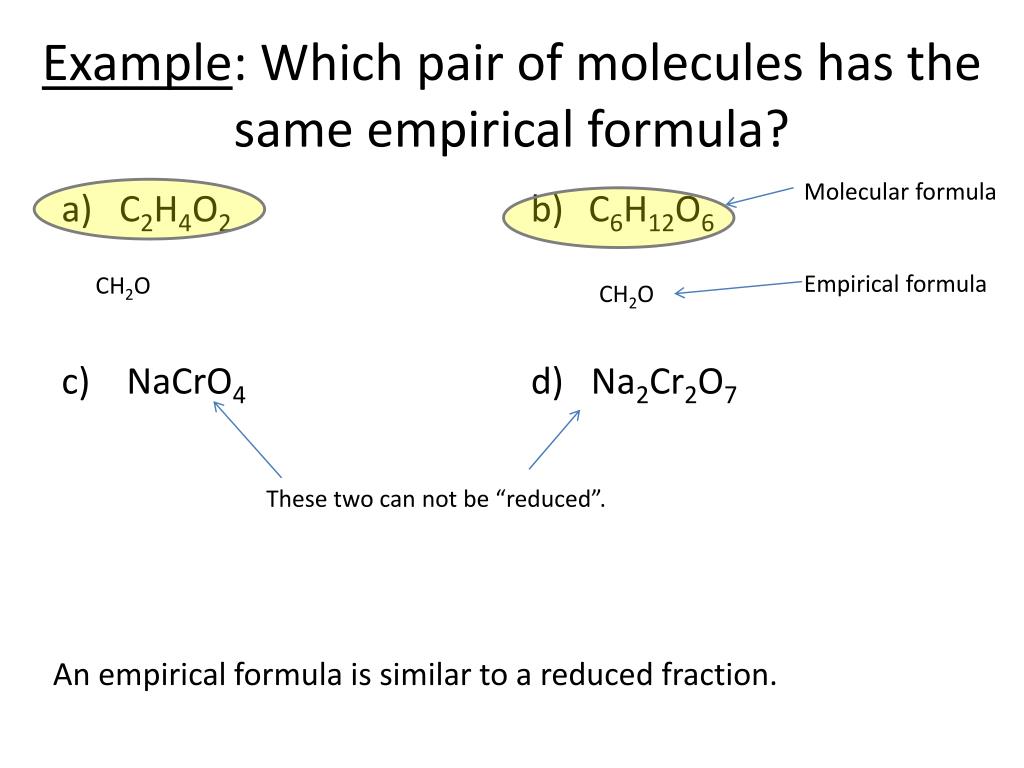
Which Pair Of Compounds Has The Same Empirical Formula
Hey there, science curious folks! Ever wonder if there's a hidden connection between seemingly different things? Well, get ready for a little chemical magic trick that's surprisingly fun. We're talking about a concept that might sound a bit fancy, but it's actually super cool when you break it down.
Imagine you have two different recipes for delicious cookies. They might look and taste a bit different, but what if the basic building blocks of those recipes were exactly the same? That's kind of what we're diving into today. It's all about finding a special pair of compounds that share a secret identity.
This isn't about super complicated lab experiments or memorizing tons of chemical names. Nope, this is more like a fun puzzle. We're going to explore how different substances can have the same fundamental recipe. Think of it as finding two friends who look totally different but have the same favorite color or the same goofy laugh. It’s that kind of delightful similarity!
So, what exactly are we looking for? We're on the hunt for compounds that have the same empirical formula. Don't let that phrase scare you! It's actually a straightforward idea. It means that the simplest whole-number ratio of atoms in those compounds is identical.
Think of it like this: you have a bag of LEGO bricks. You can build a small car or a tall tower with those same bricks. The bricks themselves are the basic units, and how you arrange them creates different structures. In chemistry, the atoms are the LEGO bricks, and the empirical formula tells you the simplest ratio of those bricks.
Why is this so engaging, you ask? Because it reveals a hidden elegance in the universe. It shows that sometimes, complexity arises from simple, repeated patterns. It’s like discovering a repeating motif in a beautiful piece of music or a pattern in nature, like the way ferns unfurl. There's a quiet beauty in shared foundations.
It's also entertaining because it challenges our initial perceptions. When you see two different chemical formulas, you might assume they are completely unrelated. But then, bam! You discover they have this fundamental link. It’s a little “aha!” moment that’s incredibly satisfying.
This shared empirical formula is special because it hints at shared origins or potential transformations. It's like finding out two different kinds of candy are made from the same core ingredients, just processed a bit differently. There’s a fascinating story waiting to be told about their relationship.

Let’s get into the nitty-gritty, but in a super chill way. We're talking about specific pairs of compounds that have this awesome trait. One such pair involves a gas we breathe out and a common sugar. Sounds intriguing, right?
The first compound is carbon dioxide. You know, the stuff we exhale when we breathe. It's made of carbon and oxygen atoms. Its chemical formula is CO₂. This means for every one carbon atom, there are two oxygen atoms.
Now, let’s look at our sweet friend, glucose. This is a simple sugar, super important for energy. Its chemical formula is C₆H₁₂O₆. This looks much more complicated, doesn't it? It has six carbons, twelve hydrogens, and six oxygens.
Here’s where the magic happens. When we look for the simplest whole-number ratio of atoms in glucose (C₆H₁₂O₆), we can divide all the numbers by the biggest common factor, which is 6. So, 6 divided by 6 is 1, 12 divided by 6 is 2, and 6 divided by 6 is 1. This gives us a ratio of C₁H₂O₁.
And guess what? The empirical formula for carbon dioxide (CO₂) is already the simplest ratio! It has one carbon and two oxygens. So, the empirical formula for carbon dioxide is also CH₂O!
See? Carbon dioxide (CO₂) and glucose (C₆H₁₂O₆) share the same empirical formula: CH₂O. Isn't that neat? It’s like finding out that a simple, everyday greeting and a complex, beautiful poem are built using the same fundamental letters.
This is what makes exploring chemistry so rewarding. It’s not just about memorizing facts; it's about uncovering these elegant connections. It’s like finding secret passages in a familiar house.
The fact that carbon dioxide, a gas we exhale, and glucose, a fundamental energy source for life, share this basic building block ratio is pretty remarkable. It hints at deeper biological and chemical processes where these substances are related.
Think about photosynthesis. Plants take in carbon dioxide and water to make glucose and oxygen. There’s a constant cycling and transformation happening, and these shared empirical formulas are a subtle clue to that interconnectedness.
Another fun pair to consider involves acetylene and benzene. These are both important organic compounds. Acetylene is a simple alkyne, often used in welding. Its chemical formula is C₂H₂.
When we simplify the ratio of atoms in acetylene (C₂H₂), we divide both numbers by 2. This gives us CH. So, the empirical formula for acetylene is CH.

Now, let’s look at benzene. This is a more complex molecule, a ring structure with six carbons and six hydrogens. Its chemical formula is C₆H₆.
If we simplify the ratio of atoms in benzene (C₆H₆), we can divide both numbers by 6. This also gives us CH!
So, acetylene (C₂H₂) and benzene (C₆H₆) also share the same empirical formula: CH. It’s another fantastic example of how different structures can arise from the same basic atomic ratio.
Why is this so entertaining? Because it’s a reminder that the world is full of surprises. It’s like finding out that two completely different songs you love were written by the same composer, or that two distinct paintings were made with the same palette of colors. It sparks curiosity and makes you want to look closer.
It’s special because it’s a tiny window into the fundamental laws that govern matter. It suggests that there are underlying principles that simplify seemingly complex realities. It’s the scientific equivalent of finding a simple, elegant solution to a challenging problem.

This concept of empirical formulas is like a secret decoder ring for chemists. It helps them understand the basic composition of compounds, even when the full molecular structure is unknown or very complex. It's a foundational piece of the chemical puzzle.
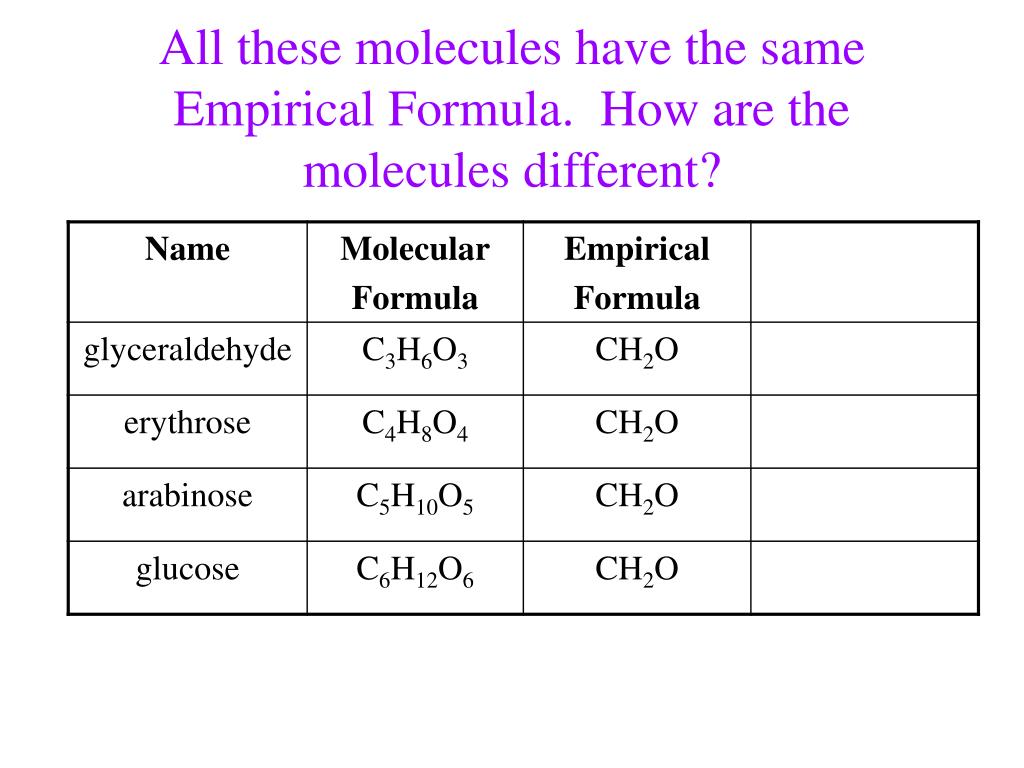
The pairs we’ve discussed, like carbon dioxide/glucose and acetylene/benzene, are just a couple of examples. There are many more out there waiting to be discovered, each with its own little story to tell about shared molecular foundations.
So, the next time you encounter different chemical compounds, take a moment to think about their empirical formulas. You might just uncover a hidden connection that makes you smile. It’s a simple idea, but it’s a gateway to understanding the beautiful, interconnected nature of chemistry.
It’s the little things, the shared ratios, that can make science feel so accessible and engaging. It’s like a scavenger hunt for fundamental truths. And honestly, who doesn't love a good hunt?
This exploration into shared empirical formulas is a perfect example of how looking at things from a different angle can reveal surprising and delightful relationships. It’s a friendly reminder that even in the world of atoms and molecules, there’s room for elegant simplicity and fascinating connections.
So go ahead, be curious! Poke around a little. You might just find yourself delighted by the hidden harmonies in the chemical world. It’s a journey that’s both informative and, dare we say, a little bit fun!
