Which Statement About The Atomic Nucleus Is Correct

Okay, let's dive into the teeny-tiny, super-duper important world of the atomic nucleus! It's like the secret heart of everything, from the air you're breathing right now to the delicious pizza you might be dreaming about. We're going to uncover which statement about this amazing little guy is the real winner.
Imagine an atom is like a miniature solar system. You've got the electrons whizzing around on the outside, kind of like planets on their orbits. But what's at the very center, holding everything together with its gravitational pull (or, you know, a slightly different kind of force)? That, my friends, is the atomic nucleus!
Now, the nucleus is not some wispy, ethereal thing. Oh no! It's packed, and I mean packed, with stuff. It’s the dense, powerhouse core of the atom. If an atom were the size of a football stadium, the nucleus would be like a tiny, super-heavy pea right in the middle! Talk about concentrated awesomeness.
Unpacking the Nucleus: What's Inside?
So, what exactly is jamming in this incredible nucleus? It’s a dynamic duo of sorts, made up of two main types of particles. Think of them as the ultimate tag team, working tirelessly to keep the atom intact.
First up, we have protons! These guys are like the positive energy bombs of the nucleus. They carry a positive electrical charge, which is a big deal when it comes to how atoms interact. Without protons, atoms would be, well, a whole lot less interesting.
Then, we have the neutrons. These are the calm, cool, and collected members of the nucleus. They don't have any electrical charge at all – they're neutral, hence the name! But don't let their chill vibe fool you; they play a crucial role in keeping those positively charged protons from freaking out and flying apart.

Together, protons and neutrons are collectively called nucleons. They're like the inseparable best friends of the atomic world, huddled together in the center. They're held together by a force so strong, it's like a superhero’s grip!
The Astonishing Density
Let’s talk about how incredibly dense the nucleus is. Remember that football stadium analogy? If you could somehow take all the material in the nucleus and squish it down, a single teaspoon of it would weigh about as much as Mount Everest! Mind. Blown.
This incredible density is because all those protons and neutrons are crammed in there super close together. There's barely any space between them. It’s like a packed concert venue where everyone’s having a fantastic time, but they’re also very, very close.
This density is a key characteristic that scientists have figured out over years of amazing experiments and brilliant thinking. It tells us a lot about the forces at play and the fundamental nature of matter.
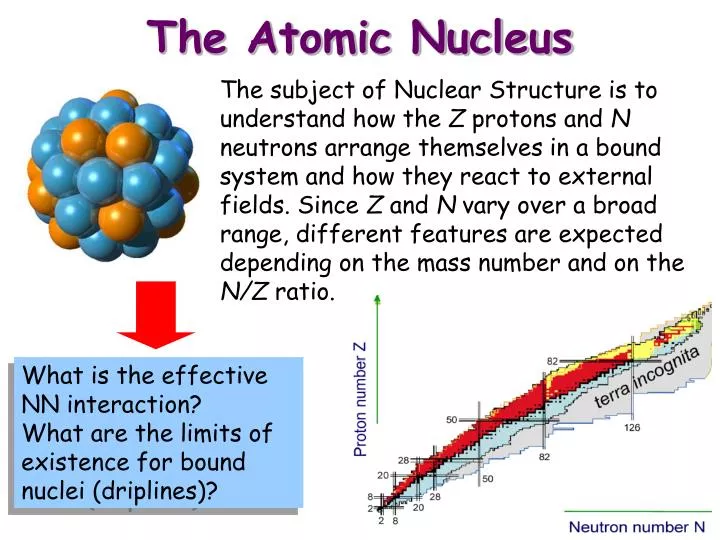
The Nucleus and Stability
The balance between protons and neutrons is super important for the nucleus to be stable. Think of it like a perfectly balanced seesaw. If you have too many of one and not enough of the other, things can get a bit wobbly.
Sometimes, a nucleus might have an unstable number of neutrons. This is when things get really interesting and we enter the realm of radioactivity! These unstable nuclei will spontaneously break down, releasing energy and particles to become more stable. It's nature’s way of tidying up!
This process of radioactivity is what powers things like smoke detectors and even certain medical treatments. So, even in its instability, the nucleus is doing important work! It’s a constant dance of energy and transformation.
Electrons and the Nucleus: A Relationship of Opposites
Now, let’s not forget those speedy electrons zipping around. They’re attracted to the positively charged protons in the nucleus. It’s like a cosmic game of magnets, but with electrical charges.

This attraction is what keeps the electrons in their orbits and defines the overall behavior of the atom. Without the nucleus’s positive pull, the electrons would just zoom off into the void. The nucleus is the anchor, the gravitational center (metaphorically speaking, of course!).
So, while the electrons are busy doing their thing on the outer edges, the real action, the heavy lifting, the fundamental identity of the atom, all happens right there in that incredibly dense, charged core. It's the ultimate control center!
The Verdict: Which Statement is Correct?
Alright, drumroll please! We’ve explored the packed nature, the components, and the crucial role of the atomic nucleus. Considering all this, let's look at some potential statements and see which one truly rings true.
Could it be that the nucleus is mostly empty space? Nope! We’ve established it’s incredibly dense. Imagine trying to squeeze an entire elephant into a teacup – that’s how not empty it is!

Perhaps the nucleus is made up of only electrons? Absolutely not! Electrons live outside the nucleus, much like planets orbit a star. The nucleus is where the protons and neutrons hang out.
And what about this: The atomic nucleus is a dense region at the center of an atom, containing protons and neutrons? YES! That is the champion, the grand prize winner, the undisputed truth!
The atomic nucleus is a dense region at the center of an atom, containing protons and neutrons.
This statement perfectly captures the essence of what we’ve been chatting about. It’s compact, it’s at the heart of the atom, and it's home to those vital nucleons. It's the true story of this incredible subatomic structure.
So, the next time you marvel at the world around you, remember the unsung hero: the atomic nucleus. It’s small, but mighty, and the foundation of absolutely everything we see, touch, and experience. Isn't science just the coolest? Keep exploring, keep wondering, and keep appreciating the tiny marvels that make up our vast universe!
