Which Of These Molecules Or Ions Exhibit Resonance

Ever feel like some molecules are just a little bit... indecisive? They can't quite make up their minds about where their electrons want to hang out. Well, guess what? That's exactly what's happening with this super cool phenomenon called resonance! It's like a molecule doing a little electron dance, shifting around to find the most stable groove.
Imagine a group of friends trying to decide where to go for pizza. One friend says pepperoni, another says mushroom, and a third insists on veggie. They can't settle on just one! So, what do they do? They compromise, maybe get a half-and-half, or even a combo that has a little bit of everything. Molecules with resonance are kind of like that, but with their electrons. Instead of one fixed arrangement of atoms and electrons, they exist as a sort of blended average of a few different possible arrangements. It's like they're saying, "I could be this, or I could be that, but really, I'm a bit of both!"
So, when we ask, "Which of these molecules or ions exhibit resonance?" we're basically looking for those electron-shy characters. We're on the hunt for the ones that are a little more fluid, a little more adventurous with their electron distribution. It's not about them being wrong or confused; it's about them being smarter and more stable because they can spread their electron love around.
Think about it like this: if you have a single, strong bond between two atoms, it's pretty straightforward. But if those electrons are able to wiggle and jiggle between multiple atoms, it's like they're sharing the load. This sharing makes the molecule or ion more chill, more at peace, and therefore, more stable. It's the chemical world's way of saying, "Hey, sharing is caring, and it makes you stronger!"
"Resonance is like a molecular personality trait that makes certain molecules extra special."
And honestly, it's a blast to figure out which ones are doing the resonance shuffle. It involves looking at their structures, seeing where the electrons could go, and realizing that they're not sticking to just one place. It’s like being a detective, but instead of solving a mystery, you’re uncovering the hidden electron pathways!
.jpg)
Let's take a peek at some of the usual suspects. You might have heard of ozone, that layer way up in the sky protecting us from the sun's harsh rays. Ozone is a fantastic example! It's not just one specific way it's put together; its electrons are happily delocalized, meaning they're spread out over all the atoms. This makes it a pretty resilient molecule.
Then there are ions, which are just atoms or molecules with a little electrical charge. Take the carbonate ion, for instance. It's got a negative charge, and those negative charges get to play around and spread out across the oxygen atoms. It’s like a party where everyone gets a chance to hold the sparkly decorations. This spreading of charge is a big clue that resonance is at play.
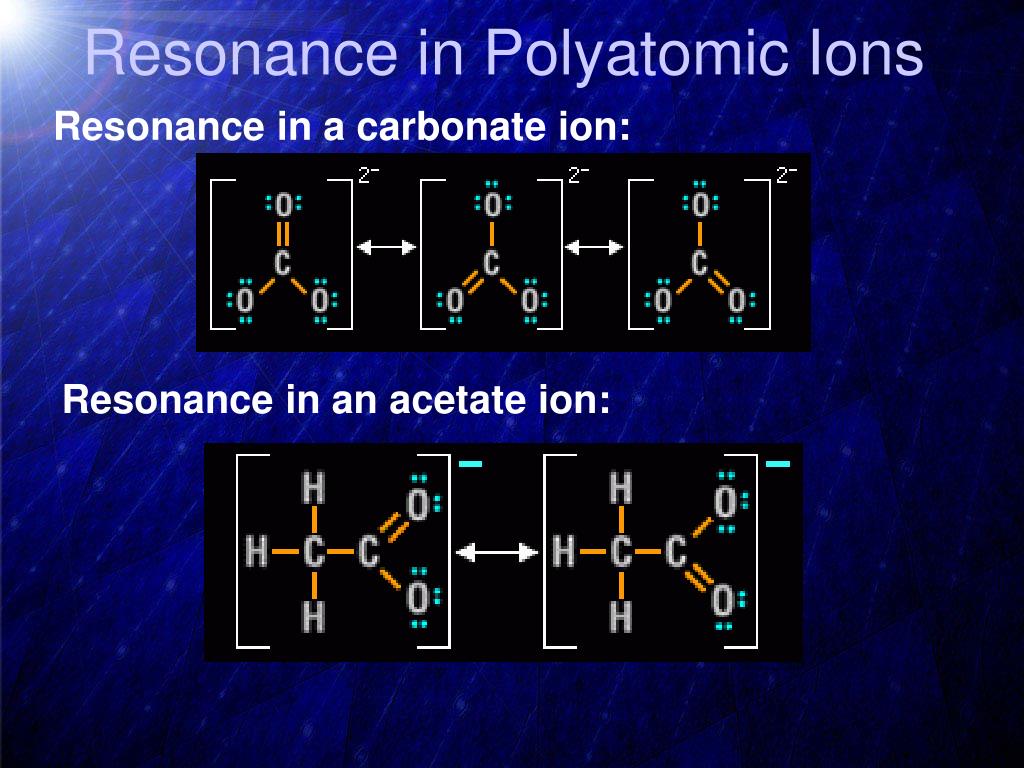
Another common one is the nitrate ion. Just like carbonate, its negative charge is distributed among the oxygen atoms, making it stable and flexible. It's a classic example you'll see in chemistry textbooks, and for good reason! It perfectly illustrates the concept.
What's so engaging about this is the idea of delocalization. It’s a fancy word, but it just means spreading out. When electrons are delocalized, they aren't tied down to just two atoms forming a single bond. They can roam freely, forming partial bonds across three or more atoms. This creates a much more energetic and stable situation. It's like having a superpower that allows you to be in multiple places at once, in a way!

So, when you look at a molecule or ion and see a situation where electrons can be moved around to create different, but equally valid, Lewis structures, that’s your cue! It's like seeing a blinking "Resonance Ahead!" sign. These structures aren't real, independent entities. They're just our best attempts to draw a picture of something that’s more complex and dynamic.
The true structure of a resonance-stabilized molecule is a hybrid of all these contributing structures. It's like tasting a delicious fruit punch – you can identify hints of pineapple, orange, and grape, but the punch itself is its own unique flavor, a blend of all those individual fruits. The molecule is more stable than any of its individual contributing structures would be on their own. This extra stability is the "magic" of resonance.

"It's the molecular equivalent of having your cake and eating it too, but then also getting another slice!"
Why does this matter? Well, it explains so much about how molecules behave! It affects their reactivity, their physical properties, and how they interact with other molecules. Understanding resonance is like unlocking a secret level in the game of chemistry. It helps us predict how things will react and why they form the way they do.
So, the next time you're looking at a chemical structure and you think, "Hmm, those electrons could totally be over there too," you might just be looking at a molecule that exhibits resonance! It’s a wonderful reminder that in the world of chemistry, things are often not as simple as they first appear, and that's precisely what makes it so endlessly fascinating and fun to explore. Keep your eyes peeled for these electron-dancing molecules; they’re the stars of the show!
It's all about recognizing the potential for electron movement. If you can draw more than one valid Lewis structure for a molecule or ion by simply moving electrons (and sometimes rearranging double and single bonds), then bingo! You've likely found a resonance performer. It's a delightful puzzle that reveals the underlying flexibility and stability of these tiny chemical building blocks.
