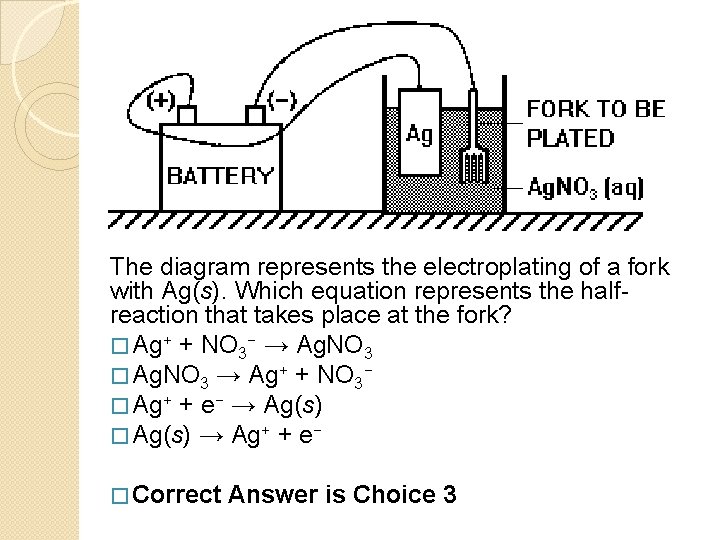
Which Part Of The Electroplating Is Represented By The Fork

Picture this: you're at a fancy dinner party, or maybe just a really good Tuesday night meal. You reach for your trusty fork, ready to tackle that delicious pasta or a perfectly roasted chicken. It feels solid in your hand, doesn't it? You probably don't give it much thought beyond its immediate job: getting food to your mouth. But here's where things get surprisingly interesting. That humble fork, especially if it’s got that lovely shiny coating – the kind that glints under the candlelight – is a tiny, everyday marvel of electroplating. And guess what part of that magical process it represents?
It’s the Cathode!
Now, I know what you might be thinking. "Cathode? Sounds like some kind of sci-fi robot!" And in a way, it kind of is, but a very helpful and delicious one. Think of the electroplating process like a very polite, very controlled magic trick. We want to give something a beautiful, protective, or even just a more appealing outer layer. In our fork's case, we're likely giving it a coat of something like nickel, chromium, or even a lovely touch of gold to make it look extra special and stop it from getting all dull and rusty.
So, how does our fork become the star of the show as a cathode? Imagine a big tub filled with a special liquid – we call this the electrolyte. This liquid is like a bustling city square where all sorts of charged particles are hanging out, ready to do some work. Now, we take our plain, perhaps a little boring, metal fork and we connect it to the negative side of a battery. This is where the magic really begins.
Because the fork is now connected to the negative terminal, it becomes negatively charged. It's like giving the fork a big, welcoming hug from all the electrons. These eager electrons are buzzing around, and they're looking for a positive friend to hang out with. Meanwhile, in that same electrolyte bath, we have the metal we want to coat the fork with – let’s say, those shiny nickel atoms. These nickel atoms, in the electrolyte, have a positive charge. They’re like the popular kids at the party, looking for someone to dance with.

And who do they find? Our negatively charged fork! The positive nickel ions (that's the technical term for our positive metal atoms) are totally drawn to the negative fork. It’s an irresistible attraction, like magnets doing a little dance. They zoom over to the fork and, with a gentle (and very fast!) process, they start to stick. They’re essentially depositing themselves onto the fork, one tiny atom at a time.
It's like the fork is saying, "Come on over, shiny metal friends! I've got plenty of negative energy to share!"
Oxidation Reduction Regents Review Zns Pb 2 aq
And that’s it! The fork, acting as the cathode, is the surface where all the beautiful, new metal ions decide to settle down and form a lovely, even coating. It's the receiver, the welcoming committee for the precious metal that will transform it from ordinary to extraordinary. The other part of the story, the part that makes all this happen, is the anode. The anode is usually a piece of the same metal we want to deposit (like a chunk of pure nickel) and it’s connected to the positive side of the battery. It's like the supplier, giving up its electrons to keep the whole operation going. But today, our spotlight is on the fork, the magnificent cathode.
So, the next time you’re twirling spaghetti or spearing a delicious piece of steak, take a moment. Appreciate that gleaming utensil in your hand. It's not just a tool; it's a testament to a clever scientific process. It's a little piece of everyday art, made possible by the humble fork playing its crucial role as the cathode, happily accepting its shiny new makeover. It’s a story of attraction, of transformation, and of how a little bit of electricity can make our lives, and our dinners, a whole lot more delightful.