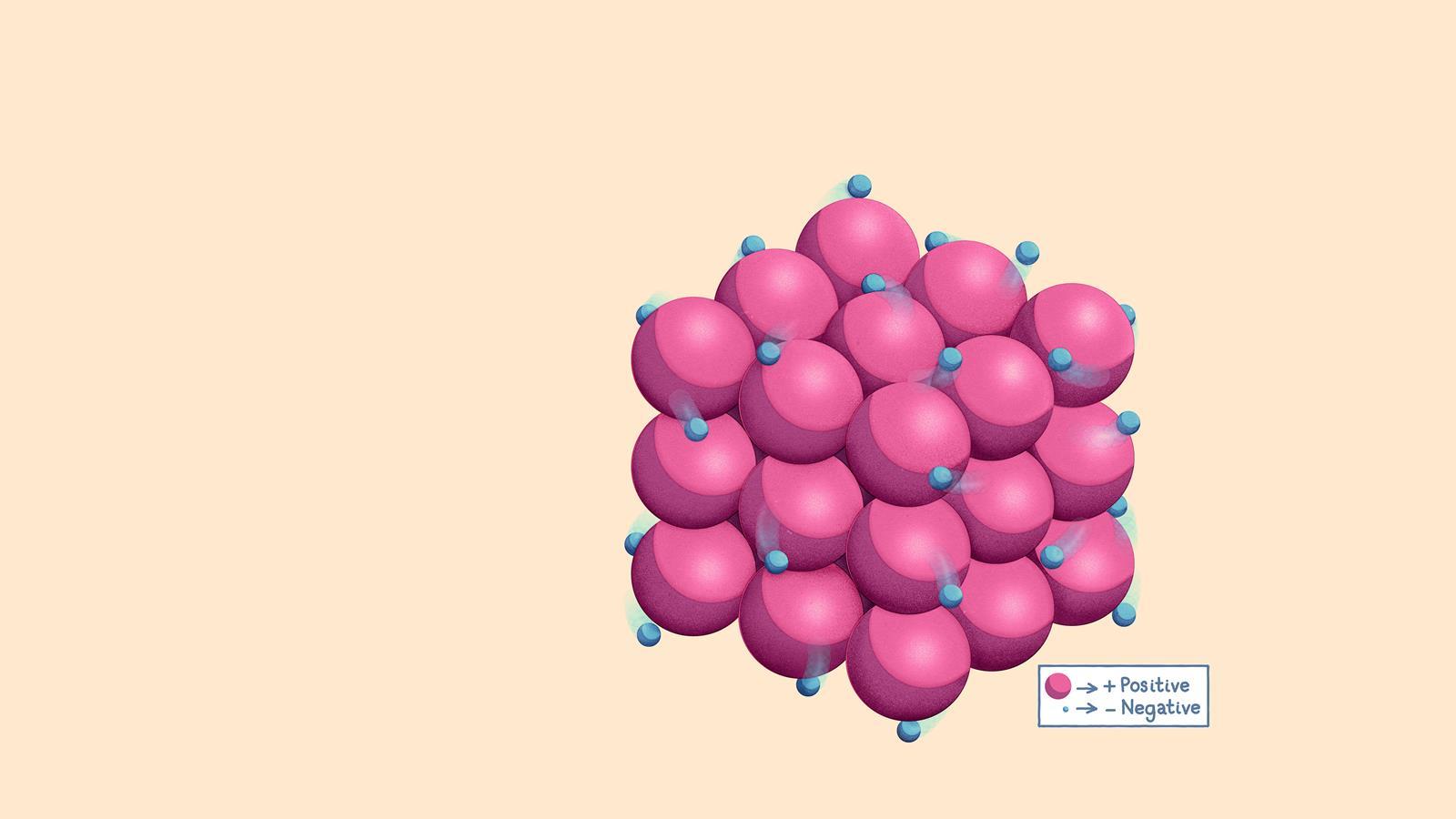
Essentially, metallic bonding describes a way metal atoms are held together. Instead of sharing electrons like in some other bonds, in metallic bonding, the outer electrons of metal atoms are delocalized, meaning they form a sort of 'sea' or 'cloud' of electrons that surrounds the positively charged metal ions. This 'sea of electrons' is what allows metals to conduct electricity and heat so well, and it also gives them their characteristic shiny appearance. It's a beautiful dance of charged particles that results in the sturdy, adaptable materials we rely on every single day. It’s a powerful concept that, once grasped, makes you see the metallic world around you in a whole new, and quite fascinating, light.
