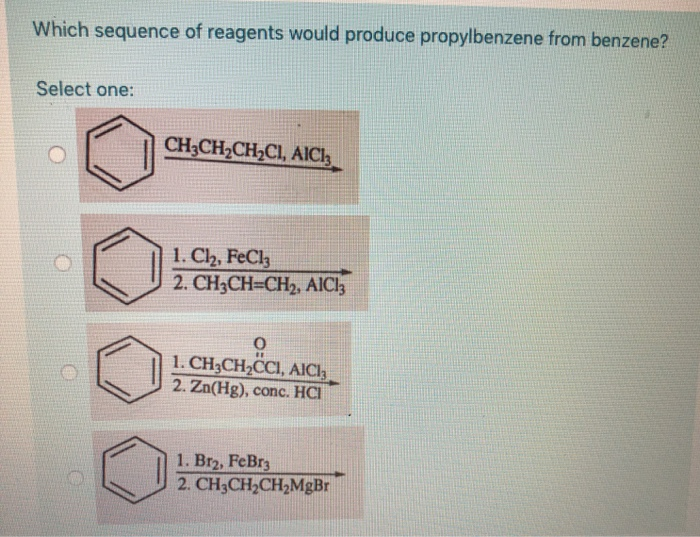
Which Sequence Of Reagents Would Produce Propylbenzene From Benzene
Ah, the tantalizing world of organic chemistry! While some might picture bubbling beakers and complex formulas, for many, it's a source of immense satisfaction and a fascinating puzzle to solve. Think of it like assembling an intricate LEGO set, but instead of plastic bricks, you're working with atoms and molecules. The thrill comes from understanding how different pieces fit together to create something entirely new and incredibly useful. It's a mental workout that rewards precision, patience, and a touch of creative thinking.
This intricate dance of molecules, particularly in synthesizing specific compounds, serves a vital purpose in our everyday lives, even if we don't always realize it. The ability to precisely build molecules is the backbone of countless industries. From the medicines that keep us healthy to the advanced materials in our electronics and the vibrant dyes in our clothing, it's all thanks to chemists who understand how to guide reactions to produce desired outcomes. For example, the plastic in your phone, the ibuprofen in your cabinet, or even the artificial sweeteners in your diet soda – all began as careful, step-by-step molecular construction projects.
Let's dive into a specific, yet illustrative, challenge: how to transform a simple benzene ring into propylbenzene. Imagine benzene as a plain, round foundation. We want to add a three-carbon chain to it. This isn't as straightforward as just sticking it on! We need a series of carefully chosen "reagents" – think of these as specialized tools or ingredients – to make this happen. The most common and effective sequence involves two key steps, each with its own brilliant chemical logic.
First, we need to introduce a precursor to our propyl chain onto the benzene ring. This is typically achieved through a process called Friedel-Crafts Acylation. We’d use an acyl halide, like propanoyl chloride (which contains our three-carbon chain in a reactive form), and a Lewis acid catalyst, such as aluminum chloride (AlCl₃). This powerful combination effectively “electrophilically attacks” the benzene ring, attaching the three-carbon carbonyl group. You’re essentially attaching a ketone to your benzene.
Now we have a benzene ring with a three-carbon carbonyl group attached. The next step is to get rid of that oxygen atom and turn the carbonyl group into a simple alkane chain. This is where reduction comes in. A classic and robust method for this is the Clemmensen Reduction, using zinc amalgam (an alloy of zinc and mercury) and concentrated hydrochloric acid. This aggressive combination strips away the oxygen, leaving behind our desired propylbenzene. Alternatively, a milder Wolff-Kishner Reduction using hydrazine and a strong base can also accomplish this transformation.
To enjoy this process, or the understanding of it, more effectively, think about visualizing the steps. Use molecular models if available, or draw out the structures and the movement of electrons. Understanding why each reagent is chosen is key. For Friedel-Crafts, it's about creating a strong electrophile that the electron-rich benzene can attack. For the reduction, it's about cleaving that specific carbon-oxygen bond. Embrace the elegance of the reactions and appreciate the precision required. It's a journey of transforming one molecule into another, and the satisfaction of seeing it through is truly rewarding!
