Which Solution Will Have The Highest Boiling Point

Hey there, science curious pals! Ever wondered about those little bubbles that pop up when you’re boiling water? Or maybe you've seen a science show and thought, "Whoa, what's going on there?" Well, today we're diving headfirst into the wonderfully wacky world of boiling points. And trust me, it’s way more interesting than you think!
So, what even is a boiling point? Think of it as the party trick of a liquid. It’s the exact temperature where a liquid decides, "Okay, I’m done being a liquid, it’s time to become a gas!" It’s like the liquid’s big moment to break free and float around. And different liquids? They have different party times. Some are eager to get going, others are a bit more… chill.
We’re gonna play a fun game today: "Who's Got the Hottest Boiling Point?" Imagine we have a lineup of common solutions, like those you might find in your kitchen or a science lab. We're gonna guess which one will throw the biggest boiling party, meaning it needs the most heat to turn into a gas.
The Contenders: A Quirky Crew!
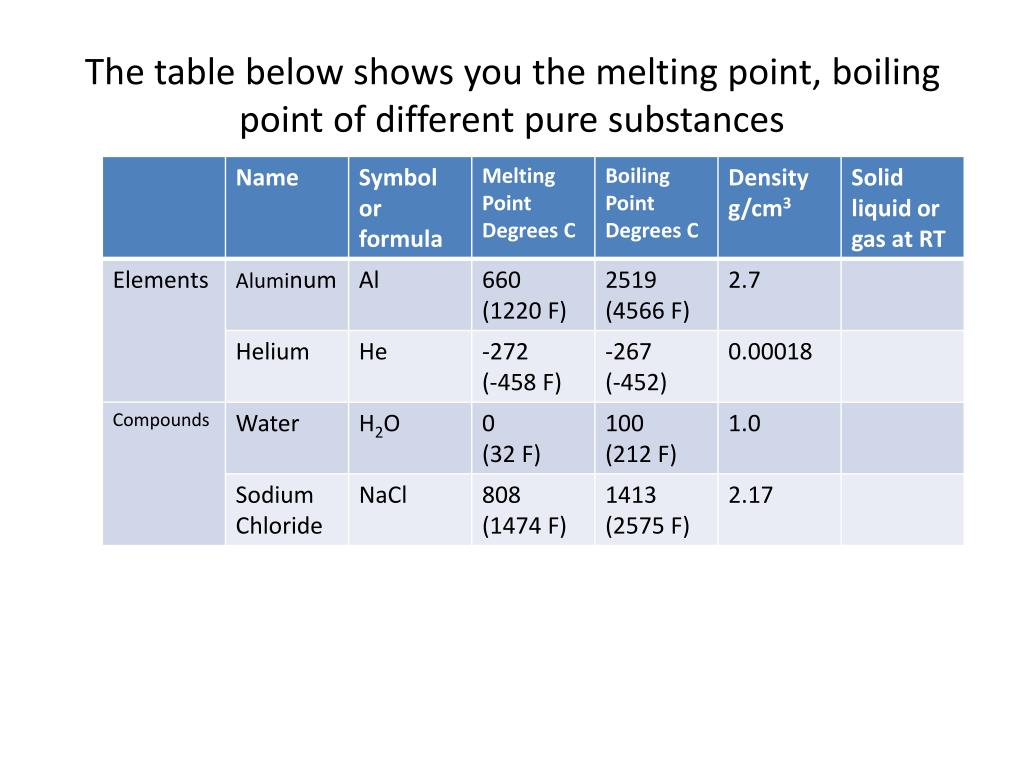
Let’s meet our contestants! We've got the everyday hero, plain old water. Boring? Maybe. But it’s our baseline, our starting point. Water boils at a nice, round 100 degrees Celsius (or 212 Fahrenheit). Pretty standard, right? Like that friend who’s always on time.
Then we have saltwater. Now, this is where things get spicy! What happens when you dissolve salt in water? Does it make the party harder to start, or easier? It’s like adding extra guests to the party. Do they make it more fun or more crowded and chaotic?
Next up, the sweet stuff: sugar water. Similar to salt, sugar dissolves. But is sugar just like salt in terms of boiling? Are all dissolved things created equal? This is where the mystery deepens, my friends.
And let's throw in something a little more… oily. How about vegetable oil? Now, oil and water don’t mix. This is a whole different ball game. Does it play by the same rules? Or does it do its own thing?
Finally, let’s add a wild card. What about something with alcohol, like rubbing alcohol (isopropyl alcohol)? This stuff evaporates super fast. Does that mean it boils at a lower temperature? The plot thickens!
The Science Shenanigans: Why the Fuss?
So, why do these liquids have different boiling points? It all comes down to the tiny, invisible forces holding them together. Think of the liquid molecules as little magnets, holding hands. To boil, they need enough energy (heat!) to break free from their buddies and become independent gases.
Intermolecular forces, that’s the fancy term. It’s like how strong are those handshakes between the molecules? Stronger handshakes mean more energy (heat!) is needed to pry them apart. Weaker handshakes? Easier to break free, lower boiling point!
Now, when you add something like salt or sugar to water, you're not just adding stuff. You're actually changing the concentration of those little handshakes. It’s like adding more people to the dance floor. It can get a bit more crowded, making it harder for the water molecules to escape.

Saltwater vs. Sugar Water: The Dissolved Drama!
Let’s zoom in on saltwater and sugar water. When salt (like sodium chloride) dissolves, it splits into ions – positively and negatively charged bits. These ions are like super-sticky magnets that really hold onto the water molecules. They create a stronger overall "stickiness" in the solution. This means more energy is needed to overcome these interactions and turn the water into a gas.
Sugar, on the other hand, dissolves as whole molecules. It still forms those hydrogen bonds with water, which increases the stickiness. But generally, salt has a bigger impact on raising the boiling point than sugar. Why? Because salt breaks down into more individual particles (ions) than sugar does (which stays as whole molecules). More particles, more interactions, higher boiling point!
So, if we had to pick between plain saltwater and plain sugar water (with the same amount of dissolved stuff, which is tricky to compare directly!), the saltwater would likely have a higher boiling point. It’s the salty underdog winning the heat competition!
Oil: The Odd One Out
Vegetable oil is a whole different beast. It’s made of long hydrocarbon chains. These molecules are held together by weaker forces (van der Waals forces) compared to the strong hydrogen bonds in water. This means oil molecules don’t hold onto each other as tightly. So, you might think it would have a lower boiling point, right? Wrong!
Oil itself has a much higher boiling point than water. It needs a ton more energy to vaporize. However, the question is about the solution. If we were comparing pure vegetable oil to pure water, the oil would win the high boiling point race by a landslide. But when we talk about a "solution," we usually mean something dissolved in a solvent, often water. So, if we're talking about oil as the solvent, it's a different story. But typically, when we talk about "solutions" in this context, water is the solvent.

The key takeaway here is that the type of substance dissolved and how it interacts with the solvent are super important. And for our typical "solutions" in everyday life, water is usually the star solvent.
Rubbing Alcohol: The Evaporation Expert
Now for our wild card, rubbing alcohol (isopropyl alcohol). This stuff feels "colder" to the touch because it evaporates so quickly. Evaporation is related to boiling, but they aren't the same thing! Evaporation happens at the surface, even below the boiling point.
Isopropyl alcohol actually has a lower boiling point than water – around 82.5 degrees Celsius (180.5 Fahrenheit). So, it’s the early bird of the boiling world, ready to break free much sooner than water. It’s like the friend who’s always ready to leave the party.
So, Who Wins the Boiling Point Battle?
Drumroll, please! If we’re comparing the common solutions we’ve talked about, and assuming we’re talking about dissolving things in water:

The solution that will likely have the highest boiling point is the one with the dissolved substances that create the strongest interactions and require the most energy to overcome. Between salt and sugar dissolved in water, saltwater is usually the winner for a higher boiling point.
Why? Remember those ions? Salt breaks into more charged particles than sugar. These ions pull strongly on water molecules, making it much harder for the water to escape into the gas phase. You need to crank up the heat significantly to get that salty water to boil compared to plain water.
The Quirky Science of Everyday Life
Isn’t it fascinating how adding a pinch of salt can change something as fundamental as boiling water? It’s a small change with a big effect. This is called a colligative property – a property of a solution that depends only on the number of solute particles, not on their identity. The more particles you add, the higher the boiling point goes!
Think about cooking. When you add salt to pasta water, it doesn’t just make it taste better; it slightly raises the boiling point. This means the water might be a tiny bit hotter when your pasta is cooking. It’s not a huge difference, but it’s there!
So, the next time you’re in the kitchen, or you see something boiling, remember the invisible dance of molecules and the power of dissolved stuff. It’s a little piece of science hiding in plain sight, making everyday life a bit more interesting. Keep asking questions, keep wondering, and keep exploring the wonderfully weird world of science!
