Which Reaction Below Represents The Second Electron Affinity Of S

Hey there, lovelies! Ever feel like you’re juggling a million things and your brain feels like it’s on a perpetual coffee break? Yeah, me too. Life these days is a whirlwind of deadlines, social media scrolls, and trying to remember if you’ve actually eaten anything besides cereal. But amidst all the hustle, sometimes it’s fun to dive into something a little… different. Something that sparks a tiny bit of curiosity and, dare I say, might even make you feel a tad smarter. We’re talking about the weird and wonderful world of chemistry, but don’t worry, we’re keeping it light, breezy, and totally jargon-free. Think of it as a mini-vacation for your brain, a little detour into the microscopic, where even tiny things have big personalities.
Today, we’re shining a spotlight on an element that’s pretty darn important, even if it doesn't get the same star power as, say, gold or platinum. We’re talking about sulfur, or ‘S’ as it’s known to its science-savvy friends. You know, the stuff that makes matches light up, or the not-so-pleasant smell from a rotten egg? Yep, that’s our guy. Sulfur is actually all around us, playing a crucial role in everything from the proteins in our bodies to the volcanic eruptions that remind us the Earth is a living, breathing entity. It’s like the unsung hero of the periodic table, always there, doing its thing.
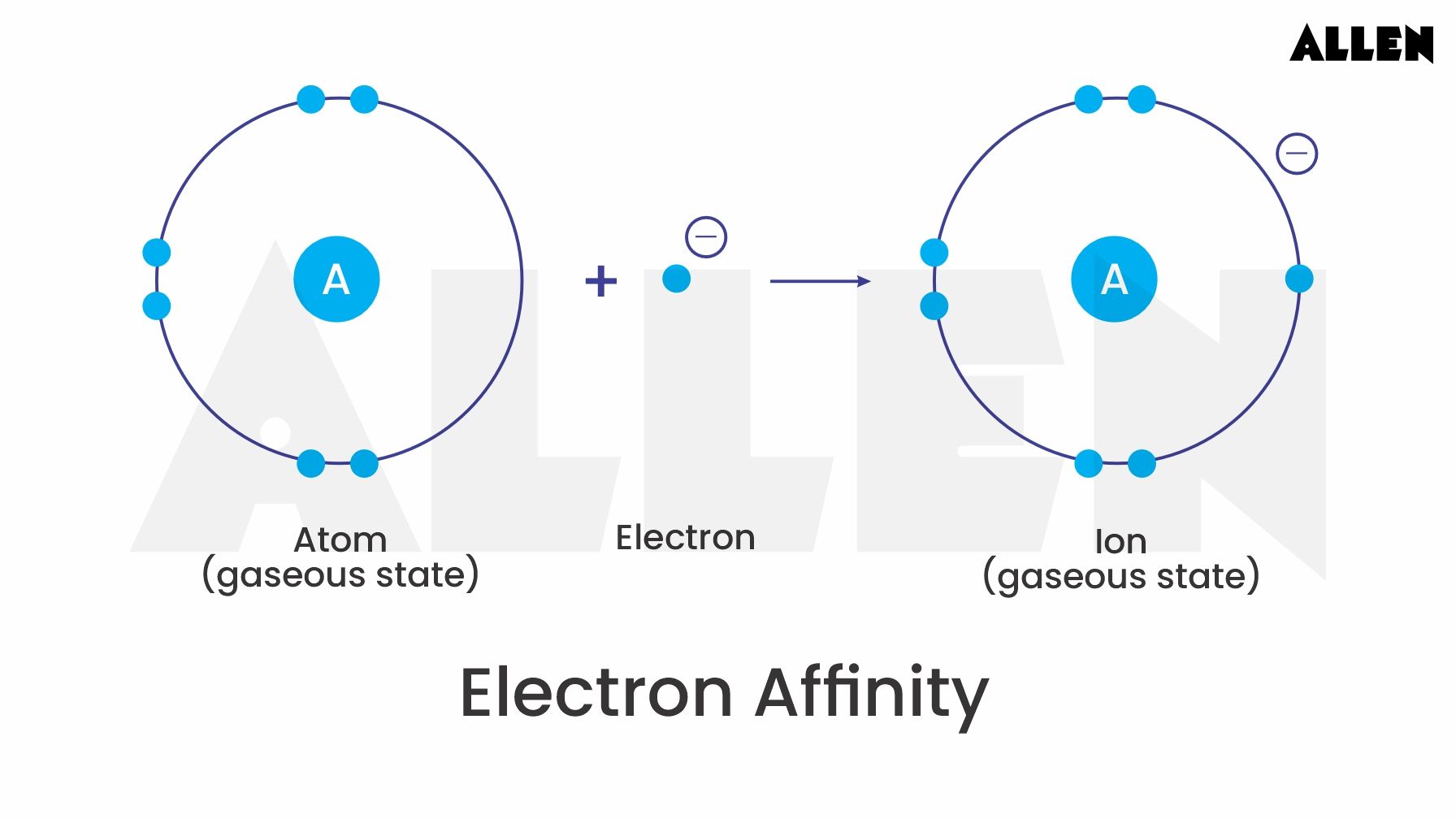
Now, let’s get a little bit meta and talk about how elements like sulfur behave when they’re trying to… well, make friends. In the atomic world, making friends often involves either giving away or grabbing electrons. It’s like a cosmic game of tag, where elements are constantly looking to achieve a stable, happy electron configuration. Think of it like finding the perfect playlist to get you in the zone – once you find it, everything just clicks. For many elements, gaining electrons is their jam. And when they’re really good at it, we call them electronegative. Sulfur, as you might have guessed, is pretty good at this electron-grabbing business.
We’ve probably all heard of the first electron affinity. This is basically the energy change when an atom picks up its first electron. It’s like that initial excitement when you start a new hobby – there’s a definite energy boost, a feeling of accomplishment. Sulfur, being the eager beaver it is, readily accepts a lone electron to become a negatively charged ion, S-. This process releases energy, which is pretty neat. It’s like getting a little dopamine hit for being so accommodating. This first electron affinity is generally a comfortable, predictable process, much like your favorite comfy sweats – always reliable, always providing a sense of ease.
But here’s where things get really interesting. What happens when our newly formed S- ion, already feeling pretty cozy with its extra electron, tries to grab another one? This is where we delve into the second electron affinity. And let me tell you, it’s a whole different ballgame. Imagine you’re already stuffed after a huge Thanksgiving dinner, and someone offers you dessert. You might make room, but it’s probably going to take a bit more convincing, right? It’s definitely not going to be as effortless as that first slice of pie.
So, the question we’re pondering today, the one that might tickle your curious bone, is: Which reaction below represents the second electron affinity of S? This isn’t just about memorizing chemical equations; it’s about understanding the underlying principles and appreciating the nuanced dance of atoms. Think of it as understanding why you crave that second cup of coffee on a Monday morning – there’s a reason, and it’s not always straightforward!
Decoding the Reactions
Let’s break down what we’re looking for. We need a reaction where a sulfur atom already carrying a negative charge (meaning it has gained one electron) accepts another electron. This is key. The starting point isn’t a neutral sulfur atom; it's a sulfur ion that's already a bit electron-rich.
When an atom gains electrons, it becomes a negative ion, also known as an anion. The first electron affinity deals with a neutral atom becoming a singly charged anion. The second electron affinity, then, must involve a singly charged anion becoming a doubly charged anion. This is a crucial distinction, like the difference between a casual acquaintance and a best friend – the relationship has deepened, and the dynamics have shifted.

Consider the forces at play. Our S- ion already has a negative charge. Now, we're asking it to welcome another electron, which is also negatively charged. What do we know about things with the same charge? They repel each other, right? It’s like trying to push two north poles of magnets together – they just don’t want to cooperate. So, for sulfur to accept this second electron, it’s going to require a significant input of energy. It’s fighting against its own inherent electron-richness. This is a concept that’s pretty fundamental to understanding chemical bonding and the overall stability of elements.
Think of it like this: the first electron affinity is like getting a free sample at the grocery store – easy, appealing, and you’re happy to take it. The second electron affinity, however, is like trying to squeeze into a crowded elevator when you’re already carrying a lot of shopping bags. It’s possible, but it’s going to take more effort, more pushing, and you might not feel as good about it afterwards.
The Energy Game
This energy aspect is where things get really interesting. The first electron affinity for sulfur is exothermic, meaning it releases energy. This is why sulfur readily forms S-. It’s a energetically favorable process, like finding a parking spot right outside your destination – pure bliss! However, the second electron affinity for sulfur is endothermic, meaning it requires energy to occur. We have to put energy in to force that second electron onto the already negatively charged sulfur ion. This is a critical point, and it’s what differentiates the two processes.
Why is this important? It tells us a lot about how elements interact and the types of compounds they form. Elements with highly positive second electron affinities (meaning they strongly resist gaining a second electron) are less likely to form doubly negative ions on their own. This is why we don't typically find stable S2- ions floating around in everyday situations. It takes a lot of work to get them there!
This concept extends beyond just sulfur. It's a fundamental principle that helps chemists predict the behavior of elements and design new materials. Understanding these energy changes allows us to build more efficient batteries, create stronger alloys, and even develop new catalysts for industrial processes. It’s like knowing the secret recipe to your grandma’s cookies – once you have it, you can replicate the magic!
So, when you’re looking at the reactions, you’re not just looking for a sulfur atom gaining an electron. You’re looking for a sulfur ion (specifically, S-) gaining another electron, and ideally, you’d see an indication that energy is being absorbed rather than released. This energy aspect is often represented by a positive ΔH value if enthalpy is being discussed, or simply by the fact that the reaction is written with energy on the reactant side.

Let’s visualize this. If we see a reaction like:
S-(g) + e- → S2-(g)
This is the fundamental process we’re after. The (g) denotes that the species are in the gaseous state, which is typically how these affinities are measured to avoid interference from surrounding molecules. The key here is the S- starting material and the addition of a single electron (e-) to form S2-.
Now, let’s consider what a first electron affinity reaction for sulfur would look like:
S(g) + e- → S-(g)
See the difference? The first one starts with a neutral sulfur atom (S), while the second one starts with a sulfur ion that already has a negative charge (S-). This subtle but crucial distinction is what separates the first from the second electron affinity.
Often, when these questions are presented in a multiple-choice format, you'll see similar-looking reactions. The trick is to identify the starting species and the overall charge change. You’re essentially looking for the reaction that describes adding a second electron to an already electron-rich sulfur species.
Think of it like a popular band playing a concert. The first time they release an album (first electron affinity), everyone’s buzzing, and it’s a massive hit. The second album (second electron affinity)? It's still great, but it might take a bit more effort for it to gain the same traction, and the band might have to work harder to impress their existing fans. The energy output is different.
Furthermore, sometimes the options might include energy values. If a reaction shows energy being released (exothermic), it's likely the first electron affinity. If it shows energy being absorbed (endothermic), that's your cue for the second electron affinity. It’s like looking at the weather report: a sunny day (exothermic) versus a day where you need an umbrella and a jacket (endothermic).
Fun Facts and Cultural Tidbits
Did you know that sulfur has been used for centuries in various applications? Ancient civilizations used it for fumigation and in medicinal concoctions. It's also the key ingredient in gunpowder, which, while not exactly a lifestyle choice we're advocating for, has certainly shaped history! Imagine the alchemists, hunched over their bubbling beakers, trying to unlock the secrets of sulfur. It's like a scene straight out of a fantasy novel, but it was real science!
And sulfur’s distinctive smell? That’s often due to compounds like hydrogen sulfide (H₂S). While unpleasant to some, this smell is also a sign of life, or rather, decomposition. Microorganisms break down organic matter, releasing sulfur compounds. So, the next time you encounter that distinctive aroma, you can think, "Ah, the symphony of microbes at work!" It's a natural alarm system, of sorts, reminding us of the constant cycle of life and decay.
In popular culture, sulfur sometimes pops up as a symbol of the underworld or hell, likely due to its association with volcanic activity and its pungent smell. Think of those dramatic scenes in movies where characters are descending into a fiery abyss – you can bet there's a whiff of sulfur in the air!
Culturally, the element sulfur is deeply embedded in folklore and mythology. In some traditions, it's associated with purification and protection. It’s also been linked to the divine and the demonic, highlighting its dual nature – both beneficial and potentially destructive. It’s a reminder that even the smallest things can have profound and diverse meanings.
When it comes to identifying the correct reaction, think of it like choosing your perfect outfit for the day. You have options, but only one truly fits the occasion. You wouldn't wear a ballgown to the grocery store, and you wouldn't wear gym clothes to a formal dinner. Similarly, you need to match the reaction to the specific process of second electron affinity. It's all about the context and the energy required for that particular transformation.
So, to recap, when you’re faced with this question, keep your eyes peeled for these key indicators:
- The reaction should start with a sulfur species that already has a negative charge (S-).
- It should show the addition of a single electron (e-).
- The product should be a sulfur species with a doubly negative charge (S2-).
- Ideally, there might be an indication that energy is being absorbed for this process to occur.
These are the hallmarks of the second electron affinity. It's about understanding that gaining that second electron isn't as easy as the first. It’s a step up, a challenge, and it requires a different energetic approach.
A Daily Dose of Chemistry
It’s easy to think of chemistry as something confined to sterile labs with white-coated scientists. But the truth is, chemistry is happening all around us, all the time. It’s in the way your coffee brews, the way your phone battery charges, and even the way your body digests that tasty snack you’re probably reaching for right now. Understanding these fundamental principles, like electron affinities, gives you a secret superpower. You start to see the hidden workings of the world, the invisible forces that shape everything we see and experience.
Life is a series of energy exchanges, much like these chemical reactions. Sometimes, giving a little (like offering a listening ear to a friend) comes easily and feels rewarding. Other times, it takes a lot of effort to overcome resistance, to push through challenges, and to absorb the energy needed to make things happen. And that’s okay. Recognizing this ebb and flow, this need for effort in certain situations, is part of growing and understanding ourselves and the world around us. So, the next time you’re faced with a complex situation, remember the second electron affinity of sulfur. It’s a reminder that not every gain is effortless, and sometimes, the greatest rewards come from the most challenging endeavors.
