Which Reaction Of An Alkene Proceeds With Anti Addition

Ever wondered how chemists tweak molecules to create everything from life-saving medicines to the plastics that fill our everyday lives? It's a fascinating world of reactions, and today, we're diving into one that’s particularly neat: anti addition reactions with alkenes. Why is this so cool? Because understanding how atoms arrange themselves as they join a double bond can unlock secrets to controlling chemical transformations and designing new materials. It's like learning a secret handshake for molecules, a way to predict exactly where things will end up. This isn't just academic curiosity; it has real-world implications in synthesizing complex organic compounds, ensuring the right pieces fit together in the right way, which is crucial for the effectiveness and safety of many products we rely on.
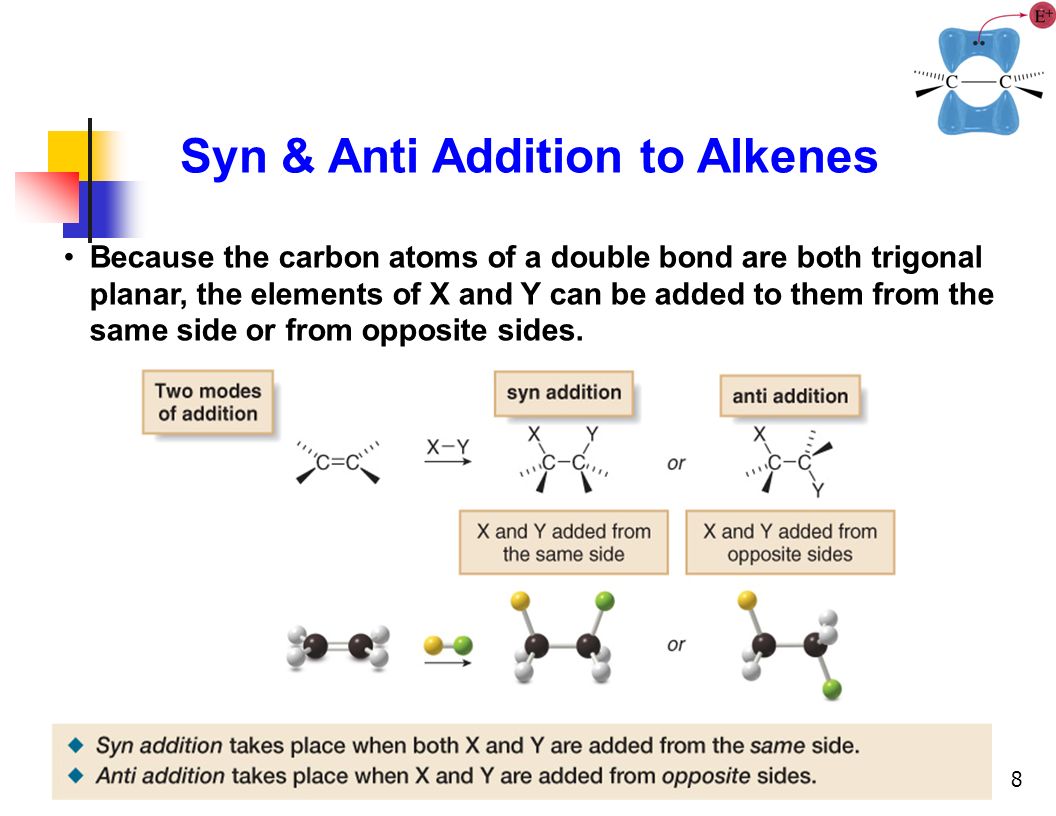
So, what exactly is anti addition? Imagine an alkene, that’s a molecule with a double bond (think of it as a handshake with two hands instead of one!). When a new group of atoms wants to join this double bond, they can do so in different ways. Anti addition is a specific type of reaction where the two new atoms or groups add to opposite sides of that original double bond. This is in contrast to syn addition, where they’d both join on the same side. Think of it like two people meeting in the middle of a dance floor and each stepping away from each other to greet someone new – they don't both try to hug the same spot on the floor!
The beauty of anti addition lies in its predictability and the specific stereochemistry it creates. Stereochemistry, in simple terms, is the 3D arrangement of atoms in a molecule. For many biologically active molecules, like drugs, getting the 3D shape just right is absolutely critical. A molecule that's a perfect fit for a target in your body might be completely inactive, or even harmful, if its atoms are arranged slightly differently. Anti addition offers a reliable way to control this crucial 3D arrangement, leading to the formation of specific isomers (molecules with the same chemical formula but different spatial arrangements of atoms).
There are several key reactions that famously proceed via anti addition, and each has its own unique charm and utility. One of the most prominent and widely studied is the halogenation of alkenes. In this reaction, a halogen molecule, like bromine (Br₂) or chlorine (Cl₂), reacts with an alkene. As the halogen molecule approaches the double bond, it forms a cyclic intermediate called a halonium ion. This intermediate is key to the anti addition pathway. It’s like a temporary bridge formed between the two carbons of the double bond, and it forces the incoming halogen atoms to approach from opposite sides. When the second halogen atom attacks, it opens this bridge, resulting in the two halogen atoms being positioned on opposite carbons of what was once the double bond, and importantly, on opposite faces of the molecule. This leads to the formation of a vicinal dihalide, with the halogens on adjacent carbons.
Another important example is the hydrohalogenation of alkenes in the presence of peroxides. While simple hydrohalogenation (adding H-X, where X is a halogen) without peroxides usually follows Markovnikov's rule and can lead to either syn or anti addition depending on the specific conditions, the presence of peroxides changes the game. Peroxides initiate a radical mechanism. This radical pathway often favors anti addition, meaning the hydrogen and the halogen end up on opposite sides of the original double bond. This is a subtle but significant difference that can dramatically alter the outcome of the reaction and the structure of the final product. It’s a great example of how a small change in reactants can lead to a completely different mechanistic pathway and, consequently, a different stereochemical outcome.
Furthermore, the dihydroxylation of alkenes using osmium tetroxide (OsO₄), while typically a syn addition, can be manipulated to achieve anti addition through indirect routes, showcasing the versatility of chemical synthesis. However, for direct anti addition, the halogenation of alkenes is the classic and most straightforward illustration. Understanding these distinctions is not just about memorizing reaction types; it's about appreciating the underlying principles of chemical reactivity and orbital interactions that dictate molecular behavior. The controlled placement of atoms is a cornerstone of synthetic chemistry, enabling chemists to build intricate molecular architectures with precision. This knowledge is invaluable when designing synthetic routes for complex pharmaceuticals, agrochemicals, and advanced materials, where even subtle changes in molecular structure can have profound effects on properties and function. So, the next time you encounter a reaction involving an alkene, remember the elegance of anti addition and how it helps shape the molecules that shape our world!
