Which Ph Value Is Consistent With The Indicator Results
So, you're hanging out, maybe doing some baking or playing with some cool science stuff, and you’ve got this mystery on your hands. What is it? It’s all about pH!
Yeah, pH. Sounds fancy, right? But it’s actually super chill. Think of it as the secret handshake of liquids. Some are happy and bubbly (alkaline), some are a bit more… well, acidic. And some are just cruising right in the middle.
And how do we know which is which? That’s where our trusty sidekick comes in: the pH indicator. These guys are like tiny mood rings for your liquids. They change color to tell you what's going on. Pretty neat, huh?
The Rainbow Detectives
pH indicators are basically nature's little chemists. They’re often made from plants! Yep, stuff like red cabbage. Who knew your salad could be a science experiment?
Red cabbage juice is a rockstar. It can be purple in neutral water, red or pink in acids, and blue, green, or even yellow in bases. Talk about a glow-up!
Imagine you’re a detective. Your mission? To uncover the pH of a mysterious liquid. Your tools? Some super cool pH indicators. Your clue? The color!
Each indicator has its own personality, its own color chart. It’s like having a secret codebook. You see a color, you check the book, and BAM! You know the pH. Easy peasy.
Why pH Even Matters (Besides Being Fun)
Okay, so it's fun to make things change color, but why do we even care about pH? Well, it's everywhere!
Your body needs a specific pH to work right. Too acidic or too alkaline? Not good. Your stomach acid? Super acidic. Your blood? Needs to be pretty darn close to neutral.
Gardening? pH is key. Some plants love acidic soil, others prefer it more alkaline. It’s like choosing the right shoes for a hike – gotta match the terrain!
And food! Ever had a sour candy? That's the acid talking. Fermenting foods, like yogurt or kimchi? pH plays a huge role in how that happens.
So, while we're having a blast with our color-changing experiments, we're actually learning about the world around us. Pretty cool, right?
Decoding the Colors: What's the pH?
Let's get to the juicy part. You've got your indicator, you've dipped it in your liquid, and it's turned a certain color. Now what?
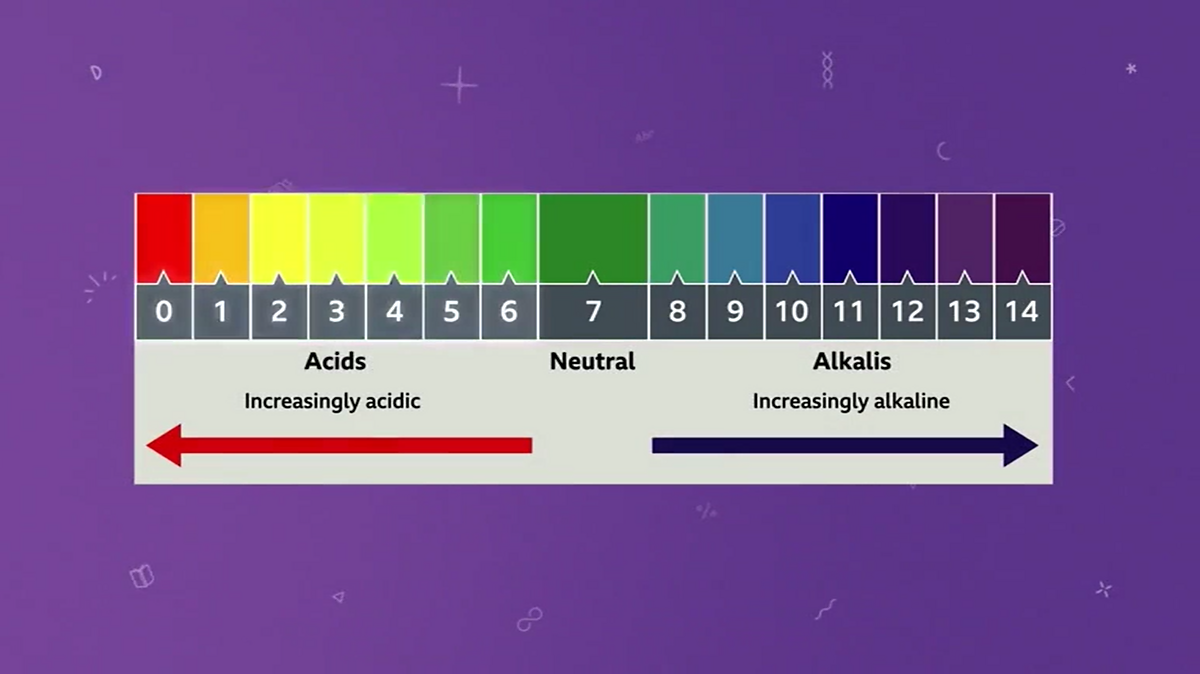
This is where the pH scale comes into play. It's a scale from 0 to 14. Think of it like a thermometer, but for acidity.

0 to 6.9? That's the acidic zone. Think lemon juice, vinegar, battery acid (but please, don't taste battery acid!).
7? That's the sweet spot, the neutral zone. Pure water is the classic example. Nothing too extreme going on here.
7.1 to 14? Welcome to the alkaline (or basic) side. Think baking soda, soap, bleach (again, be careful with bleach!).
So, if your red cabbage indicator turns bright red, you’re probably deep in the acidic zone. If it’s a lovely blue, you’re likely near neutral. And if it’s a sunny yellow? Hello, alkaline party!
The Quirky Truths of Indicators
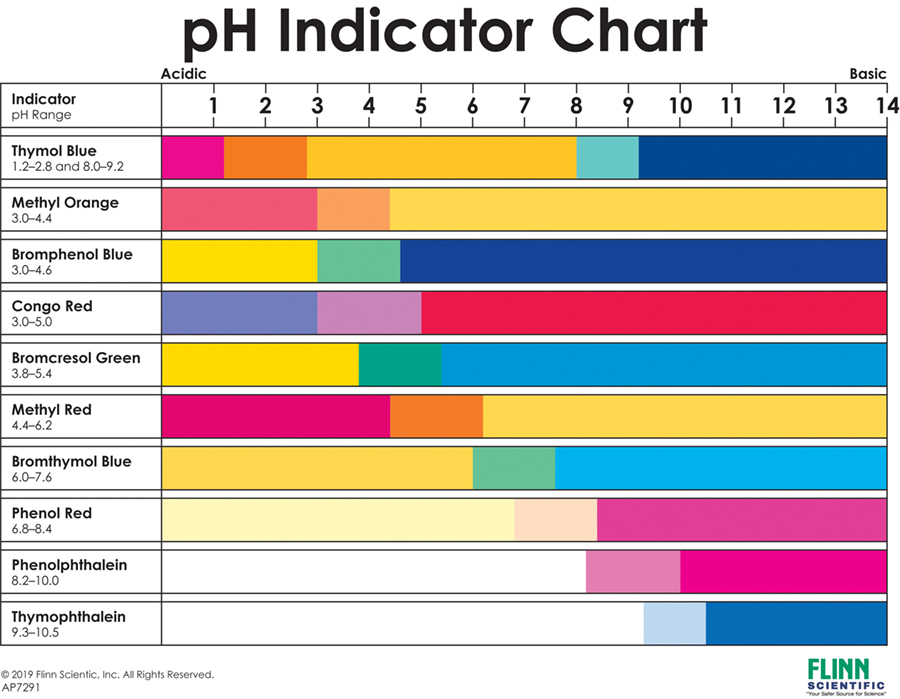
Here’s a funny little detail: different indicators have different transition ranges. That's just a fancy way of saying they only change color within a certain pH range.
It’s like having a bunch of different thermometers. One might be great for measuring your body temp, but useless for a blast furnace. Similarly, one pH indicator might be perfect for detecting weak acids, while another is better for spotting strong bases.
For example, phenolphthalein is a popular one. It’s colorless in acidic solutions and turns a vibrant pink or fuchsia in alkaline solutions (above pH 8.2). It’s not much of a detective in the acidic or neutral ranges, but when it comes to spotting bases? It's like a neon sign!
And what about litmus paper? Those classic strips you see in science labs? They're simple but effective. Red litmus paper turns blue in alkaline solutions, and blue litmus paper turns red in acidic solutions. They give you a quick yes/no for acidity or alkalinity, but not a super precise number.
It’s like having a set of colored lenses. You put them on, and the world changes color. Each lens reveals a different aspect of the liquid's true nature.
Putting It All Together: The Fun Part!
So, you’ve got your indicator results. Let’s say you tested a mystery liquid with red cabbage juice. It turned a beautiful shade of green.
Now, you consult your mental (or actual!) red cabbage pH chart. You remember (or look up) that green typically falls in the alkaline range. It's definitely not red (acidic) and not purple (neutral).
So, the pH value consistent with your indicator result of green is likely above 7. You might not know if it's exactly 7.5 or 8.2 without a more precise indicator or a pH meter, but you've got a solid answer: it's alkaline!

What if you used phenolphthalein and it turned pink? Easy! That means your liquid is alkaline, so its pH value is greater than 8.2.
It’s this detective work, this visual puzzle-solving, that makes pH indicators so darn fun. You're not just reading numbers; you're observing a transformation.
A World of Wonder, One Color at a Time
Think about it: we can use plants to understand the invisible properties of liquids. It's like unlocking a hidden language of the universe.
From the tang of your orange juice to the froth of your dish soap, pH is an unseen force shaping our world. And with pH indicators, we get a front-row seat to the show.
So next time you're feeling curious, grab some red cabbage or a pack of litmus paper. Play around. See what colors you can create. You might just surprise yourself with how much fun you can have unraveling the mysteries of pH.
Remember, the pH value is consistent with the indicator result when the color you observe matches the color associated with a specific pH range on that indicator's chart. It's a beautiful, colorful dance between chemistry and observation!
