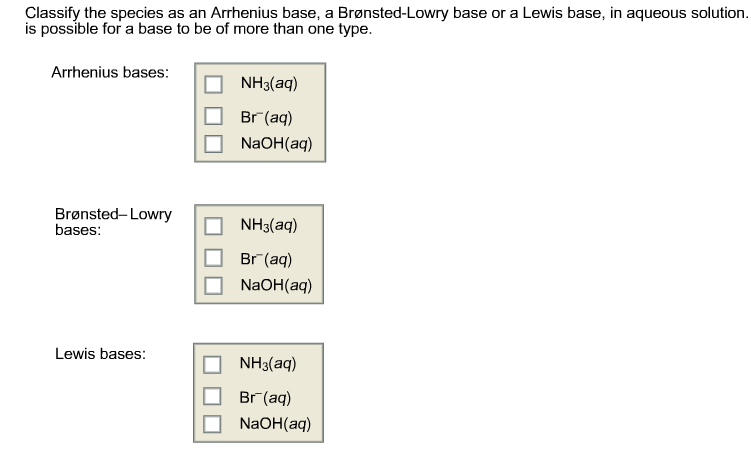
Which Species Is Classified As An Arrhenius Base
Ever wondered why some cleaning products are so good at tackling grease, or what makes antacids work so quickly? It all boils down to some fascinating chemistry, and a core concept we can explore together: Arrhenius bases. Now, don't let the fancy name scare you off! Thinking about which species fall into this category is actually a bit like being a detective, uncovering the hidden properties of everyday substances. It's fun because it demystifies the world around us, helping us understand the "why" behind so many things we encounter.
So, what exactly is an Arrhenius base, and why should we care? In essence, an Arrhenius base is a compound that, when dissolved in water, increases the concentration of hydroxide ions (OH⁻). Think of hydroxide ions as tiny, reactive particles that have a particular set of skills. They're the reason bases feel slippery, taste bitter (though it's never safe to taste chemicals!), and can neutralize acids. The purpose of understanding Arrhenius bases is fundamental to chemistry. It's a cornerstone for comprehending chemical reactions, particularly acid-base reactions, which are incredibly important in everything from biological processes within our bodies to industrial manufacturing.
The benefits of knowing about Arrhenius bases extend far beyond a textbook. In education, it's a vital stepping stone for students learning chemistry. Understanding this concept opens doors to comprehending more complex topics like pH, buffers, and titrations. In our daily lives, this knowledge helps us make informed choices. For instance, knowing that ammonia (NH₃) is an Arrhenius base helps explain why it's a powerful cleaning agent for certain surfaces. Similarly, understanding that magnesium hydroxide (Mg(OH)₂) is an Arrhenius base explains why it's a common ingredient in over-the-counter antacids, helping to soothe indigestion by neutralizing stomach acid.
The beauty of exploring Arrhenius bases lies in its accessibility. You don't need a full-fledged laboratory to start learning! A simple and safe way to explore this is by observing common household items. For example, you might notice how baking soda (sodium bicarbonate, NaHCO₃) can react with vinegar (acetic acid) to create fizzing – this is an acid-base reaction in action! While baking soda isn't a strict Arrhenius base in its pure form (it needs water to release hydroxide ions, or it can react with water to produce them indirectly), its behavior illustrates the principles. You can also look at the ingredients lists of cleaning products; you might spot compounds containing hydroxide ions, hinting at their basic nature. For those curious to delve a bit deeper, readily available litmus paper can be a fun tool. You can test the pH of various safe solutions (like diluted vinegar or baking soda solutions) to see how they react – acids turn blue litmus paper red, while bases turn red litmus paper blue. It's a visual and engaging way to see chemistry at play right in your own home!
