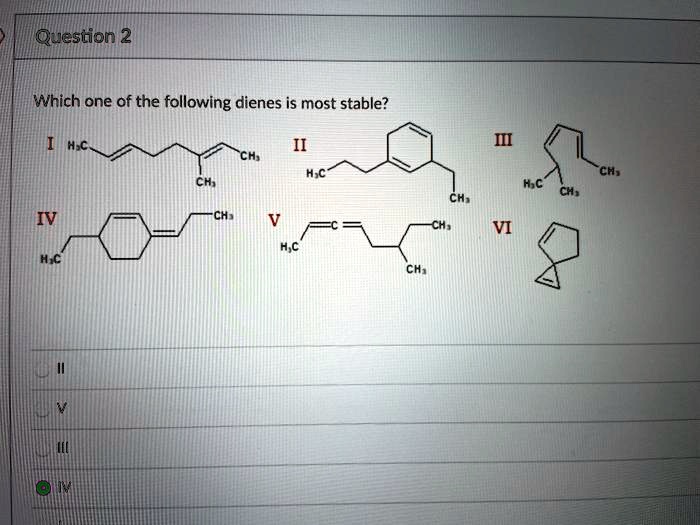
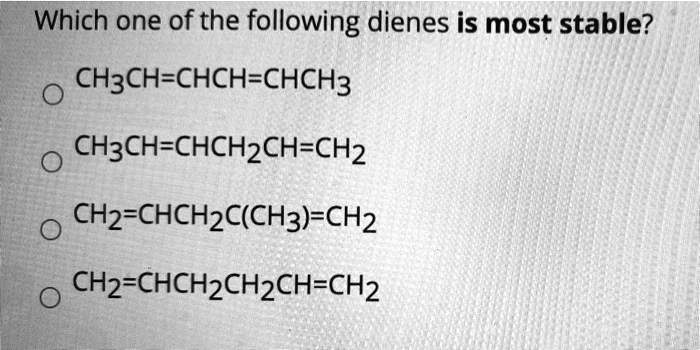
Which One Of The Following Dienes Is Most Stable
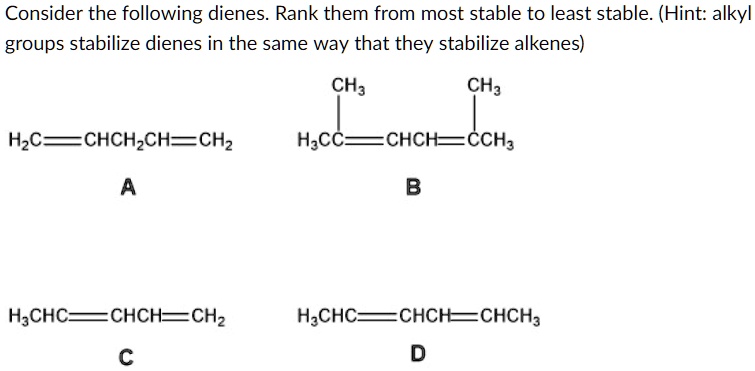
Ever wondered about the hidden world of molecules and why some are just… better than others? It’s not all about fancy lab coats and complex equations! Believe it or not, understanding molecular stability is like solving a fun, tiny puzzle with huge implications. Think of it like figuring out which building blocks are the sturdiest or which recipe is most likely to succeed. In the realm of organic chemistry, a key concept is diene stability. Now, "diene" might sound a bit intimidating, but it simply refers to a molecule with two double bonds. And when we talk about their stability, we're essentially asking: which of these molecules is the most relaxed, the most content, the least likely to spontaneously fall apart or react wildly?
This isn't just a nerdy academic pursuit. Understanding why certain dienes are more stable than others helps chemists predict how molecules will behave, design new materials, and even develop more efficient chemical processes. It's like knowing that a well-built house will withstand a storm better than a rickety shack. For the everyday person, this translates to everything from the plastics in your everyday gadgets to the ingredients in your medicines. The principles of molecular stability are woven into the fabric of modern life.
So, let’s dive into the fun part: figuring out which one of the following dienes is the most stable. We’re going to be looking at some contenders and unpacking the reasons behind their inherent chill-out factor. The benefit here is gaining a glimpse into the forces that govern the molecular world, making abstract chemistry feel a little more tangible and, dare we say, exciting!
The Contenders: A Look at Diene Stability
Imagine you have a group of friends, and you’re trying to figure out who’s the most relaxed and easy-going. In chemistry, we do something similar with molecules! We’re going to consider a few different types of dienes and see why one might be considered more “stable” than the others. Stability in this context means the molecule has a lower energy state, making it less reactive and more content to just… be.
First up, let's consider cumulated dienes. Think of these like a chaotic pile-up of double bonds. In a cumulated diene, the two double bonds share a central carbon atom. An example is allene, CH2=C=CH2. While perfectly valid molecules, this arrangement puts a bit of strain on the system. The double bonds are quite close, and their electron clouds can get a little… frisky with each other, leading to higher energy and thus, less stability. They're like that friend who's always a bit on edge, ready to jump into action (or drama!).
Next, we have conjugated dienes. These are the stars of our stability show! In a conjugated diene, the two double bonds are separated by just one single bond. A classic example is 1,3-butadiene, CH2=CH-CH=CH2. Now, why are these guys so special? It’s all about something called resonance. The electrons in the pi bonds (the ones involved in the double bonds) aren't just stuck in their own little worlds. They can actually delocalize, meaning they spread out over the entire system. Imagine a group of friends sharing their energy and resources – it makes the whole group stronger and more stable.
This spreading out of electrons, this resonance, lowers the overall energy of the molecule. It's like having a well-distributed workload; no single person (or bond) is overwhelmed.
This delocalization makes conjugated dienes significantly more stable than their cumulated cousins. They're the chill, laid-back friends who are happy to go with the flow.
Finally, we might encounter isolated dienes. These are dienes where the two double bonds are separated by two or more single bonds. An example would be 1,4-pentadiene, CH2=CH-CH2-CH=CH2. In these molecules, the double bonds essentially act independently. There’s no special interaction or delocalization between them. They’re like friends who live in different neighborhoods and only see each other occasionally – their interactions are limited, and they don’t offer the same kind of shared stability as the conjugated system. They fall somewhere in between cumulated and conjugated dienes in terms of stability.
So, when we ask which one is the most stable, we're looking for the arrangement that allows for the greatest electron delocalization and therefore the lowest energy. That, my friends, points squarely towards the conjugated dienes. Their ability to share electron density across multiple bonds is the secret to their superior chill factor. It's a beautiful example of how subtle differences in molecular structure can lead to significant differences in behavior and stability, a concept that’s both fascinating and incredibly useful in the world of chemistry!
