Which Starch Solution Will Decrease In Volume As Osmosis Occurs
Hey there, starch lovers! Ever wondered what happens when your favorite starchy goodies get a little… thirsty? We’re talking about things like fluffy mashed potatoes, chewy bread, and maybe even a sweet corn on the cob. These guys are packed with something called starch, which is like a tiny, energy-packed treasure chest for our bodies. But here’s a fun little secret: starch isn't always a lone wolf. It often hangs out with its buddy, water. And when these two get together, some pretty cool stuff can happen, especially when we introduce them to a new environment.
Imagine you’ve just made a big ol’ pot of mashed potatoes. They’re steaming, they’re creamy, and they’re calling your name. Now, what if we were to take some of those glorious mashed potatoes and put them in a special kind of a situation? Not boiling water, not just sitting on the counter, but something a bit more… persuasive. Think of it like this: imagine your mashed potatoes are at a party, feeling all nice and cozy. But then, someone starts playing really loud, really tempting music from outside the party room. What do you think might happen?
Well, believe it or not, some starch solutions are a bit like those partygoers. They have a natural tendency to want to reach a kind of balance, a state of sweet, sweet harmony. And when they’re surrounded by something that has a lot more water – like, imagine a whole swimming pool compared to a tiny puddle in your mashed potatoes – the water from the swimming pool starts to get a little jealous of the water inside the starch. It wants to join the party, so to speak!
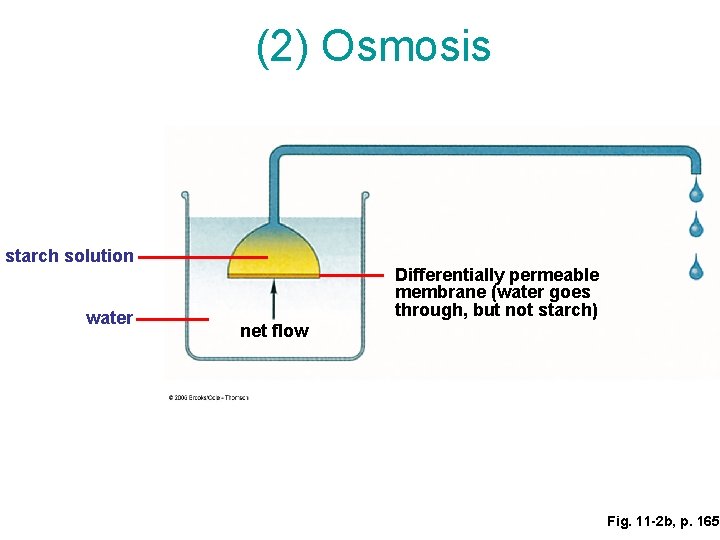
This is where things get really interesting, and frankly, a little bit magical. We’re talking about a process called osmosis. Don't let the fancy name scare you! It's just a fancy word for water being a bit of a nosy neighbor, moving from where there’s a ton of it to where there’s a little less, especially if there’s something really interesting on the other side, like our starchy friends. It’s like water sees the starch and thinks, "Ooh, that looks like fun! I'm going in!"
Now, not all starch solutions are created equal when it comes to this water-gathering party. Some are super eager to soak up water, like a brand new sponge. Others are a bit more reserved, like someone who prefers to watch the party from the sidelines. But the really surprising part is that some starch solutions, when faced with a situation where there’s way more water outside than inside them, can actually decide to shrink. Yes, you heard that right. They might actually decrease in volume!

Think about it. You’d expect more water to go in, right? Making them bigger, plumper, more delicious. But in certain very specific circumstances, the opposite can happen. It’s like the starch itself is saying, "You know what? All this extra water out there is just too much. I’m going to hold onto what I’ve got and maybe even let some of my own water go, just to keep things calm and collected." It’s a starch’s way of setting boundaries, perhaps? It’s a surprisingly self-preserving move for something we usually associate with puffing up and becoming soft and yielding.
This isn't about soggy bread or mushy potatoes turning into tiny little pebbles, mind you. It's a subtle shift, a gentle dance of water molecules. But the fact that it can happen at all is pretty mind-boggling. It's a little reminder that even the most familiar things in our kitchens have hidden depths and unexpected behaviors.

So, the next time you’re enjoying a meal rich in starch – perhaps a comforting bowl of rice, a slice of freshly baked bread, or even a bite of a starchy root vegetable – take a moment to appreciate the invisible ballet happening within. There’s a whole world of water-moving, volume-changing magic going on, and some of those starchy heroes are surprisingly good at holding their ground, even shrinking back to protect their own inner peace. It's a little bit of science that adds a whole lot of wonder to our everyday plates.
It’s a testament to the fascinating, often invisible, world that exists within the foods we love. Who knew that something as simple as starch and water could have such a dramatic, albeit subtle, story to tell? It makes you wonder what other secrets are hiding in your pantry, just waiting for the right conditions to reveal themselves. Maybe your flour is secretly a master negotiator, or your pasta is a shrewd water manager. The possibilities, much like the water molecules, are endless!
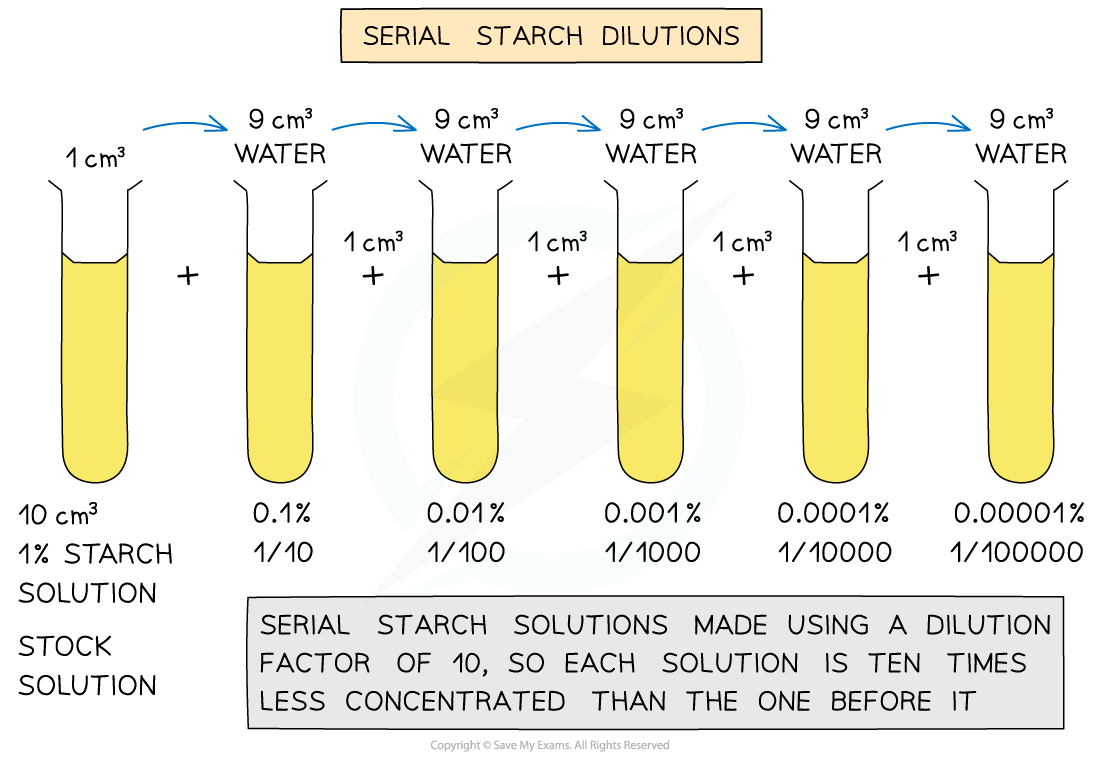
"It’s a little reminder that even the most familiar things in our kitchens have hidden depths and unexpected behaviors."
And the starch solution that decreases in volume as osmosis occurs? Well, it’s not the one you might first expect. It’s the one that, when faced with an overwhelming amount of external water, decides to get a little more compact, a little more… self-contained. It’s the quiet achiever of the starch world, the one that reminds us that sometimes, less really can be more, especially when it comes to water management.
So, next time you see those starchy staples, give them a little nod of appreciation. They’re not just filling our bellies; they’re performing miniature scientific miracles, all while looking delicious. Isn't that something to smile about?
