Which One Of The Following Compounds Is Aromatic

Alright, settle in, grab your latte, and let's talk about something that sounds like it belongs in a secret wizard's spellbook, but is actually just… well, chemistry. We're diving into the weird and wonderful world of aromatic compounds. And by "diving," I mean gently dipping our toes in, because this stuff can get intense. Think of it like trying to understand quantum physics after one too many espressos – a little fuzzy, but undeniably fascinating.
So, the big question, the one that keeps chemists up at night (probably fueled by more coffee), is: which one of these mystery compounds is aromatic? It's like a chemistry-themed game of "Who's the Divo?" or "Guess Which Molecule is a Rock Star?" We've got a lineup of contenders, and only one of them truly embodies that elusive "aromatic" vibe. And trust me, this isn't about smelling nice, although some aromatics do smell fantastic. We're talking about a whole different kind of charisma.
What's the Big Deal with Aromaticity Anyway?
Before we start pointing fingers at our unsuspecting molecules, let's demystify "aromatic." It’s not just about smelling like a rose garden, although that’s a nice bonus. In chemistry, aromaticity is a property that makes a molecule unusually stable. Like, super stable. Imagine a molecule that’s so chill, so unbothered by the chaotic forces of the universe, that it’s basically meditating on a mountaintop. That’s an aromatic compound for you.
Think of it like this: most molecules are like a bunch of toddlers at a birthday party – bouncing off the walls, eager to react, constantly getting into mischief. Aromatic molecules, on the other hand, are like the wise old librarian who just sips her tea and observes the chaos with a knowing smile. They've got this internal harmony, this perfect little arrangement that makes them reluctant to change. They’ve achieved peak molecular zen.
This stability comes from a special arrangement of electrons, specifically a phenomenon called delocalization. Imagine electrons not being tied down to just one or two atoms, but instead, they’re like a free-spirited traveler, roaming all over the molecule, making friends with everyone. This electron-sharing party makes the whole molecule feel more comfortable and, therefore, more stable.
The Rules of the Aromatic Club
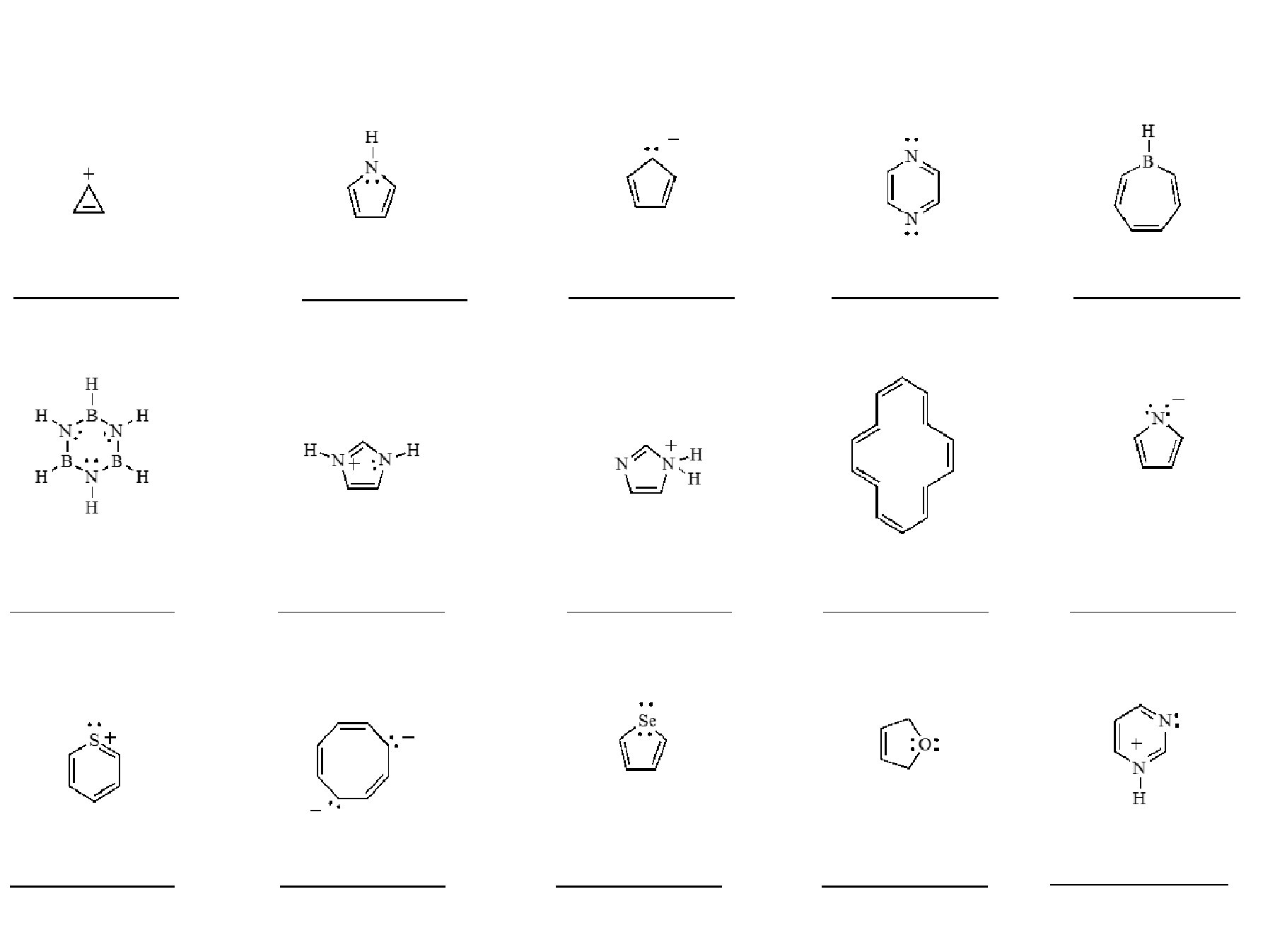
Now, every cool club has its membership requirements, right? The aromatic club is no different. To get into this exclusive group, a molecule has to follow a few simple (but crucial) rules:
- It needs to be cyclic: It’s gotta be a ring, a closed loop. No open chains allowed. Think of it as needing a membership card that’s shaped like a circle.
- It needs to be planar: The atoms in the ring have to lie flat, like a perfectly ironed shirt. No bumps or wiggles allowed. They gotta be in the same plane.
- It needs to have (4n + 2) pi electrons: This is the real kicker, the secret handshake. 'N' is just some whole number (0, 1, 2, etc.). So, you're looking for 2, 6, 10, 14… electrons that are part of this special pi system. This is where the electron-sharing party happens.
- Every atom in the ring must have a p-orbital that can overlap: This is like needing to be able to hold hands with everyone in the circle. Each atom needs to be able to contribute to that electron-sharing network.
If a molecule checks all these boxes, BAM! It’s officially an aromatic superstar. It’s got the swagger, the stability, and the electrons doing the conga line. It’s the Beyoncé of the molecule world.
Meet Our Contenders!
Okay, let’s look at the suspects. Imagine these are our contestants in a high-stakes chemistry beauty pageant. We've got:
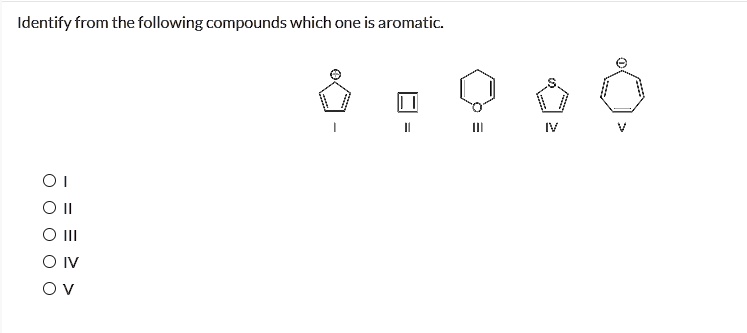
Compound A: Cyclohexane. This is your basic, everyday six-membered ring. It’s like the comfy sweater of molecules. Stable, sure, but is it… aromatic? Let’s check the rules. Cyclic? Yes. Planar? Well, it can adopt a somewhat flat shape, but it’s more of a floppy chair. And the pi electrons? Uh oh. Cyclohexane is saturated, meaning all its carbon atoms are happily holding onto as many hydrogens as possible. No room for a delocalized electron party here. So, it’s a solid "no" for aromaticity. It’s more like the "regular" in "regular coffee."
Compound B: Cyclopentadienyl anion. Now this one's interesting. It's a five-membered ring, and it’s got a little something extra: a negative charge. That negative charge means it has extra electrons. Let’s see. Cyclic? Yes. Planar? Yep, it can be pretty flat. Pi electrons? It has some double bonds and that extra pair of electrons from the negative charge. When we crunch the numbers for the (4n + 2) rule, with n=1, we get 6 pi electrons. Six! That’s a magic number in the aromatic world. So, this little anion is definitely a contender. It’s got that potential for electron-sharing bliss. It's like the contestant who brought their own glitter for the talent show.
Compound C: Cyclooctatetraene. This one’s an eight-membered ring with alternating double and single bonds. Looks promising, right? Cyclic? Check. Pi electrons? It’s got 8 pi electrons. Now, let's plug that into our magic formula: (4n + 2). If we try n=1, we get 6. If we try n=2, we get 10. So, 8 doesn't fit our (4n + 2) rule. It’s actually following a different, less stable rule called Hückel’s rule for anti-aromatic compounds (which is like being the exact opposite of cool, but that’s a story for another coffee). Plus, this molecule isn't even planar! It's more like a crinkled-up piece of paper. So, for our aromatic purposes, this one’s a "nope." It’s the contestant who showed up in pajamas.
Compound D: Benzene. Ah, benzene. The OG. The granddaddy of aromaticity. You’ve probably heard of it, maybe even seen its iconic hexagon-with-a-circle structure. It’s the molecule that made chemists go, "Whoa, what is that?" Cyclic? Absolutely. Planar? Perfectly flat. Pi electrons? It’s got 6 pi electrons (from its alternating double and single bonds). Plug n=1 into our (4n + 2) rule, and what do you get? Yep, 6! Benzene nails every single rule. It’s the perfectly posed, perfectly radiating superstar. It’s the diva who actually deserves the crown.
The Verdict is In!
So, after all that detective work, who is our aromatic champion? Drumroll, please… It’s Benzene!
While the cyclopentadienyl anion is very close to achieving aromatic glory (and in its anionic form, it is aromatic!), benzene is the classic, the undisputed king of aromatic stability. It’s the molecule that inspired a whole field of chemistry. It’s the one that smells like industrial solvents but is found in everything from medicines to plastics. It’s like finding out your quiet neighbor is secretly a concert pianist with a Nobel Prize.
The other compounds are either too floppy, don't have the right number of electrons for that special dance, or are just plain uninvited to the electron-sharing party. They might be good, they might be useful, but they don't have that extra spark, that je ne sais quoi, that makes a molecule truly aromatic.
So next time you’re pondering the mysteries of the universe, or just trying to decide between a croissant and a muffin, remember benzene. It’s a reminder that even in the seemingly mundane world of chemistry, there are molecules with extraordinary stability and a special kind of charm. They’re the rock stars, the divas, the zen masters – all rolled into one perfect, stable, and undeniably aromatic package.
