Which One Of The Following Is A Bronsted Lowry Acid

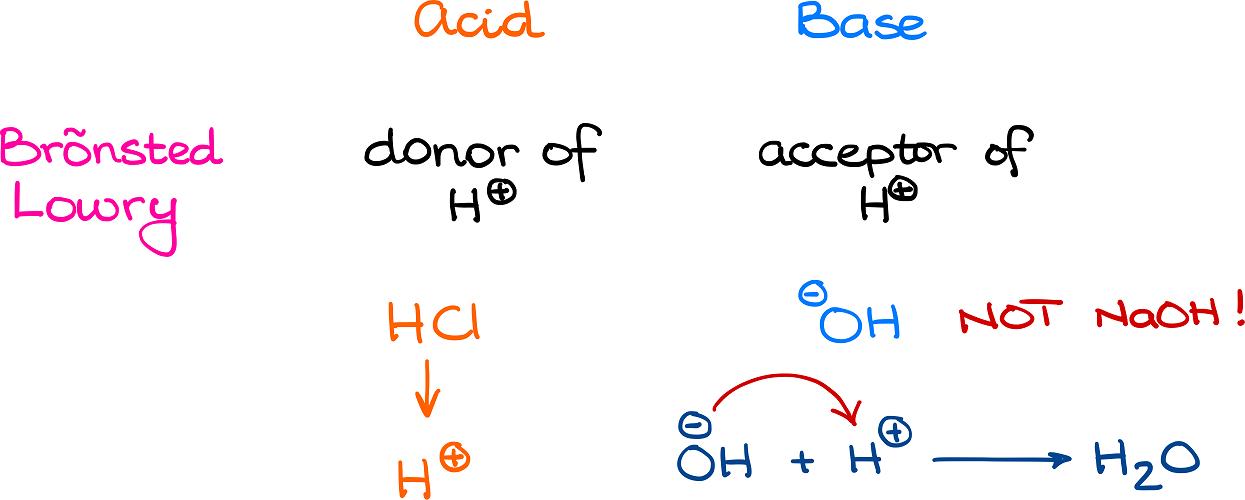
Okay, friends, gather 'round! We're diving into a little bit of science today. Don't worry, it's the fun kind. The kind that makes you feel smart without actually having to crack open a textbook. We're talking about something called Brønsted-Lowry acids. Sounds fancy, right? Like something you'd order at a really intimidating cafe. But it's actually way simpler than it sounds. Imagine a superchill scientist named Brønsted and his equally cool buddy Lowry. They had this brilliant idea about how acids work. And guess what? It's all about sharing. Like sharing your last slice of pizza. Or your Wi-Fi password. Or that really good meme. Acids, in their world, are basically proton donors. They're the generous ones. The ones who are always ready to lend a hand, or in this case, a proton.
Now, what's a proton in this context? It's not the guy who sits in the front row and asks all the questions. It's a positively charged particle. Think of it like a tiny, energetic cheerleader for molecules. And acids are the ones who say, "Here, have a cheerleader! You look like you could use some positive energy!" It's really quite touching, if you think about it. So, when we talk about which one of the following is a Brønsted-Lowry acid, we're basically asking: Which one of these guys is the generous proton sharer? Who's the molecule that's happy to give up a proton and make another molecule feel better?
Let's imagine a party. A molecular party. Everyone's chilling, maybe dancing to some funky electron beats. Then along comes a molecule that's a bit of a loner. It's got an extra proton, like a spare party favor. And then, another molecule walks by, looking a little down. It's missing that certain oomph. That's where our Brønsted-Lowry acid steps in. It sees its fellow molecule struggling and says, "Hey, buddy! Take this proton. It'll cheer you right up!" And poof! A chemical reaction happens. It's like a tiny act of kindness in the microscopic world.
So, we're presented with a list. A lineup of suspects. And we have to pick out the one who's the ultimate proton donor. The one who’s basically wearing a sign that says "Free Protons Here!" It's like a game of chemical musical chairs, but instead of chairs, it's protons. And everyone wants a seat. Or, in this case, everyone wants to give one away. It’s a bit of an unpopular opinion, but sometimes, being the giver is the coolest thing you can be.
Think about it in everyday terms. If you have a cookie, and your friend looks hungry, you offer them a piece, right? You're donating a little bit of your cookie goodness. That's kind of what a Brønsted-Lowry acid does with its protons. It's sharing its molecular deliciousness. It's not hoarding its protons like a dragon with its gold. It's out there, spreading the proton love. And that, my friends, is a noble cause.
Now, let's get down to the nitty-gritty, but keep it light, okay? We're not going to get bogged down in complex formulas. We're just going to look at the players. Imagine you're at a talent show. And you've got a bunch of contestants. You've got a juggler, a singer, a magician. And then you've got someone who’s really good at giving away free samples. That's our Brønsted-Lowry acid. It’s the one with the generous spirit.
Sometimes, it's really obvious. Like, if you see a molecule that’s practically begging to get rid of a hydrogen atom. It’s like it’s got a hydrogen on its back and it’s saying, “Please! Take it! I can’t carry it anymore!” That’s your classic Brønsted-Lowry acid right there. It’s the tired traveler with too many bags.

Other times, it's a little more subtle. Like a ninja in the night. It might not look like it's giving anything away, but if you look closely, it's got that proton ready to spring. It’s a master of disguise, this acid. You have to pay attention to the details. Like noticing when someone’s had a haircut. You might not have seen the haircut itself, but you know it happened.
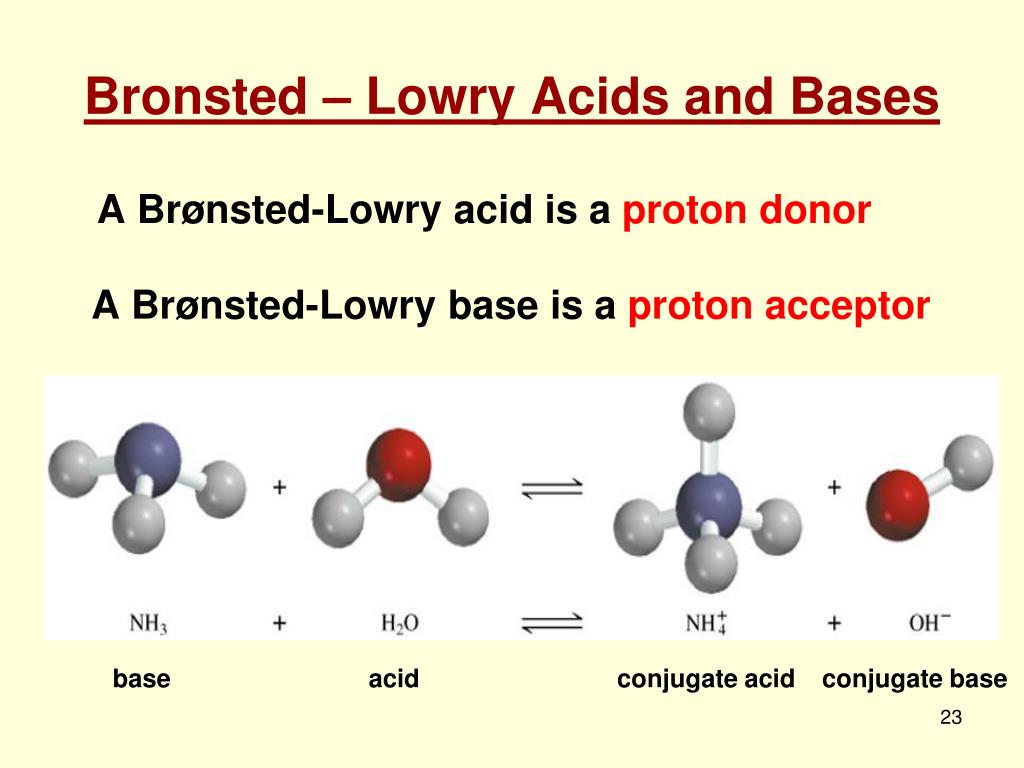
It's all about the transfer of a proton. Simple as that. Like passing a baton in a relay race. Except the baton is a proton, and the runners are molecules. And the finish line is a chemical reaction. Sounds exciting, doesn't it?
Bronsted-Lowry Acid-Base Equilibrium — Organic Chemistry Tutor
So, when you’re faced with that question, “Which one of the following is a Brønsted-Lowry acid?”, just think about who’s feeling generous. Who’s ready to share the wealth, or in this case, the positive charge? Who’s the molecule that’s most likely to say, “Here, you take this proton. You clearly need it more than I do.” It’s about empathy, really. Molecular empathy.
We’re not talking about things that just sit around and look acidic. Like a lemon. Lemons are acidic, sure. But are they Brønsted-Lowry acidic? That’s the million-dollar question, isn't it? It’s about the action. The giving. The sharing. It’s not about what you are, it’s about what you do.
So, next time you hear the term “Brønsted-Lowry acid,” don't get intimidated. Just picture a friendly molecule handing out protons like they’re candy. It’s the ultimate sharer. The proton philanthropist. The molecule with a heart of gold… or rather, a heart of positive charge. And that, in my humble, possibly unpopular opinion, is pretty darn cool.