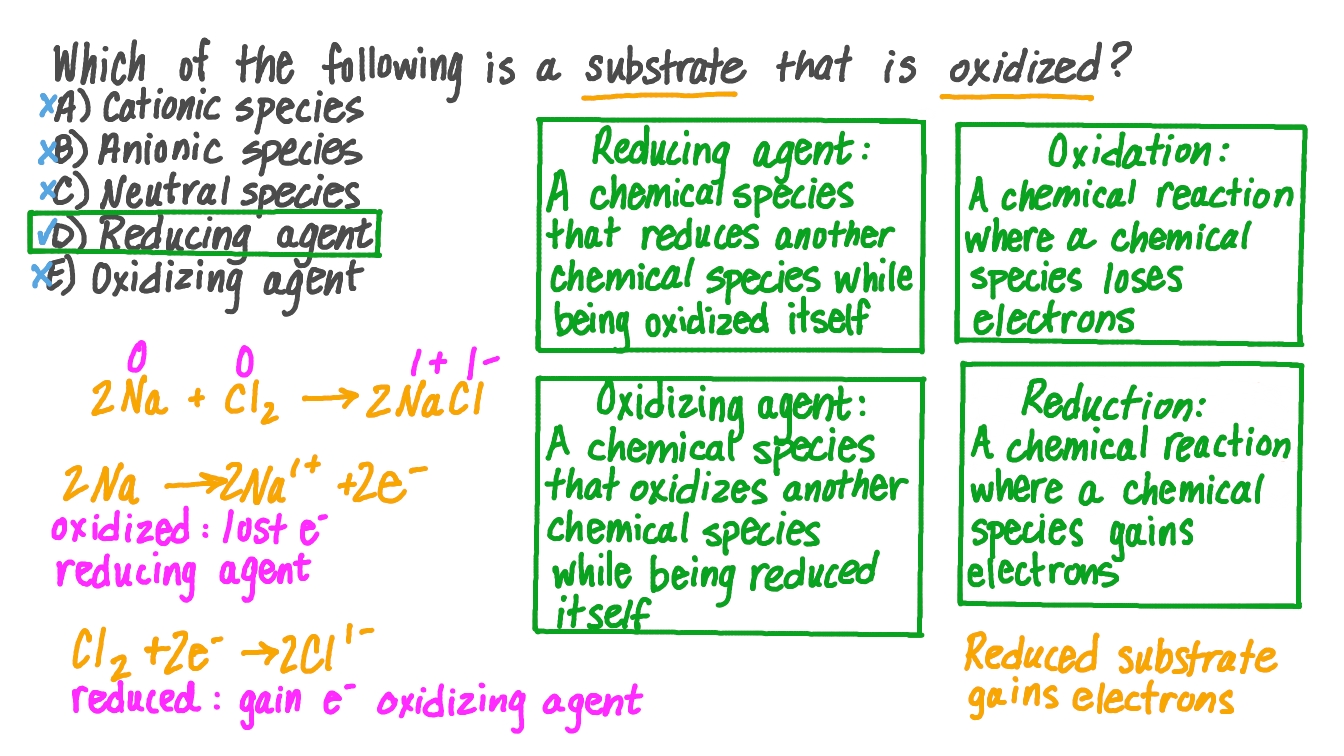
Which Species Is Oxidized When The Switch Is Closed
Ever wondered about those little switches that control so much of our modern world? They seem so simple, right? Just a flick, and poof, something happens! But behind that satisfying click is a miniature drama playing out, a tiny battle of the elements. And when you close that switch, a fascinating change occurs. It’s all about something called oxidation. Sounds a bit science-y, but stick with me, it's actually super cool.
Imagine a microscopic stage. On this stage, we have our main players. When the switch is open, they're all minding their own business, doing their own thing. But then, clack! The switch closes, and the spotlight hits. Suddenly, one of our players is thrust into a starring role. This is where the magic happens, or rather, the chemical reaction. We're talking about a specific kind of transformation, a bit like when iron rusts, but happening way faster and in a much more controlled environment.
So, which species gets to be the star of this show? It's all about who's ready to give up a little something. In the world of electricity and switches, we often have two main characters: a metal and something that can carry the electric flow. Think of it like a relay race. When the switch is closed, it's like the baton being passed. And in this pass, one participant is essentially giving away an electron, a tiny particle with a negative charge.
This giving away of an electron is what we call oxidation. It's not a dramatic explosion, but a subtle, yet significant, chemical change. The species that loses an electron becomes, well, oxidized. It’s like it’s undergone a tiny makeover, changing its chemical identity. And the species that receives that electron? That's called reduction. It’s a dynamic duo, always working together. One oxidizes, the other reduces. They're the ultimate chemical tag team.
Now, why is this so entertaining? Because it’s happening constantly, all around us, in devices we use every single day! From the tiny circuits in your phone to the lights in your room, this dance of oxidation and reduction is the unsung hero of modern technology. It’s the silent engine driving our digital lives. And when you close that switch, you’re not just completing a circuit; you’re initiating this fascinating chemical transformation.

Think about the sheer elegance of it. A simple mechanical action – the closing of a switch – triggering a precise chemical reaction. It’s a beautiful symphony of physics and chemistry, all happening at the speed of light. And the best part is, it’s not some abstract concept confined to a lab. It’s tangible. You can see the results of this process every time you turn on a light, send a text, or scroll through your feed. That little flicker, that responsive screen – that's the work of oxidation and reduction.
It's like a microscopic stage where elements perform a dazzling chemical ballet, all triggered by the simple flick of a switch!
The star of this particular show, the one that gets oxidized when the switch is closed, is usually the metal that's part of the conductive pathway. Metals are often keen to lose electrons. They’re like generous givers in the electron economy. When the electrical pathway is completed, they’re perfectly positioned to make that donation. It’s their natural inclination, their chemical destiny in that moment.

What makes it special? It’s the fundamental building block of so much of what we take for granted. Without this process, our electronics wouldn’t work. Imagine a world without electricity! It’s hard to even fathom. This seemingly simple act of closing a switch is the key that unlocks a universe of possibilities, all thanks to the willing sacrifice of an electron by a metal species.
The beauty lies in its universality. This principle applies across a vast range of applications. Whether it's a simple battery powering a toy or a complex industrial circuit, the underlying chemistry remains the same. It's a testament to the elegance and efficiency of natural laws. They’re simple at their core, but their implications are monumental.
And the species that gets reduced? That's the electron acceptor, the one that’s happy to take on that extra electron. Often, this is another component within the circuit that’s designed to readily accept electrons. It’s a perfect partnership, a give-and-take that keeps everything running smoothly. It’s the chemical equivalent of perfect teamwork.
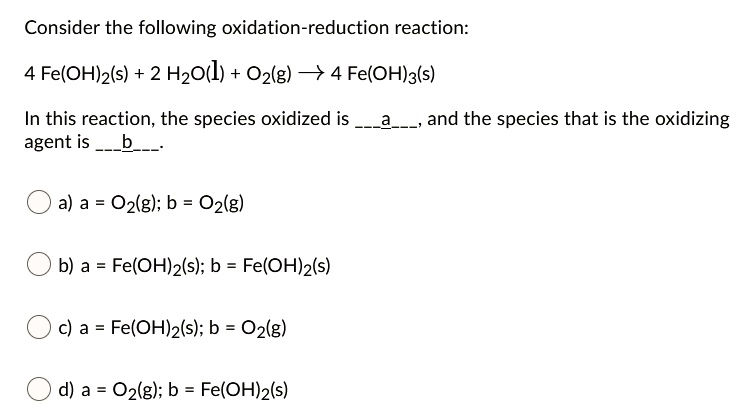
So, the next time you flick a light switch, take a moment. Appreciate the silent, unseen drama that unfolds. A metal species, perhaps something like copper or silver, is stepping into the spotlight. It’s giving up an electron, undergoing oxidation, and enabling the flow of electricity. It’s a small act with huge consequences.
It's this fundamental interplay that makes our modern world possible. From the tiniest transistor to massive power grids, the principles of oxidation and reduction are at play. It's not just about making things work; it's about understanding the elegant dance of atoms and electrons that powers our lives.
And it's incredibly entertaining because it’s so vital and yet so invisible. We interact with its effects constantly, but rarely do we consider the intricate chemical ballet happening beneath the surface. It’s the ultimate hidden magic show, powered by the willingness of certain elements to share their electrons.

The species that gets oxidized is essentially the one that’s ready and willing to let go of an electron when the circuit is completed. It’s a chemical predisposition, a characteristic of that particular element or compound. Think of it as its personality trait in the context of an electrical circuit.
It’s this inherent property of certain materials, their readiness to participate in redox reactions, that makes them so valuable in electronics. They are the willing participants in the energy transfer that lights up our lives and connects our world. It's a constant, reliable performance that we often overlook.
So, if you’re curious about the hidden workings of the world, about the tiny, invisible processes that make our technology tick, then this is a great place to start. It's a peek behind the curtain, a glimpse into the fascinating chemical universe that surrounds us. The next time you see a switch, remember the little chemical drama playing out, and the star species that gets oxidized to make it all happen!
