Which Statement About The Self Ionization Of Water Is Correct

Hey there, science curious pals! Ever thought about water? Like, really thought about it? We all know it's super important for, well, everything. But did you know water can be a little bit of a drama queen? Like, it can totally do its own thing, all by itself.
We're talking about the self-ionization of water. Sounds fancy, right? But it's actually way cooler and a lot less intimidating than it sounds. Think of it like water having a secret life when no one's looking.
Water: Not as Simple as You Think!
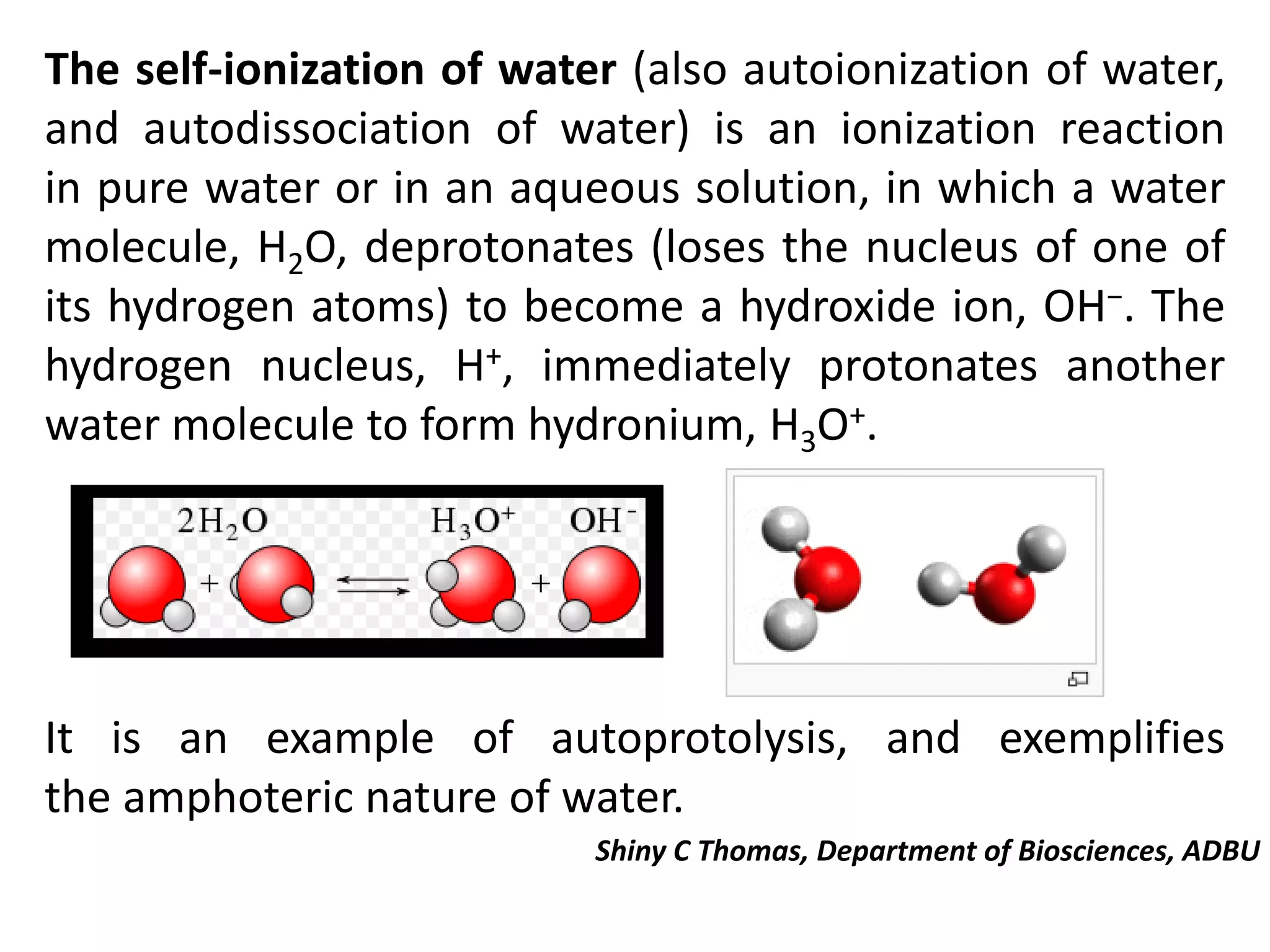
So, what's this "self-ionization" jazz? Imagine water molecules, H2O, chilling together. Most of the time, they're just vibing. But sometimes, just sometimes, one water molecule is like, "You know what? I'm gonna share my hydrogen atom with my neighbor."
It's like a tiny, microscopic act of generosity. Or maybe just a molecular identity crisis. Who knows? But this little transfer creates something pretty neat. One molecule becomes a hydroxide ion (OH-). It's got a negative vibe, you know?
The other molecule, the one that lost the hydrogen, becomes a hydronium ion (H3O+). This one's got a positive outlook. Get it? Positive charge!

So, in the grand scheme of pure water, you've got these hydronium ions and hydroxide ions hanging out. It’s a super, super tiny fraction of the total water. Like, if you had a million water molecules, only a couple would be doing this little dance. It’s a very exclusive club.
The Constant Tug-of-War
This whole process is happening all the time, in both directions. Water molecules are breaking apart and reforming, creating these ions. It's a dynamic equilibrium, which is just a fancy way of saying it’s a constant, balanced back-and-forth. Like a tiny, silent tug-of-war inside your glass of water.
And here's the super fun part: the balance between these ions is key to whether something is acidic or basic. That's where pH comes in! Remember pH from chemistry class? This is its origin story!

Which Statement is Actually Correct? Let's Break It Down!
Okay, so you've got this self-ionization thing going on. Now, let's look at some statements you might hear about it. One of them is the real deal. Think of this as a little pop quiz, but with way less pressure and way more fun facts.
Statement 1: Pure water has a pH of 7 because it's completely neutral and has no ions.
Uh oh, red flag! This one’s trying to trick you. Remember our little ion dance? Pure water absolutely has ions. It's not an empty void of neutrality. The reason it's pH 7 is because of the balanced presence of hydronium and hydroxide ions. If there were no ions, what would be there to make it neutral? This statement is like saying a party is empty because there are only a few people dancing. Nope, those dancers are what make it a party!
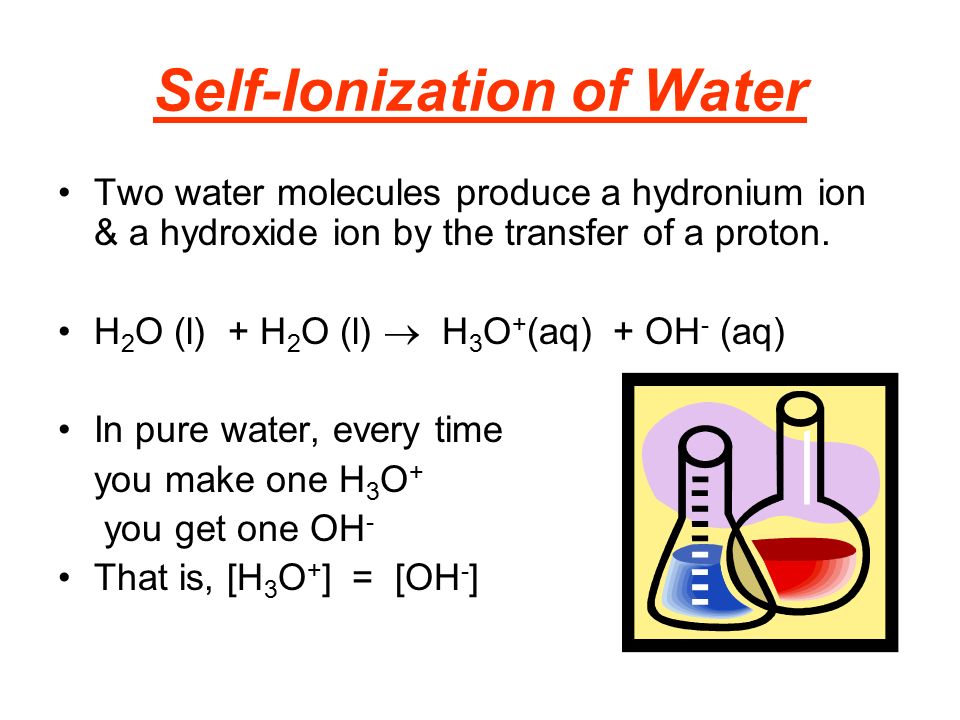
Statement 2: In pure water, the concentration of hydronium ions is equal to the concentration of hydroxide ions.
Ding, ding, ding! We have a winner! This statement is spot on. Because the self-ionization process creates one hydronium ion for every hydroxide ion, their concentrations in pure water are always, always, always equal. This perfect balance is what gives pure water its neutral pH of 7 at standard temperatures. It's like a perfectly matched pair, always in sync. They’re the ultimate power couple of pure water!
Statement 3: The self-ionization of water is a process that only happens when you add something acidic or basic to it.
Nope, not quite! This statement misses the fundamental nature of self-ionization. Water does this all by its lonesome, even when it’s just plain old, pure water. Adding acids or bases changes the balance of these ions, but it doesn't start the process. Think of it like this: the water is already making a tiny bit of juice and a tiny bit of lemonade. Adding store-bought juice or lemonade just tips the scale, it doesn’t invent juice and lemonade.
Statement 4: Water molecules are perfectly stable and never break apart.
Myth Busted! If this were true, we wouldn't have acids, bases, or pH as we know it. Water molecules are dynamic! They're constantly interacting, and a tiny, tiny fraction of them are indeed breaking apart and reforming. It’s like a microscopic perpetual motion machine, but instead of energy, it’s creating ions.
Why This Quirkiness Matters (and is Fun!)
So, why is this tiny bit of water ionization such a big deal? Because it's the foundation of all our understanding of acids and bases. Think about cooking – that sour taste of lemon juice? That’s all about hydronium ions!
Or even your own body! Your blood has to maintain a very specific pH, and that balance relies on the chemistry of water and its ions. It’s wild to think that something so fundamental to life happens because water molecules can't quite make up their minds about who gets the hydrogen.
It's these little quirks of nature that make science so fascinating. Water, this everyday substance, has a hidden, dynamic personality. It’s always doing its own thing, creating a delicate balance that affects everything around it.
So next time you drink a glass of water, just remember the tiny, electrifying dance happening within. It’s a reminder that even the simplest things can hold incredible, complex secrets. And isn't that just the coolest?
