Which Shows The Electron Configuration For Zinc Zn
Imagine you're at a bustling city fair, and you spot a row of prize booths. Each booth has a different game, and each game requires a specific set of tokens to play. Now, think of an atom like a very tiny, very orderly fairground. The electrons, those zippy little particles, are like the players trying to get into the games (which we call orbitals). So, the electron configuration is basically the list of which games each electron is playing and how many are in each game.
Today, we're talking about Zinc (Zn), a metal you might know from things like galvanized nails or even that classic superhero movie. Zinc's electron configuration is like a secret handshake, a unique way its electrons arrange themselves. It's not as complicated as it sounds, but it does have a little quirk that makes it stand out from the crowd.
Let's pretend we have a special "Electron Lottery" for every atom. This lottery has different drawing rooms, each a bit like a different type of game. We have the 's' rooms, the 'p' rooms, the 'd' rooms, and the 'f' rooms. The 's' rooms are cozy, only holding two electrons. The 'p' rooms are a bit larger, holding six. The 'd' rooms are even bigger, holding ten, and the 'f' rooms are the biggest of all, fitting fourteen!
Now, these rooms aren't just filled randomly. There's a specific order in which they get filled, like a queue at the popular rollercoaster. Electrons generally prefer to fill the lower-energy rooms first, the ones that are easier to get into, before they start aiming for the pricier, higher-energy ones. It's all about efficiency, even for tiny particles!
So, for a plain old atom, you might see a configuration like "1s² 2s² 2p⁶...". This just means two electrons in the first 's' room, two in the second 's' room, and six in the second 'p' room. It's like saying, "Okay, the first two games are full, and the next two games are full too, and then we have six players in the third game."
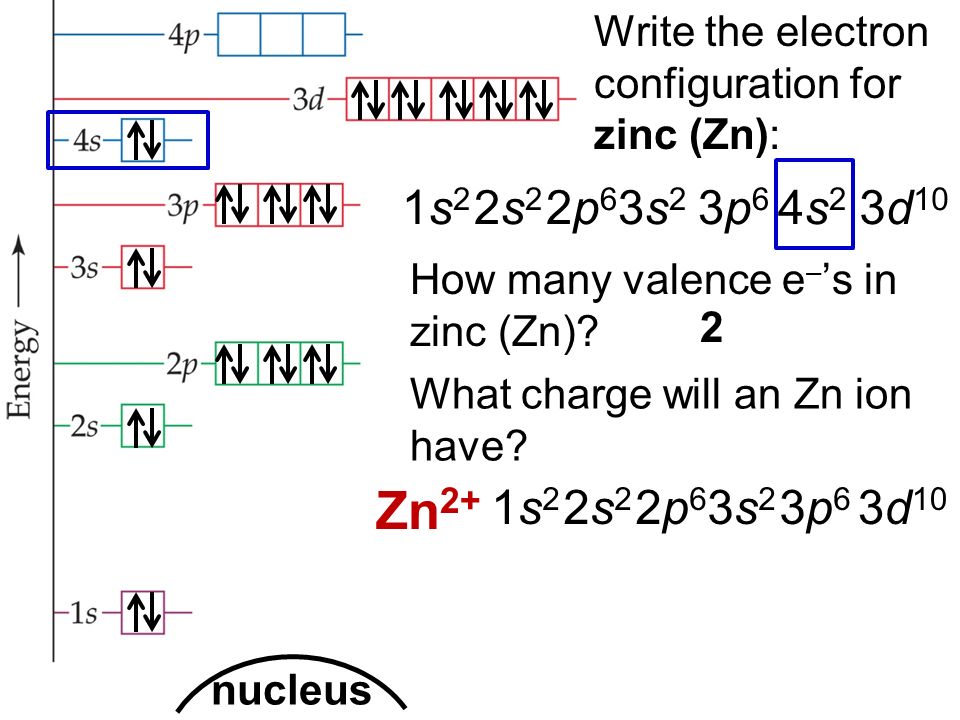
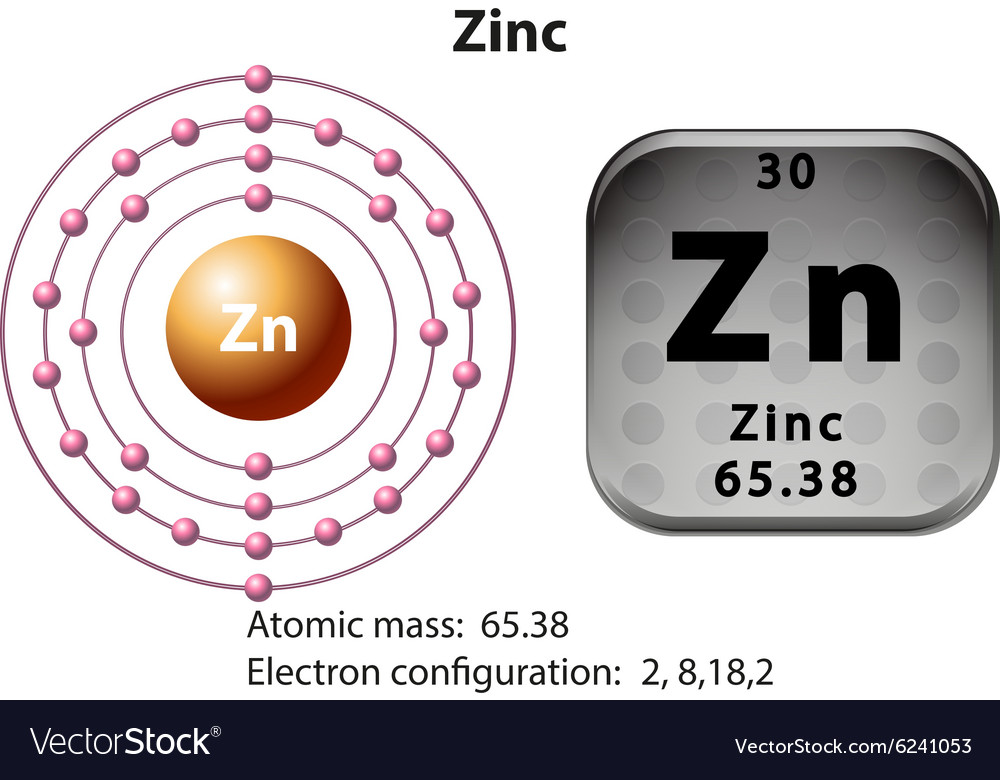
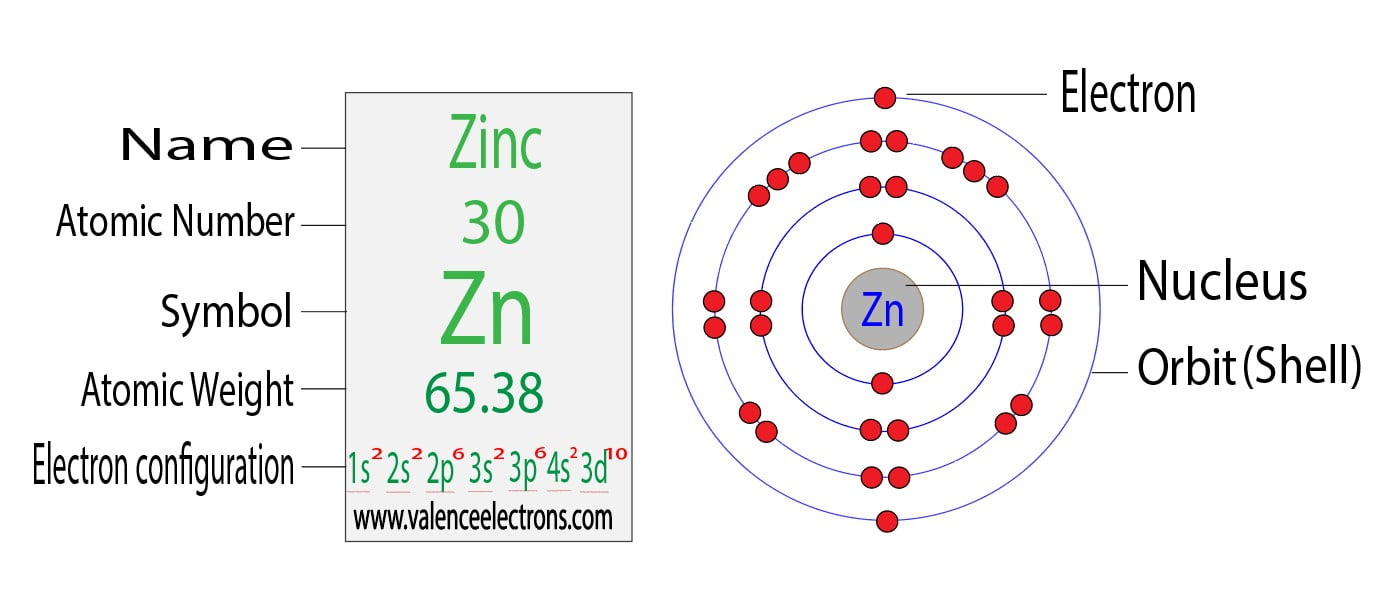
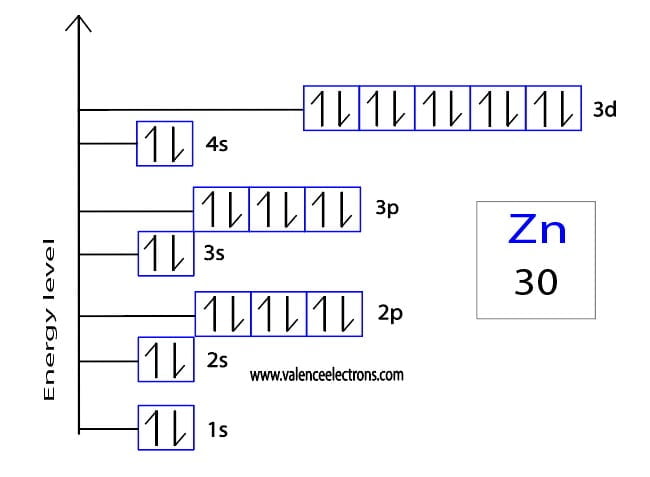
But Zinc (Zn), with its atomic number of 30, has 30 electrons to place. You might expect a straightforward filling of all the rooms up to its point. Following the usual order, we'd fill up to the '3d' orbitals. This would look something like [Ar] 4s² 3d¹⁰. Notice how the '4s' orbital gets filled before the '3d' orbital, even though '3d' has a lower number. That's the quirky rule of filling!
The exciting part about Zinc is what happens in those 'd' orbitals. Unlike many other elements, where the 'd' orbitals might be only half-filled or have some empty spaces, Zinc's '3d' orbitals are completely full. They've got all 10 electrons they can hold, all snug and happy.

Think of it like a professional athlete training for a championship. They meticulously fill their training schedule, making sure every practice session, every workout is accounted for. Zinc's electrons do something similar. They fill up the '4s' orbitals, and then they go and perfectly fill the '3d' orbitals.
This full '3d' shell is like a perfectly balanced meal or a flawlessly executed dance routine. It's incredibly stable. This stability is why Zinc behaves the way it does in chemical reactions. It doesn't get as excited to give away or gain electrons because its electron arrangement is already so content.
Now, if you were to write out the entire electron configuration for Zinc without using the shorthand (the [Ar] part), it would be a bit of a mouthful. You'd see: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰. That's a lot of numbers and letters, like a long, complicated recipe.
But the important thing to remember is that the last part, the 3d¹⁰, is the star of the show for Zinc. It's what makes it special. It's like a musician nailing every single note in a difficult passage of music; it's a sign of mastery and perfection.
This perfect filling of the '3d' orbitals makes Zinc quite unique in the periodic table. It means that when you see Zinc in a chemical equation, you can often predict its behavior with a bit more certainty. It's not prone to the wild swings of reactivity that some other elements might be.
It's almost like Zinc is the reliable friend at the party. While others might be jumping from one game to another, changing their minds, and getting into all sorts of kerfuffles, Zinc is calmly sitting back, its electrons perfectly arranged, its '3d' room full and content.
So, next time you encounter Zinc, whether it's in a practical application or just as a name in a chemistry textbook, remember its electron configuration. Think of those perfectly filled '3d' orbitals as the reason for its quiet strength and stability. It's a beautiful example of how even the tiniest, most invisible parts of matter have their own intricate stories to tell.
The way electrons arrange themselves in atoms is a fundamental concept in chemistry. For Zinc, the configuration [Ar] 4s² 3d¹⁰ is a testament to this order. It shows us that even in the seemingly chaotic world of atoms, there's a profound sense of balance and predictability.
It’s not just about memorizing symbols; it’s about understanding the underlying logic. The electron configuration for Zinc, specifically the complete 3d¹⁰ shell, is a perfect illustration of this. It's a tiny, atomic achievement.
Think of it as Zinc's atomic superpower: a full and stable outer electron shell. This makes it less likely to react explosively or unpredictably. It's the quiet confidence of a well-prepared individual.
The simplified notation [Ar] 4s² 3d¹⁰ tells a story of electron arrangements. It highlights how the '4s' orbitals are filled first, and then the '3d' orbitals are completed to their maximum capacity. This is a key characteristic of Zinc.
So, while other elements might be experimenting with half-filled orbitals, Zinc is like the student who has already mastered the entire chapter and is now leisurely reviewing the material. Its electron configuration is a sign of that mastery.
The complete filling of the '3d' orbitals gives Zinc a certain inertness compared to its neighbors. It’s not eager to gain or lose electrons, making it a stable and predictable element.
This stability is what makes Zinc so useful in many applications. Its reliability stems directly from this electron configuration, a testament to nature’s elegant design.
When you see the electron configuration for Zinc, you are looking at a snapshot of its atomic personality. It’s a picture of perfect electron harmony.
The story of Zinc's electron configuration is a story of stability, order, and a touch of atomic elegance. It reminds us that even in the smallest building blocks of the universe, there's a captivating order waiting to be understood.
The notation [Ar] 4s² 3d¹⁰, far from being just a jumble of letters and numbers, is a coded message revealing the stable heart of the Zinc atom. It's a beautiful secret shared by the element itself.
Understanding this configuration allows us to appreciate why Zinc acts the way it does in the grand theatre of chemistry. It’s all about that perfectly full 3d shell!
