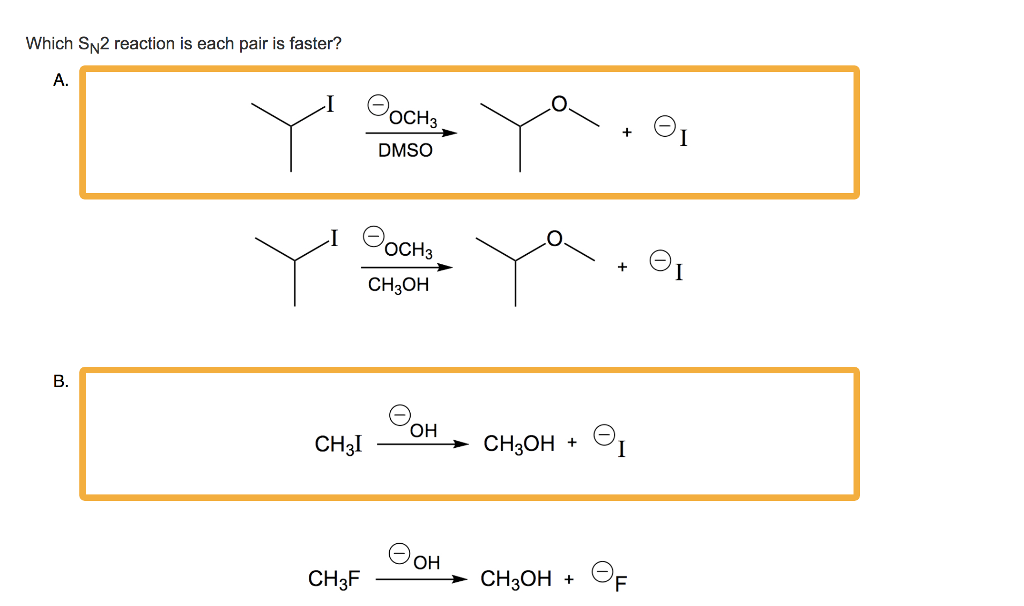
Which Sn2 Reaction In The Following Pair Is Faster
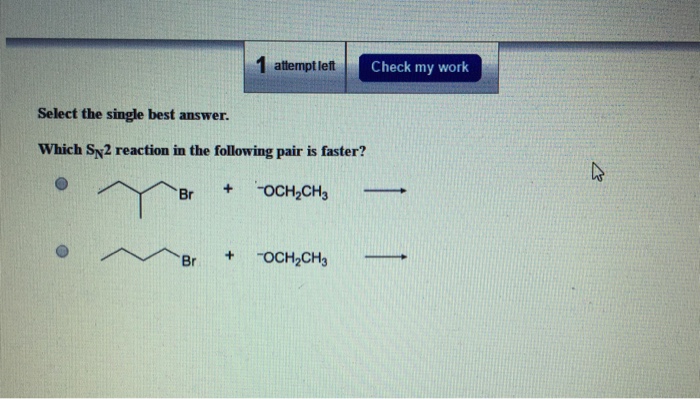
Imagine you’ve got two identical boxes of cookies, right? And you’ve got a little helper who absolutely loves to swap things out. Our little helper has two different jobs to do, and we want to know which job will get done quicker. It’s a bit like a race, but instead of speed, we’re looking at how easily our little helper can get the job done.
In the world of tiny molecules, we have something similar happening called an SN2 reaction. Think of it as a molecular dance where one little piece, let’s call it the nucleophile (our cookie-loving helper!), comes in and nudges another little piece, the leaving group (the bit that needs to go), right off a central molecule (our cookie itself!). The whole point is that the nucleophile is super keen to attach itself, and the leaving group is just waiting for a good reason to pack its bags and leave. It’s a graceful, one-step process where the nucleophile arrives just as the leaving group departs. No waiting in line, no awkward goodbyes, just a swift, decisive exchange!
Now, we have a bit of a playful challenge. We have two scenarios, two different sets of molecules trying to play this SN2 game. Let’s meet our contenders! On one side, we have methyl iodide. Think of this as a super-simple cookie – a small central carbon atom with a few tiny hydrogen bits, and then a rather large, iodine atom hanging on. Iodine, bless its heart, is a bit of a drama queen. It’s not very tightly attached, and it’s always looking for an excuse to leave. This makes it a fantastic leaving group. On the other side, ready to swoop in, is our trusty nucleophile, let’s say it’s a happy little hydroxide ion – a bit of oxygen with a hydrogen and a negative charge, eager to join the party.
In our first race, the hydroxide ion has to deal with methyl iodide. It’s like our cookie helper approaching a very neatly packaged, small cookie with a loosely attached bow. The hydroxide ion can easily get to the carbon atom from the back, giving that iodine a firm nudge. Because the carbon atom is so small and uncluttered with the tiny hydrogens, there’s plenty of room for our nucleophile to do its work. It’s like trying to unwrap a small present with a ribbon that’s already coming undone – pretty straightforward, wouldn’t you say?
Now, let’s introduce our second contender: tert-butyl bromide. This one is a bit more… complex. Imagine our cookie is now a giant, frosted cake, piled high with three fluffy layers of meringue on top of our central carbon. Instead of tiny hydrogens, we have these rather chunky methyl groups taking up a lot of space. And our leaving group here is bromide, which is pretty similar to iodine in its eagerness to depart. But here’s the catch. Our little hydroxide ion, our cookie helper, has to navigate this molecular obstacle course! It needs to get to the carbon atom to do its swapping, but oh boy, those bulky methyl groups are like bouncers at a very exclusive party, standing in the way.
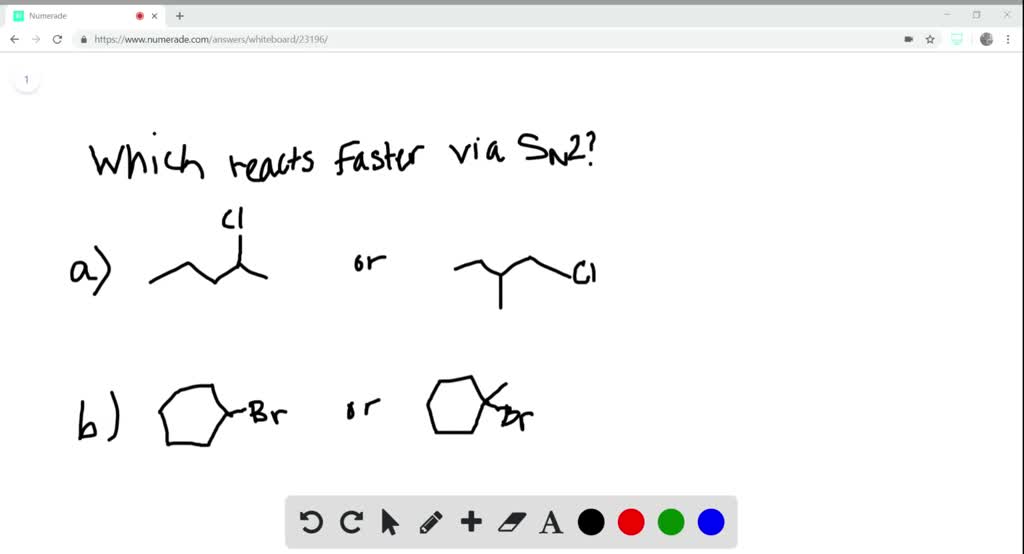
So, in our second race, the hydroxide ion approaches tert-butyl bromide. It’s like our cookie helper trying to get to that loosely attached bow on our giant cake, but there are three huge piles of frosting and decorations blocking the path. The hydroxide ion tries to sneak around, but it’s a tight squeeze! It’s like trying to get a single finger through a very crowded, bustling market. There’s just not enough space for our nucleophile to get a good grip and give that bromide the nudge it needs. The leaving group might be ready to go, but the path to the action is just too congested.
The bigger the crowd around the dance floor, the harder it is for new partners to join the fun!
Solved Which SN2 reaction is each pair is faster? A. OCH3 | Chegg.com
So, when we compare these two scenarios, the first one – the race involving methyl iodide – is going to be the clear winner. Our cookie helper, the hydroxide ion, has a much easier, more direct route to do its swapping. There are no bulky distractions, no molecular traffic jams. It’s a smooth, efficient transaction.
The second scenario, with tert-butyl bromide, is a bit of a mess. Our nucleophile is essentially fighting for space. It’s like trying to perform a delicate operation in a cramped closet. Even though the bromide is happy to leave, the sheer physical obstruction of those bulky groups around the carbon atom makes the SN2 reaction incredibly slow, almost to a standstill. It’s the difference between a quick, efficient handshake and trying to high-five someone who’s wearing oven mitts and standing behind a sofa.
It’s quite fascinating, isn’t it? How the simplest change – the size and arrangement of the bits around our central molecule – can make such a massive difference to how quickly a reaction can happen. It’s a little reminder that even in the microscopic world, space and accessibility are everything. It’s a tiny drama playing out, a race against crowding, where the less cluttered molecule always wins the speed round. And sometimes, the simplest molecules are the ones that can perform their little chemical dances with the most effortless grace!