Which Statement About Surface Tension Is False

Ever watched a water strider dance across a pond, defying gravity as if it were on an invisible trampoline? Or wondered why a tiny droplet of water forms a perfect sphere? This magical phenomenon is all thanks to something called surface tension, and it's not just for science geeks! It’s a hidden force that shapes our everyday world in surprising and delightful ways. From the way your morning coffee steams to the effectiveness of your laundry detergent, surface tension is silently at play. Understanding it makes us appreciate the ordinary a little bit more and unlocks a whole world of fascinating science experiments you can do right at home.
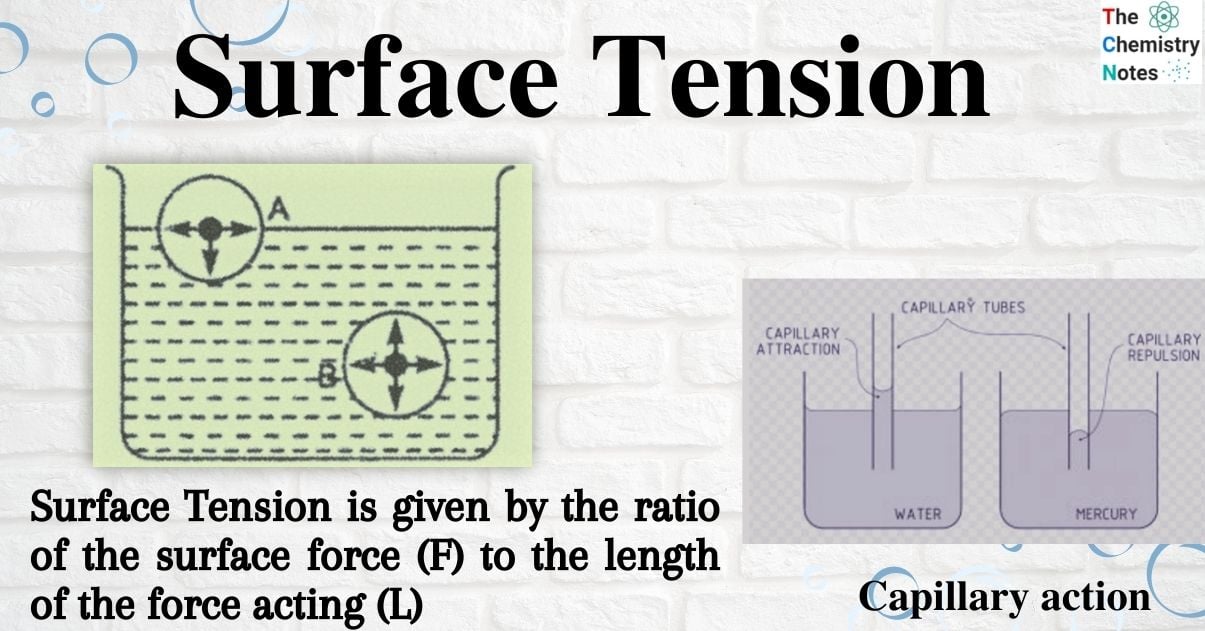
So, what exactly is this superhero force? Imagine the surface of a liquid, like water. The molecules at the surface are a bit like lonely teenagers at a party, surrounded by their friends (other water molecules) below, but with fewer pals on one side (the air). This means they’re being pulled inward and sideways by their neighbors, creating a sort of elastic skin. This inward pull is surface tension. It’s what allows insects to walk on water, makes needles float if carefully placed, and causes water to bead up on a waxy surface. It’s a constant battle between the cohesive forces (molecules sticking together) within the liquid and the forces pulling them outwards.
The Wonderful World of Surface Tension
The benefits of understanding surface tension are pretty widespread. For starters, it’s crucial in nature. Many tiny organisms rely on surface tension to move and survive. Think about how raindrops cling to leaves or how dew forms on spiderwebs – all beautiful examples of this principle in action. In our homes, surface tension plays a role in how soaps and detergents work. These clever substances are designed to reduce surface tension, allowing water to spread out and penetrate fabrics more effectively, lifting away dirt and grime. Without this reduction, your washing wouldn't get nearly as clean!
But it’s not just about cleaning. In medicine, understanding surface tension is vital for things like lung function. The tiny air sacs in our lungs (alveoli) are coated with a fluid, and if surface tension were too high, these delicate sacs would collapse. A special substance called surfactant is naturally produced in our bodies to lower this surface tension, keeping our lungs inflated and allowing us to breathe. Premature babies often have underdeveloped surfactant production, which is why they can experience breathing difficulties.
Even something as simple as pouring a glass of water involves surface tension. You might have noticed that water doesn't always flow smoothly; sometimes it forms a little meniscus, a curved surface at the edge of the container. That’s surface tension at work, along with another related force called adhesion (water molecules sticking to the container). And let's not forget cooking! The way oil and water separate, or how a sauce coats the back of a spoon, are all influenced by surface tension.
Testing Your Knowledge: Which Statement is False?
Now, let's get to the fun part! We’ve talked about what surface tension is and why it matters. But like any scientific concept, there are statements made about it, and not all of them are true. To really get a handle on surface tension, it’s important to be able to identify what’s fact and what’s fiction. Think of it like being a detective, gathering clues and spotting the odd one out.
Let's consider a few common ideas about surface tension:

- Statement A: Surface tension is a force that pulls liquid molecules towards each other.
- Statement B: Adding soap to water increases its surface tension, making it a better cleaner.
- Statement C: Insects like water striders can walk on water because of surface tension.
- Statement D: Surface tension is stronger in liquids with weaker intermolecular forces.
Take a moment to ponder these. We’ve already discussed a few of these concepts. Which one doesn't quite add up based on what we've learned? It’s a good way to solidify your understanding and to start thinking critically about scientific claims.
The beauty of surface tension lies in its simplicity and its profound impact. It’s a concept that’s both easily observable and deeply complex. From the tiny water droplet to the vast ocean, surface tension is a fundamental property of liquids that continues to amaze and inform us.

Think about a time you've seen something intriguing on the surface of water – maybe a tiny bug perfectly balanced, or the way water beads on a freshly washed car. That's surface tension saying hello! It’s a reminder that science isn’t just in labs; it’s all around us, in the most ordinary moments, making the world a more interesting and, dare we say, a bit magical place.
"The surface of water is a window into the molecular world."
So, the next time you see a water strider, remember the invisible forces at play, the cohesive dance of molecules creating an elastic skin. It’s a testament to the power of physics, working in silent, elegant ways. And who knows, understanding these principles might just inspire your next backyard science experiment, proving that learning about the world around us can be incredibly fun and rewarding.
