Which Of The Molecules And Polyatomic Ions Cannot Be Adequately
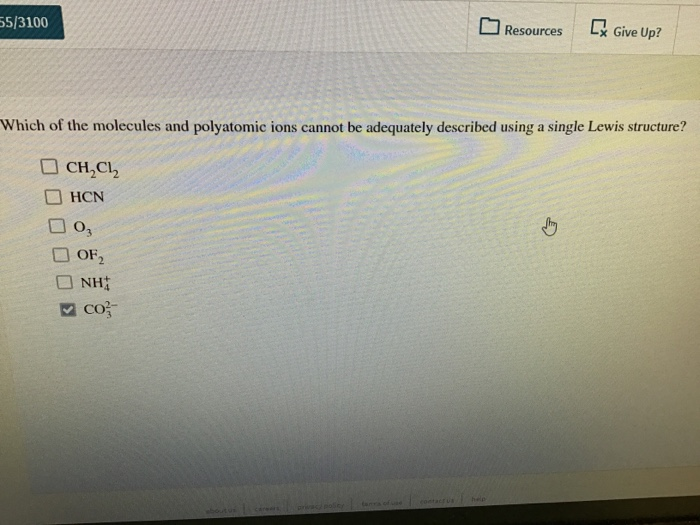
Hey there, fellow science enthusiasts! Grab your favorite mug, because we're about to dive into something that can be a little… tricky. You know how sometimes you look at a molecule, or a little group of atoms holding hands (that's what polyatomic ions are, basically!), and you think, "Yeah, I can totally draw this thing out, map its electrons, figure out its shape"? Well, sometimes, my friends, those molecules and ions just don't play by the rules. They're the rebels of the chemical world, the ones that make you scratch your head and go, "Huh?"
Today, we're going to chat about which of these guys just cannot be adequately described using our usual, go-to methods. Think of it like trying to fit a square peg into a round hole. It just doesn't quite work, does it? And that's totally okay! The universe is full of wonderfully weird stuff, and chemistry is no exception.
The Usual Suspects: What We Normally Do
Before we get to the troublemakers, let's quickly recap what we usually do. You know, the stuff that works like a charm for, like, 90% of molecules. We're talking about drawing out those Lewis structures, right? Those little dots and lines showing all the electrons and bonds. It's like a blueprint for a tiny, molecular house. And then, from that, we can usually figure out the VSEPR theory. That's Valence Shell Electron Pair Repulsion theory, for those who need a refresher. Basically, it says that electron pairs around a central atom want to be as far apart as possible. Like tiny magnets repelling each other! It’s surprisingly effective, predicting whether things will be linear, bent, trigonal planar, tetrahedral… you get the picture.
It’s all about minimizing that electron-electron repulsion. Super logical, right? Most of the time, it gives us a really good idea of the molecule's 3D shape. And that shape, oh man, that shape is everything. It dictates how the molecule interacts with other molecules, its properties, its reactivity… you name it!
So, why are we even talking about exceptions? Because, as we know, life isn't always textbook-perfect. And neither is chemistry. Sometimes, our trusty Lewis structures and VSEPR predictions start to wobble. They don't quite capture the full story, or they lead us down a path that feels… incomplete.
When the Blueprint Doesn't Quite Fit: The Challenges
What kind of molecules are we talking about here? Well, it's often when things get a bit more complicated than your average, everyday molecule. Think about molecules with an odd number of valence electrons. Uh oh. That immediately throws a wrench in the whole "perfect octet" idea. You know, where atoms love to have eight electrons in their outer shell? Not so easy when you've got an odd number. Someone's always going to be a little bit short, or a little bit extra. It's like trying to pair up dancers at a party when there's one person left over. Awkward!
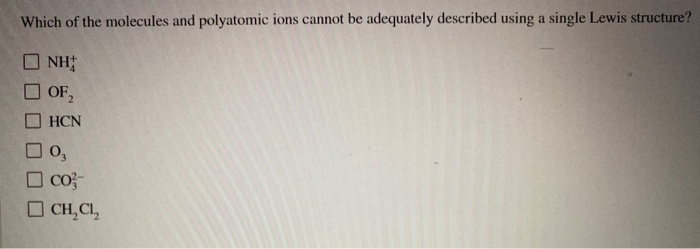
Another one? Molecules with expanded octets. Now, you might think, "Wait a minute, I thought octets were sacred!" For many elements, especially the second-period ones like carbon and nitrogen, yes, they really stick to that octet rule. But when you get to heavier elements, like phosphorus or sulfur or chlorine, they've got those extra "d" orbitals hanging around, just waiting to be used. So, they can sometimes shove more than eight electrons around themselves. It’s like they’ve got a bigger house and can fit more furniture. Our simple Lewis structures sometimes struggle to accurately represent that extra furniture.
And then there are those really fascinating cases where a molecule could have multiple valid Lewis structures that are essentially equivalent. We call these resonance structures. Imagine you have a molecule, and you can draw its electron arrangement in a couple of different ways, and the actual molecule is like a hybrid, a blend of all these possibilities. Our simple dot-and-line drawings can't really show that blend. They’re like showing a picture of a cat and a picture of a dog, but not the actual creature that's a cat-dog hybrid (which, let's be honest, would be amazing). It’s like our tools are a little too simplistic to capture the full reality.
The Oddballs: Molecules That Make Us Think
So, let's get down to the nitty-gritty. Which molecules and polyatomic ions often give our standard models a bit of a headache? One of the most classic examples is Nitric Oxide (NO). Just… NO. It has 11 valence electrons. Eleven! Try to draw a Lewis structure for that and give everyone a nice, tidy octet. You can't! It’s inherently a radical, meaning it has an unpaired electron. This makes it super reactive. Our usual rules just don't quite handle that unpaired electron gracefully.
Then we have things like Sulfur Dioxide (SO₂). Now, if you just draw out a simple Lewis structure, you might end up with a sulfur atom with an expanded octet, and that's… fine. But the reality is, SO₂ exhibits resonance. The double bond isn't fixed to one oxygen atom; it's delocalized, spread out over both. So, the actual molecule is a blend of two possible structures. Our single Lewis structure doesn't tell the whole story about how those electrons are distributed.

What about Carbon Monoxide (CO)? This one is a bit more subtle. While it can be drawn with a triple bond and lone pairs, the formal charges you get can be a bit awkward. It suggests a stronger polarity than might be expected, and the triple bond, while strong, doesn't perfectly explain all of CO's behavior. It’s a molecule that’s a bit more complex than a simple double or triple bond might suggest.
Let’s talk about some polyatomic ions. Nitrate (NO₃⁻) is another prime example of resonance. You can draw it with one double bond and two single bonds, but the negative charge and the bonding are actually distributed equally among all three oxygen atoms. The real nitrate ion is a perfect equilateral triangle, with equal bond lengths and strengths. Our individual Lewis structures showing a single or double bond just don't do it justice.
And what about the Sulfite ion (SO₃²⁻)? Similar to SO₂, it shows resonance. The negative charge is spread out, and the sulfur-oxygen bonds are all the same length and strength. Again, the simple Lewis structure doesn't fully capture this delocalization.
Now, here's where it gets really interesting. Sometimes, molecules or ions can exist in different states or under different conditions, and their bonding might change. Or they might be involved in reactions where the initial drawing is just a starting point. Think about things that are highly unstable or transient intermediates in reactions. Our static Lewis structures are best for describing stable, ground-state molecules.

Beyond the Dots and Lines: What's the Bigger Picture?
So, when our Lewis structures and VSEPR theory start to falter, what do we do? Do we just give up? Of course not! We have other tools in our chemist's toolkit. We can delve into more advanced theories like Molecular Orbital (MO) theory. This is where things get really sophisticated. Instead of thinking about bonds between specific atoms, MO theory looks at the entire molecule as one big unit and describes the orbitals that electrons can occupy across the whole thing. It’s like looking at the entire concert hall for your musicians, rather than just their individual dressing rooms.
MO theory is fantastic for explaining things like the delocalization of electrons in resonance structures, or the magnetic properties of molecules with unpaired electrons (like NO!). It can also explain bonding in molecules that just don't fit the simple octet rule, even with expanded octets.
Then there's the whole field of Quantum Chemistry. This is the really, really deep dive. It uses complex mathematical models to describe the behavior of electrons in molecules. It’s the ultimate truth serum for molecular structure and bonding, but it’s also way beyond what you can sketch out with a pencil and paper. Think of it as the supercomputer analysis that backs up all our simpler theories.
Sometimes, the issue isn't even with the theory itself, but with how we're trying to apply it. If we're trying to predict the exact bond angles in a very large, complex organic molecule with our basic VSEPR, we might run into limitations. Steric hindrance, the bulky nature of groups of atoms, can push and pull on bonds in ways that simple electron repulsion doesn't fully account for.
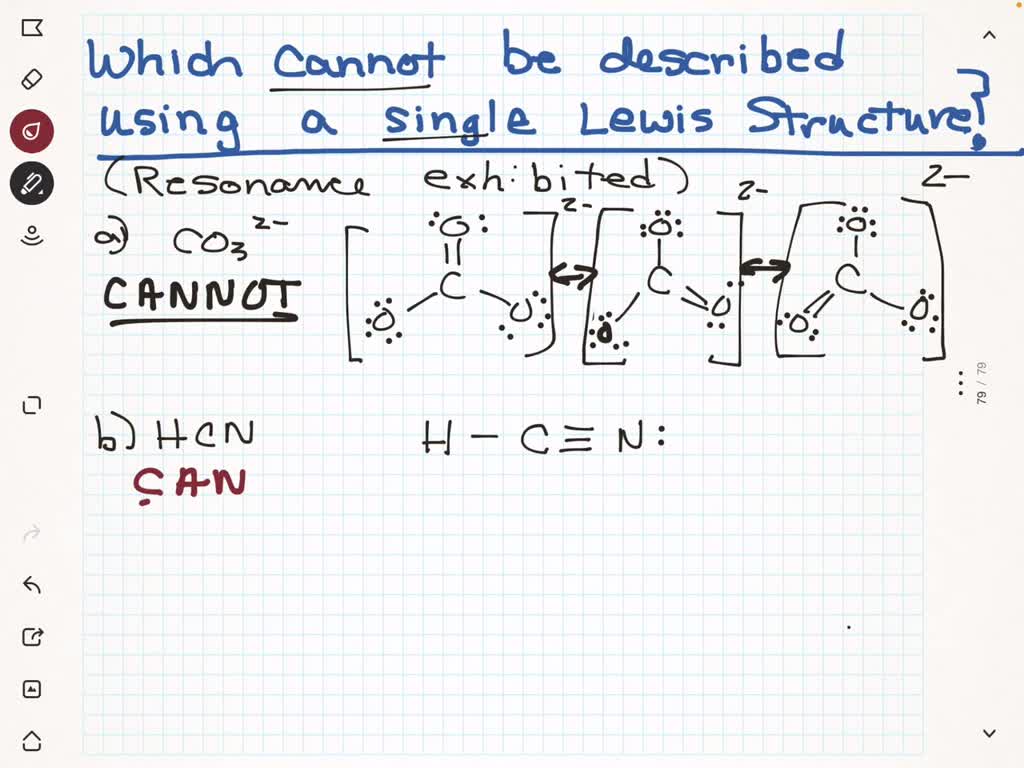
And let's not forget that chemical bonding isn't always perfectly covalent or ionic. There's a spectrum, and some molecules have characteristics of both. Our simplified models often have to choose one extreme or the other, and sometimes the reality is somewhere in between.
So, What's the Takeaway?
The main point is, it's okay for some molecules and ions to be a bit more complex than our introductory models can fully capture. It doesn't mean those models are "wrong"; they're just simplified representations. They’re incredibly useful for building our understanding, like learning to walk before you can run.
When you encounter something like NO, or a molecule with extensive resonance, or even something that seems to defy the octet rule in a way that doesn't fit the usual expanded octet exceptions, don't get discouraged! It just means you're getting to the more interesting, nuanced parts of chemistry. It's a sign that the universe is, indeed, wonderfully complex and beautiful in its chemical intricacies.
Think of it as a friendly reminder that there's always more to learn, more to explore. These "inadequately describable" molecules are the ones that push the boundaries of our knowledge and lead to new discoveries. They are the challenges that make chemistry such a dynamic and exciting field. So next time you're stuck on a Lewis structure or a VSEPR prediction, just take a breath, maybe have another sip of your coffee, and remember that sometimes, the most interesting things in chemistry are the ones that don't fit perfectly into our neat little boxes. They're the rebels, the artists, the ones that make us think a little harder. And that, my friends, is a beautiful thing.
