Which Reactants Would Lead To A Spontaneous Reaction
Ever find yourself staring at a perfectly brewed cup of coffee, that little swirl of cream just begging to mingle? Or maybe you’ve watched a dramatic cooking show, where ingredients just leap into action, transforming into something delicious? These everyday moments are actually a peek into the fascinating world of chemistry, specifically, spontaneous reactions. Think of it like nature’s ultimate “easy button.” When things just happen without a constant nudge from us, that’s usually a sign of a spontaneous process. And in the realm of reactants, some pairs are just natural-born collaborators, ready to team up and get things done. Let’s dive into what makes these chemical partnerships so effortlessly chill.
So, what exactly is a spontaneous reaction? In the simplest terms, it’s a reaction that, once initiated, will proceed on its own without any continuous external energy input. It’s not about being fast, mind you. Some spontaneous reactions are like watching paint dry, while others are as explosive as a mic drop. The key is that they don't require you to keep feeding them energy to keep going. Think of it as the universe’s way of finding the path of least resistance, a bit like how you’d rather scroll through your phone than do extra chores.
At the heart of it all are a couple of fundamental concepts: enthalpy and entropy. Don't let the fancy terms scare you! Enthalpy (ΔH) is basically the heat change during a reaction. If a reaction releases heat (exothermic, ΔH is negative), it’s like a warm hug for the universe, and the universe usually appreciates that. Entropy (ΔS) is all about disorder or randomness. Think of your desk after a productive (or not-so-productive) work session – that’s entropy in action! A reaction that increases disorder (ΔS is positive) is also generally favored.
The real magic happens when we combine these two with Gibbs Free Energy (ΔG). This is our ultimate cheat sheet for spontaneity. The equation is elegantly simple: ΔG = ΔH - TΔS. Here, 'T' is the temperature in Kelvin. If ΔG is negative, congratulations! You’ve got yourself a spontaneous reaction. It's like a cosmic "yes" stamp. If ΔG is positive, it’s a spontaneous "nope," meaning you'll need to put in energy to make it happen. If ΔG is zero, the system is at equilibrium – perfectly balanced, like a tightrope walker.
Now, let's talk about the reactants themselves. What kind of personalities do they need to have to be a spontaneous duo? It’s all about their inherent tendencies to reach a lower energy state (enthalpy) or a more disordered state (entropy), or both!
The "Hug It Out" Duo: Exothermic Reactions
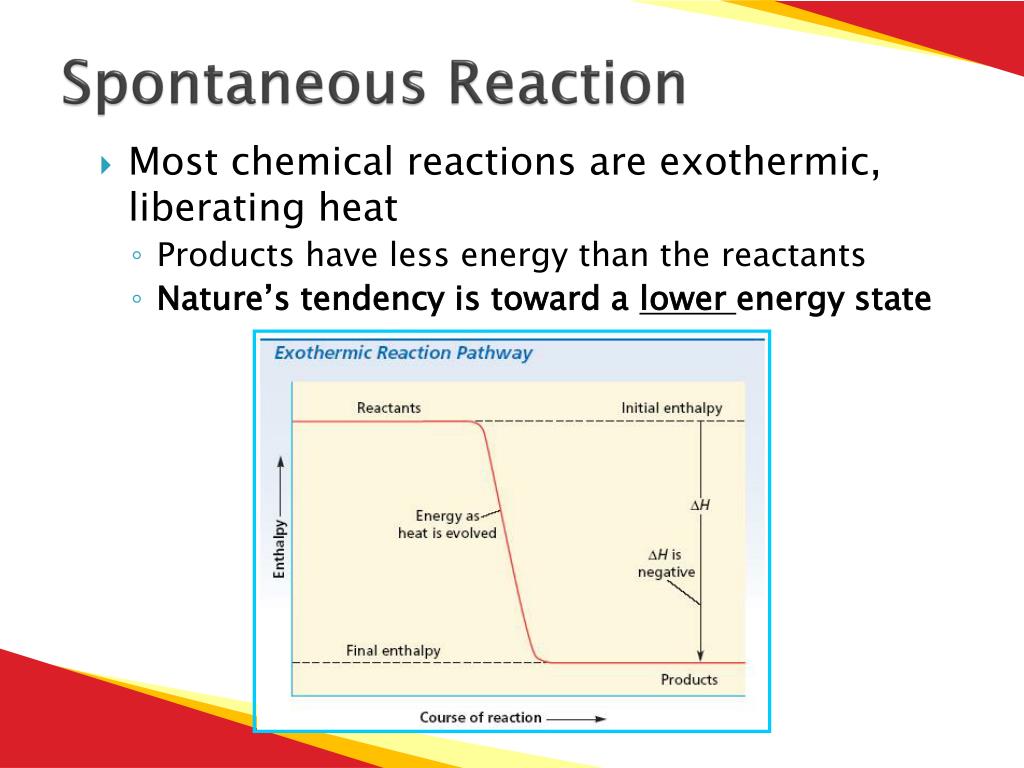
Imagine two friends who just love to chill and share their energy. That’s kind of what happens in an exothermic reaction. These reactions release heat into their surroundings. Think of the classic “hand warmer” packet. You snap it, and within minutes, it’s radiating a cozy warmth. This is because the reactants are in a higher energy state, and by bonding, they’re releasing that excess energy as heat, settling into a more stable, lower-energy configuration. It's like they’re so happy to be together, they’re giving off good vibes (and heat!).
A prime example is the combustion of fuels. When you burn wood or natural gas, you’re witnessing a highly exothermic, spontaneous reaction. The carbon and hydrogen in the fuel combine with oxygen, releasing a tremendous amount of energy as heat and light. This is why we’ve harnessed fire for millennia – it’s a readily available, energetic handshake between fuel and oxygen. It’s the chemical equivalent of a perfectly executed high-five.
Another relatable example? Dissolving strong acids in water. Think about mixing something like sulfuric acid. It gets noticeably warm. The ions in the acid are happily interacting with the water molecules, and in the process, they release energy. It's a satisfying rumble of chemical bonding that’s easy to observe.

Practical Tip: Ever noticed how your phone battery feels warm after heavy use? That’s a sign of exothermic processes happening inside. It’s also why you shouldn’t leave your phone charging in a super hot car – the heat generated can further impact the battery’s performance.
Fun Fact: The term "combustion" comes from the Latin word "combustio," meaning "to burn." It's a pretty straightforward origin story for a process that's anything but simple in its chemical dance.
The "Let It All Hang Out" Crew: Entropy Boosters
Then there are the reactants that just love a bit of chaos. These are the ones that lead to an increase in entropy. Think about a perfectly organized deck of cards. Now, shuffle them vigorously. That’s entropy increase! In chemistry, this often happens when a solid dissolves into a liquid, or when a liquid evaporates into a gas. The particles go from being neatly arranged to being much more spread out and jumbled. This increase in disorder is a powerful driving force for spontaneity, especially at higher temperatures.
Consider dissolving a spoonful of sugar in your morning tea. The sugar crystals (ordered solid) break down into individual sugar molecules dispersed throughout the tea (much less ordered liquid). This process is spontaneous, largely because the entropy of the system increases significantly. The sugar molecules are now free to roam and mingle, much like you might feel after a long week when all you want to do is kick back and let loose.
Another excellent example is the evaporation of water. Even at room temperature, some water molecules will escape the liquid phase and become water vapor. This increases the disorder, as the molecules have far more freedom to move in the gaseous state. It’s the universe saying, “Let’s spread out and have some fun!”
Practical Tip: When you’re packing for a trip, think about how things can get jumbled. Using packing cubes helps organize your clothes (decrease entropy within the cube) but the overall act of packing and unpacking involves a lot of messy moving around (increase entropy of your belongings relative to their original state). It's a fun analogy for how entropy plays out in our lives!

Cultural Reference: In Japanese culture, the concept of wabi-sabi celebrates imperfection and impermanence. This resonates with the idea of entropy – finding beauty in the natural progression towards disorder and change, rather than fighting against it.
The "Two Birds, One Stone" Champions: Exothermic & Entropy-Boosting Reactants
And then, you have the ultimate power couples. These are the reactions that are both exothermic (ΔH negative) and increase entropy (ΔS positive). These guys are practically guaranteed to be spontaneous, no matter the temperature. They’re hitting all the right notes for a negative ΔG. It’s like finding a parking spot right in front of the store on a Saturday afternoon – pure, unadulterated spontaneity!
A fantastic example is the reaction between a strong acid and a strong base, like hydrochloric acid (HCl) and sodium hydroxide (NaOH). When you mix them, they neutralize each other, forming salt (NaCl) and water (H₂O). This reaction is highly exothermic – it releases a good amount of heat. It also increases entropy because you’re going from two distinct molecular species to ions dissolved in water, which are much more spread out and mobile than the original reactants. The formation of a stable salt and water also represents a significant drop in potential energy.
Think of it as the ultimate act of chemical cooperation. They’re releasing energy and becoming more disordered simultaneously. It’s the chemical equivalent of a perfect, harmonious collaboration where everyone wins. This is why neutralization reactions are so common and predictable.
Practical Tip: When you’re doing any kind of cleaning that involves mixing chemicals (always with caution and following instructions!), you might observe heat being generated. This is often a sign of a spontaneous, exothermic reaction. Be mindful of ventilation and proper disposal.
Fun Fact: The concept of neutralization is central to many biological processes, including how your body maintains its pH balance, a crucial aspect of keeping you alive and well!

The "It Depends on the Vibe" Couples: Enthalpy vs. Entropy Dominance
Life isn't always black and white, and neither is chemistry. Some reactions have conflicting tendencies. You might have an endothermic reaction (absorbs heat, ΔH positive) that also increases entropy (ΔS positive). Or an exothermic reaction (releases heat, ΔH negative) that decreases entropy (ΔS negative).
For the endothermic-entropy-increasing case (ΔH positive, ΔS positive), spontaneity is dictated by temperature. At high temperatures, the TΔS term becomes dominant. If the increase in entropy is large enough, it can overcome the positive enthalpy, making ΔG negative. A good example is the melting of ice. Ice melting into water is endothermic (you need to add heat), but it significantly increases entropy as the water molecules become much more mobile. So, on a warm day, ice melts spontaneously!
Conversely, for an exothermic-entropy-decreasing case (ΔH negative, ΔS negative), spontaneity also depends on temperature, but in the opposite direction. At low temperatures, the negative ΔH term can dominate. If the reaction releases enough heat, it can overcome the decrease in entropy, making ΔG negative. Think about freezing water. Water freezing into ice is exothermic (it releases heat), but it decreases entropy as the molecules become more ordered. On a cold night, water freezes spontaneously!
Practical Tip: This is why keeping food in the refrigerator (low temperature) prevents it from spoiling spontaneously. Many spoilage reactions are exothermic and increase entropy, but at low temperatures, the freezing process (exothermic, decrease entropy) dominates, slowing them down. It’s like putting a chill on the chaos!
Cultural Reference: The concept of yin and yang in Taoism – representing opposing but complementary forces – can be a useful way to think about enthalpy and entropy. They often work in tandem, sometimes in opposition, to create the balance of spontaneous processes.
What About the "Nope" Reactions?
And then there are the reactions that are simply not spontaneous under normal conditions. These are the ones where you need to actively put in energy. Think about trying to make a ball roll uphill. It’s not going to happen on its own; you need to push it. These are non-spontaneous reactions, and they often have a positive ΔG. They might be endothermic and decrease entropy, or have other combinations that lead to a positive energy outcome.

An example is the production of ozone (O₃) from oxygen (O₂). This requires energy input, often in the form of UV radiation or electrical discharge. It's an endothermic reaction that decreases entropy (three oxygen atoms forming two ozone molecules). So, nature doesn't just spontaneously decide to make more ozone; it needs a kickstart.
Another common example is the process of photosynthesis. Plants use energy from sunlight to convert carbon dioxide and water into glucose (sugar) and oxygen. This is a highly endergonic (non-spontaneous) process that requires a continuous input of solar energy. Without that sunshine, the reaction wouldn't happen.
Practical Tip: When you charge your phone, you're essentially forcing a non-spontaneous reaction. You’re putting electrical energy into the battery to reverse the discharge process and store energy. It's a great example of how we can harness energy to drive necessary, but not naturally occurring, chemical changes.
Fun Fact: The term "non-spontaneous" is sometimes referred to as "endergonic," meaning "energy within," as opposed to "exergonic" (energy out) for spontaneous reactions.
The Takeaway: Nature Loves a Lazy (But Productive) Path
Ultimately, the question of which reactants lead to a spontaneous reaction boils down to their inherent tendencies to move towards a state of lower energy and/or higher disorder. The universe, in its grand, elegant way, favors processes that are less demanding. It’s like us – given a choice, we often gravitate towards the path of least resistance. Whether it’s a warm hug of an exothermic reaction, a wild dance of increasing entropy, or a perfect blend of both, nature is constantly orchestrating these spontaneous events all around us.
Thinking about spontaneity in chemistry is a lot like thinking about our own lives. We’re often looking for that flow, that feeling of things just working. Sometimes, it’s about creating the right conditions – the warmth of connection (exothermic), the freedom to express ourselves (entropy increase). Other times, it’s about recognizing that some things just require that extra effort, that intentional push. But understanding these basic chemical principles can give us a little more appreciation for the effortless magic that happens every single day, from the smallest chemical bond to the grandest cosmic dance.
