Which Set Of Quantum Numbers Cannot Specify An Orbital
Okay, so picture this: I was crammed into my tiny dorm room, textbooks piled higher than my ambition on a Monday morning, staring at a particularly gnarly chemistry problem. It involved electron configurations, quantum numbers, the whole nine yards. I was convinced I had it all figured out, you know? Like, I'd meticulously calculated each number, felt all smug and victorious. Then, my professor, bless his patient soul, walked by, peeked at my scribbled answer, and just chuckled. A gentle, knowing chuckle. "Ah, trying to define an orbital with that set, are we?" he said, a twinkle in his eye. I felt my face get hot. Turns out, I’d accidentally created a chimera of quantum numbers, something that looked plausible but was, in reality, utterly impossible. It was like trying to describe a unicorn by listing the average weight of a horse and the wing span of a pigeon. Makes about as much sense. And that, my friends, is precisely what we're diving into today: which set of quantum numbers simply cannot specify an orbital.
It’s a bit of a mind-bender, isn’t it? We’re taught these four quantum numbers – the principal (n), the azimuthal or angular momentum (l), the magnetic (ml), and the spin (ms) – as the ultimate identifiers for an electron’s state within an atom. They’re like an electron’s social security number, detailing its energy level, the shape of its "neighborhood" (orbital), its orientation in space, and its intrinsic spin. Pretty neat, right? But just like not every combination of digits makes a valid social security number (thank goodness, otherwise we’d have a real mess on our hands!), not every combination of quantum numbers can describe a real, existing orbital.
So, what’s the culprit? The one set of numbers that throws a wrench in the works? It all boils down to a fundamental relationship between two of our quantum buddies: the principal quantum number (n) and the azimuthal quantum number (l). You see, these two are practically joined at the hip. Their relationship isn't just a suggestion; it's a strict rule, a cosmic decree within the quantum realm.
The Unbreakable Bond: n and l
Let's break down what each of these numbers actually represents, just to refresh our memories. Think of the principal quantum number, n, as the main energy shell. It tells you how far, on average, the electron is from the nucleus. Higher n means higher energy and, generally, a larger spatial distribution. So, n = 1 is the innermost shell, n = 2 is the next one out, and so on. Easy peasy, right? Like floors in a building.
Then we have the azimuthal quantum number, l. This one’s a bit more descriptive. It tells us about the shape of the orbital within a given energy shell. It dictates the subshells. For a given value of n, l can take on integer values from 0 up to n-1. This is where the magic, and the potential for error, really starts to happen.
So, if n = 1, l can only be 0. This corresponds to the s subshell, which has spherical orbitals. If n = 2, l can be 0 (the 2s subshell, also spherical) or 1 (the 2p subshell, which has those dumbbell shapes). See the pattern? For n = 3, l can be 0 (3s), 1 (3p), or 2 (3d, with more complex shapes). It's always l = 0, 1, 2, ..., (n-1).
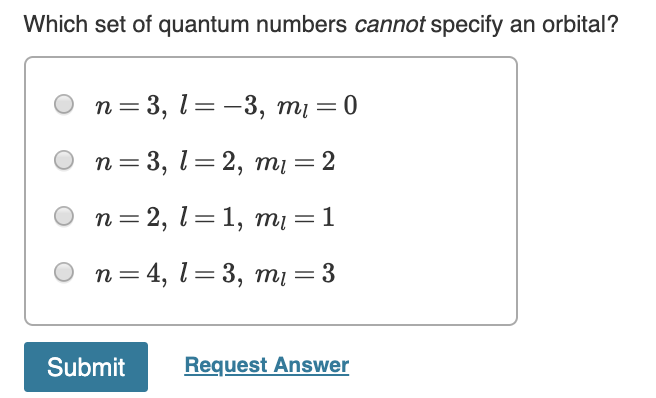
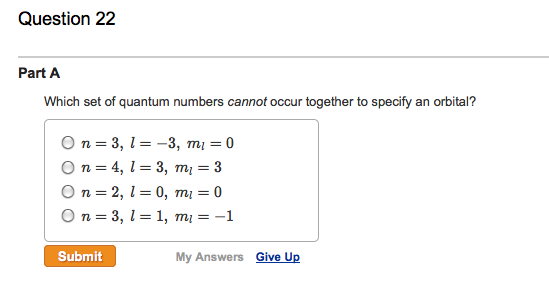
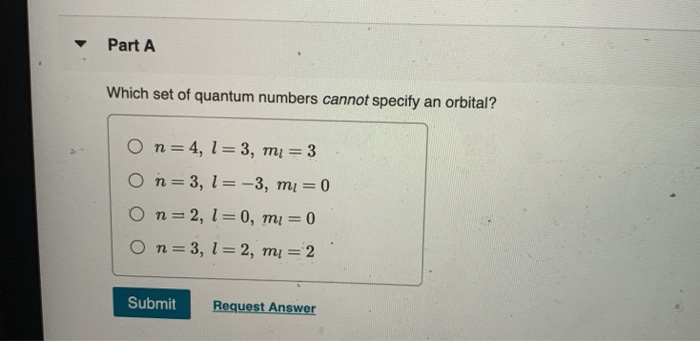
Now, here’s the crucial part: l can never be equal to or greater than n. Never. Not even once. If n represents the total number of shells available, and l describes the subshells within that shell, it's logically impossible to have more subshells than shells. Imagine trying to fit 5 books onto a shelf that can only hold 3. It just doesn't work. The universe, in its infinite wisdom (and mathematical elegance), has drawn a hard line here.
This relationship is so fundamental that if you try to propose a set of quantum numbers where l ≥ n, you're essentially describing a ghost orbital. It’s a configuration that doesn’t and can’t exist in any atom governed by quantum mechanics. It’s a non-starter, a cosmic faux pas.
The Other Players: ml and ms
Before we completely dismiss our impossible set, let's briefly touch upon the other two quantum numbers, just for completeness. The magnetic quantum number (ml) tells us about the orientation of the orbital in space. For a given value of l, ml can take on integer values from -l to +l, including 0. So, if l = 0 (an s orbital), ml can only be 0 (one orientation). If l = 1 (a p orbital), ml can be -1, 0, or +1 (three different orientations: px, py, pz). If l = 2 (a d orbital), ml can be -2, -1, 0, +1, +2 (five orientations).
And finally, the spin quantum number (ms). This one’s a bit quirky. It describes the intrinsic angular momentum of an electron, often visualized as its "spin" (though it's not a literal spinning ball). An electron can spin in one of two directions, conventionally represented as +1/2 (spin up) and -1/2 (spin down). This is what allows two electrons to occupy the same orbital without violating the Pauli Exclusion Principle.
These two, ml and ms, are dependent on l and the specific orbital respectively. They don't introduce the fundamental impossibility that the n-l relationship does. You can have valid combinations of n, l, and ml, and then assign spins to electrons within those orbitals. The real deal-breaker, the showstopper, is when n and l are at odds with each other.

Putting It All Together: The Forbidden Fruit
So, let’s construct an example of a forbidden set of quantum numbers. Remember our rule: l must be less than n.
Imagine a chemist, perhaps a bit too eager, declaring: "I’ve found an electron in a state described by n=1 and l=1!"
Immediately, a red flag should go up. Why? Because if n=1, then l can only be 0. The value l=1 is simply not allowed for n=1. It's like saying you have a third floor in a two-story building. It's a logical contradiction.
This specific combination, (n=1, l=1), therefore, cannot specify an orbital. There is no such orbital in any atom. It's a hypothetical construct that violates the fundamental rules of quantum mechanics governing atomic structure.
What about if we throw in ml and ms? Let’s say the hypothetical set is: n=1, l=1, ml=0, ms=+1/2. Does adding the other numbers make it valid? Nope! The initial violation of the n-l relationship is enough to render the entire set invalid for describing an orbital. The problem is at the very foundation.
It's like building a house: if your foundation is cracked, it doesn't matter how fancy your roof is; the whole structure is compromised. The n-l relationship is the bedrock of orbital definition.
Why Does This Rule Exist? The Deeper Dive (Optional, but Fun!)
For those of you who like to peek behind the curtain, why is this rule so strict? It stems from the mathematical solutions to the Schrödinger equation, the fundamental equation of quantum mechanics. When you solve the Schrödinger equation for an atom, the solutions (which describe the wave functions of electrons, i.e., the orbitals) naturally yield these quantum numbers.
The principal quantum number, n, emerges from the radial part of the solution, related to the energy. The azimuthal quantum number, l, emerges from the angular part of the solution, related to the shape. The mathematics dictates that for a given number of radial nodes (related to n), there are only a certain number of possible angular solutions (related to l).
Think of it like this: the 'n' value dictates the overall complexity or "size" of the wave function. The 'l' value dictates how that wave function behaves in terms of its angular distribution. You can't have more complex angular patterns than the overall complexity allowed by 'n' permits. It's an inherent constraint of how wave functions behave in three dimensions around a central potential (the nucleus).

So, the rule isn't arbitrary. It's a direct consequence of the physics that governs our universe at the atomic level. It’s elegant, really, how these abstract mathematical constraints translate into the concrete structure of atoms.
The Takeaway: Be Mindful of the Numbers!
So, what’s the practical implication for you, the budding chemist or physics enthusiast? It's a good reminder to pay attention to the constraints. When you're dealing with quantum numbers, always double-check the relationship between n and l.
If you’re asked to identify a valid or invalid orbital based on a set of quantum numbers, the first thing you should look for is a violation of the rule: l < n. If you see l ≥ n, you’ve found your culprit. The other quantum numbers (ml and ms) can also lead to invalid sets if they're not consistent with the given l and the presence of electrons, but the n-l relationship is the most fundamental barrier to orbital existence.
It’s like being a detective. You’re looking for the clue that breaks the case. In this instance, the clue is that seemingly innocent pair of numbers, n and l, and their unbreakable bond. Mess with that, and you’re creating an orbital that simply doesn't exist. It’s a great way to trip up someone (or yourself!) who isn’t paying close enough attention.
Next time you’re wrestling with quantum numbers, remember my dorm room incident and my professor’s knowing chuckle. It’s a friendly reminder that while the quantum world is full of marvels, it also has some very strict rules. And the rule about n and l? It's one of the most important for defining the very existence of an orbital. So, keep those numbers in line, and you'll be well on your way to understanding the incredible architecture of the atom!
