Which Statement Accurately Describes Radioactive Dating
Alright, let's talk about something that sounds super science-y, but honestly, it's kind of like figuring out how old your grandma's secret cookie recipe is. We're diving into the wonderful world of radioactive dating. Forget dusty textbooks and intimidating equations for a sec. Think of it like this: everything, from your car keys that seem to mysteriously vanish to the ancient rocks in your backyard, has a story, and radioactive dating is basically the universe's way of letting us peek at its timeline.
You know how sometimes you find an old photograph, and you can tell by the hairstyles and the slightly-too-bright colors that it’s from, like, the 80s? Or maybe even earlier? That’s kind of what radioactive dating does, but instead of big hair and questionable fashion choices, it looks at the tiny, almost invisible changes happening within certain elements. It's like a built-in clock, but instead of ticking, it’s decaying. Spooky, right? But also, super cool.
So, what's the big idea? Imagine you have a bag of those candies that change color as you eat them. You start with a whole bag of red ones, but over time, some of them slowly turn into blue ones. Radioactive dating is a bit like that, but with atoms. Certain atoms, called isotopes, are a little unstable. They’re like that one friend who’s always fidgeting and eventually just… changes into something else.
These unstable isotopes, called radioactive isotopes, have a tendency to break down, or decay, into a more stable form. It’s like a natural process, a bit like how bread eventually goes stale, or how your phone battery mysteriously drains overnight even when you’re not using it. Except, this decay happens at a very specific, predictable rate. It’s not random; it’s like the universe has a stopwatch running for each of these radioactive elements, and it doesn’t speed up or slow down no matter what.
Now, here’s where it gets really interesting. Think about when you bake a cake. You start with a certain amount of flour, sugar, and eggs. As the cake bakes, those ingredients transform. Radioactive dating is similar, but instead of transforming into a delicious dessert, these radioactive isotopes transform into a different element, or a different form of the same element. We call this the daughter isotope, and the original unstable one is the parent isotope. It’s like a scientific family tree, where the parent isotope eventually has a daughter isotope.
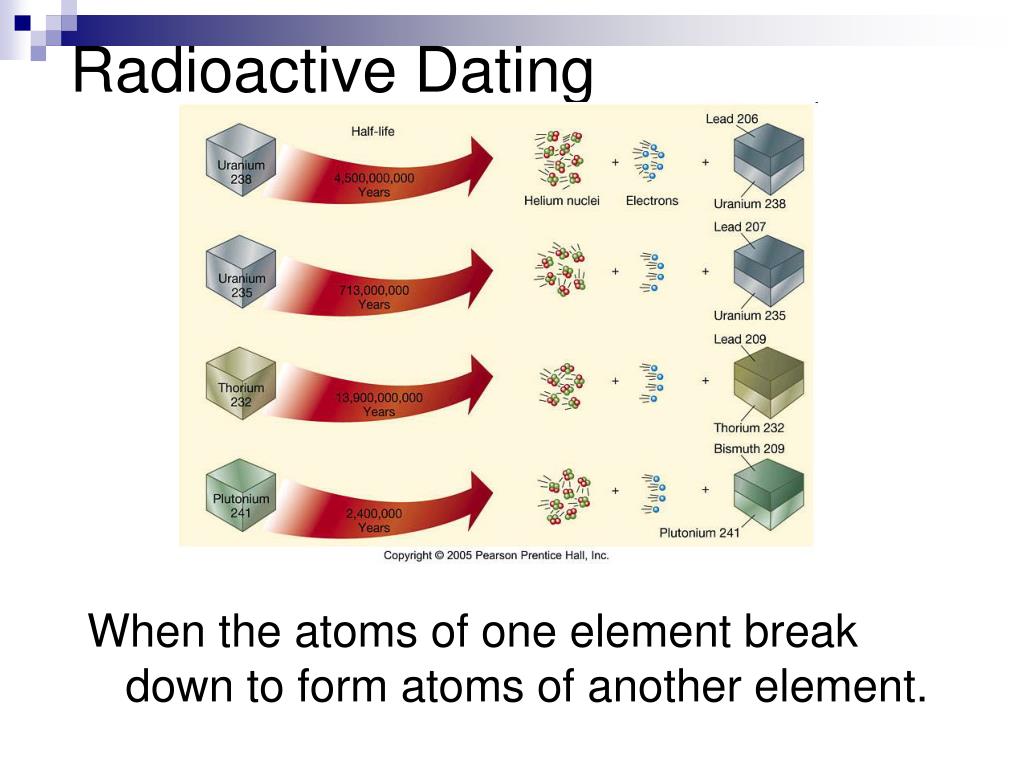
The magic of radioactive dating lies in the half-life. This is probably the most important concept, so let’s give it a good chew. The half-life of a radioactive isotope is the exact amount of time it takes for half of the parent isotopes in a sample to decay into daughter isotopes. Imagine you have 100 little radioactive atom buddies. After one half-life, you’ll have 50 of the original buddies and 50 of the new, daughter buddies. After another half-life, you’ll have 25 of the original and 75 of the daughter. It’s a consistent, reliable reduction. Like a discount that keeps halving!

This half-life thing is super crucial because it acts as our cosmic clock. Different radioactive isotopes have different half-lives. Some are super short, like seconds or minutes, which are great for dating very recent things. Others are astronomically long, like billions of years. This is why radioactive dating is so versatile. It’s like having a whole toolbox of different sized measuring tapes.
Let’s say you find a really old fossil. You can’t exactly ask it for its ID, right? So, scientists will look for rocks associated with that fossil. These rocks often contain radioactive isotopes. If they find a rock with a certain radioactive element, say, Potassium-40 (sounds fancy, right?), and they know its half-life is about 1.25 billion years, they can measure how much Potassium-40 is left and how much of its daughter isotope, Argon-40, has accumulated. By comparing the ratio, they can figure out how much time has passed since that rock solidified.
It's like finding an old, half-eaten jar of pickles in your fridge. You know the pickles weren’t always this… mushy. You can sort of gauge how long they’ve been there based on their current state. Radioactive dating just does this with a scientific eye, a lot more precisely, and without the questionable smell.

So, which statement accurately describes radioactive dating? It’s the one that captures this fundamental idea: that the predictable decay of radioactive isotopes, measured by their half-lives, allows us to determine the age of ancient materials.
Let’s break down what an inaccurate statement might sound like, just to get it crystal clear. Imagine someone saying, “Radioactive dating measures how quickly things are moving.” Nope! Not about speed, it’s about time. Or, “It works by looking at how shiny an object is.” Definitely not. It’s about the internal, atomic changes.
A more accurate statement would highlight the core mechanism. It's about a parent isotope transforming into a daughter isotope at a constant rate, defined by its half-life. This transformation is what gives us the timeline. Think of it like a secret code that nature has embedded in certain rocks and organic materials. Scientists are essentially the code-breakers, using their scientific tools to read that code.
Consider dating something like a dinosaur bone. We can’t directly date the bone itself with the long-lived isotopes used for rocks, because organic material decays differently and is less stable over millions of years. But, we can date the layers of volcanic ash or rock that surround the bone. If a layer of ash, dated using something like Uranium-Lead, shows it’s 150 million years old, and the dinosaur bone is found embedded within that layer, then we know that dinosaur lived around 150 million years ago. It’s like finding a toy car in a time capsule – the time capsule’s age tells you when the car was sealed away.

What about dating things that are closer to home, like ancient pottery or old wooden artifacts? For these, we often use radiocarbon dating. This uses Carbon-14, a radioactive isotope of carbon that’s constantly being produced in the atmosphere. Living organisms take in Carbon-14, and when they die, they stop taking it in. The Carbon-14 inside them then starts to decay with a half-life of about 5,730 years. So, by measuring the amount of Carbon-14 left in an ancient object, we can figure out how long ago it was alive.
It’s like a loaf of bread. When it’s fresh, it’s full of bouncy, airy goodness. As it ages, it gets stale and dry. Radiocarbon dating is like measuring the “staleness” of the Carbon-14 in an organic object to see how long ago it was alive and breathing. But instead of being stale, it’s just got less radioactive stuff in it!
The accuracy of radioactive dating relies on a few key assumptions. One is that the initial amount of the parent isotope was known or can be reasonably estimated. Another is that the system hasn’t been contaminated or disturbed since the object formed. If a rock has been melted and reformed, or if a fossil has been replaced by minerals, it can mess with the clock. It’s like if you dropped your phone in the ocean – it might not tell time accurately anymore!

So, when you see a statement about radioactive dating, look for these key elements: the idea of decay, the use of isotopes (parent and daughter), and the concept of a fixed, measurable rate like a half-life. It’s the science of turning tiny atomic changes into massive leaps through time.
It’s a process that has revolutionized our understanding of the Earth's history, the evolution of life, and even the age of the universe itself. From the microscopic world of atoms, we get a macroscopic view of epochs long past. It’s pretty mind-blowing when you think about it.
So, an accurate statement would boil down to something like: Radioactive dating determines the age of materials by measuring the ratio of parent radioactive isotopes to their stable daughter isotopes, which decay at a constant rate determined by the isotope's half-life. It’s the scientific equivalent of finding a really old, but still ticking, grandfather clock that’s been running perfectly since the dawn of time.
It’s not magic, although it feels pretty close. It’s just really smart science using the natural processes of the universe to tell us stories about the past. And who doesn’t love a good story, especially one that’s billions of years in the making?
