Which Of The Following Would Tend To Increase Membrane Fluidity

Hey there, science enthusiasts and fellow membrane-curious folks! Ever wondered what makes those tiny cellular borders, your cell membranes, so gosh-darn flexible? It’s like a groovy dance party happening at the molecular level, and today, we’re going to peek at the guest list and see who’s bringing the good vibes to increase the fluidity.
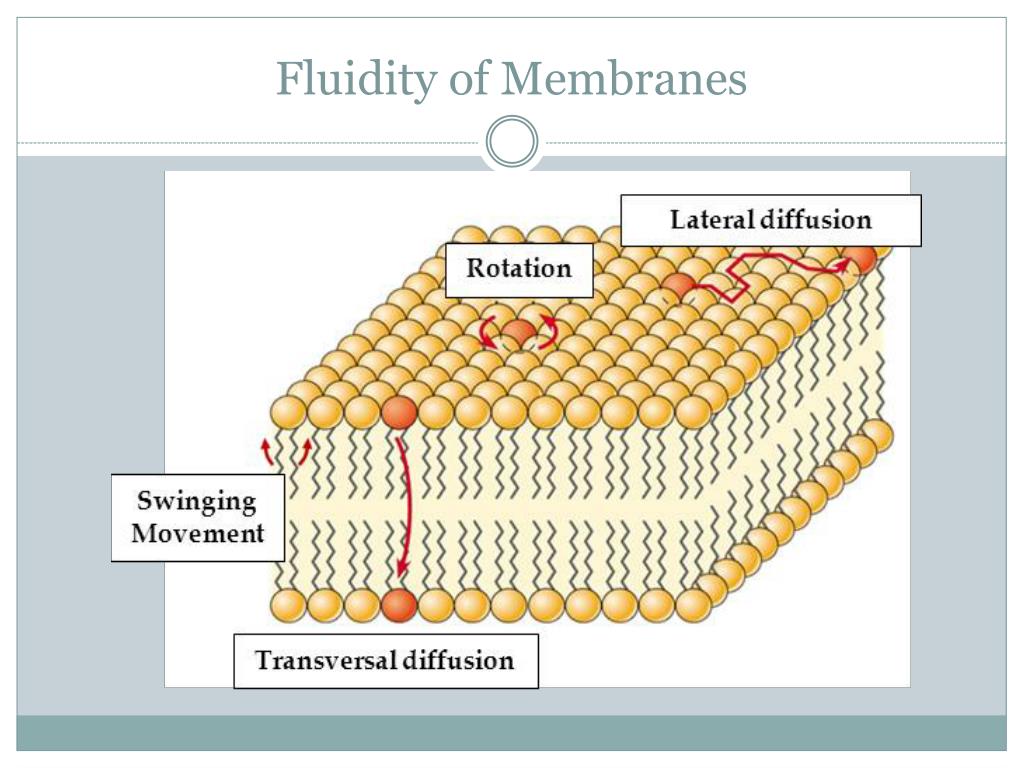
So, picture this: your cell membrane. It’s not a rigid brick wall, oh no. It’s more like a super-cool, semi-permeable mosaic, made up of all sorts of bits and bobs. The main players are these awesome things called phospholipids. They’re like little tadpoles with a water-loving head and a water-hating tail. These tadpoles love to hang out in a double layer, with their tails all tucked in, creating a barrier. Pretty neat, right?
But here’s the kicker: these phospholipids aren’t just stuck in place. They can wiggle, jiggle, and slide around. This constant motion is what we call membrane fluidity. And why is this so important? Well, a membrane that’s too stiff is like trying to navigate a frozen lake – not much fun, and probably not great for cell function. On the flip side, a membrane that’s too fluid is like a leaky sieve, and that’s definitely not what we want either. So, it’s all about finding that perfect balance, that sweet spot of wiggles and stability.
The Usual Suspects: What Makes Membranes Groovy?
Now, let’s dive into the nitty-gritty. We’re going to look at a few factors that can turn up the volume on membrane fluidity. Think of it like a DJ spinning some tunes that get the whole cell party started!
1. The Fatty Acid Tail Tale: Saturated vs. Unsaturated
This is probably the biggest player in the fluidity game. Remember those phospholipid tadpoles? Their tails are made of fatty acids, and these tails can come in two main flavors: saturated and unsaturated.
Saturated fatty acids are like the perfectly straight, well-behaved kids. They’ve got all the hydrogen atoms they can hold, so their tails are straight and can pack in really tightly. Imagine a box of perfectly aligned spaghetti. When you have a lot of saturated fatty acids in your membrane, all those straight tails snuggle up real close. This makes the membrane more rigid, less able to move around. Think of it as a really crowded dance floor – not much room to bust a move!
On the other hand, unsaturated fatty acids are the rebels of the fatty acid world. They have double bonds between some of their carbon atoms, which creates little kinks or bends in their tails. These kinks are like little elbows sticking out, preventing the phospholipids from packing too tightly. Imagine a box of spaghetti where some of the strands have little bends in them. They can’t stack up as neatly. This means the membrane is more spaced out, more able to wiggle and flow. More room for dancing! So, if you’re looking to increase fluidity, you’re going to want more of these bent, unsaturated fatty acids hanging around.
So, the take-home message here is: more unsaturated fatty acids = more kinks = more space = more fluidity! It’s like swapping out stiff, boring straight dancers for some groovy, bendy folk who love to do the wave.

2. Cholesterol: The Membrane’s Wingman (or Wingwoman!)
Ah, cholesterol. We often hear about it in relation to our arteries, but in the cell membrane, it plays a much more sophisticated role. Think of cholesterol as the ultimate wingman or wingwoman at the cell party. It’s a pretty interesting molecule because, depending on the temperature, it can actually do two very different things.
At warmer temperatures, when the membrane is already feeling pretty fluid and wiggly, cholesterol gets in there and acts like a brake. It tucks itself in between those phospholipids, kind of like a buffer. It’s saying, “Whoa there, slow down a bit, everyone!” By limiting the movement of the phospholipids, cholesterol reduces membrane fluidity at higher temperatures. It prevents the membrane from becoming too floppy and losing its structural integrity.
But here’s the cool part: at cooler temperatures, when the membrane starts to get all stiff and chilly, cholesterol becomes the party starter! It wedges itself in between the phospholipids and actually prevents them from packing too tightly together. It’s like saying, “Hey, don’t huddle up so much, spread out a little!” In this scenario, cholesterol increases membrane fluidity at lower temperatures. It’s like a tiny, molecular thermostat, helping the membrane maintain its optimal fluidity regardless of the external temperature.
So, while cholesterol isn't a simple "increase fluidity" or "decrease fluidity" factor, it’s a crucial regulator. It’s like a smart bouncer who knows when to let people mingle and when to tell them to take it easy. If we're talking about increasing fluidity specifically, we'd generally consider its role at cooler temperatures.
3. Temperature: The Ultimate Vibe Setter
This one is pretty intuitive, isn't it? Think about butter. At room temperature, it’s pretty solid. But if you warm it up, it melts and becomes all smooth and spreadable. The same principle applies to cell membranes!

Higher temperatures mean more kinetic energy. All those molecules, including the phospholipids in the membrane, are zipping around faster. This increased movement means they can slide past each other more easily, leading to increased membrane fluidity. The membrane becomes more liquid-like, more "melted."
Conversely, lower temperatures mean less kinetic energy. Everything slows down. The phospholipids don’t have as much energy to overcome their attractions to each other, and they start to pack together more tightly. This makes the membrane more rigid, or less fluid. Think of it like trying to move through cold, thick molasses – it’s tough going!
So, if you want a more fluid membrane, cranking up the heat (metaphorically speaking, of course!) is definitely the way to go. It's like turning up the music at a party – everyone starts moving more!
4. Chain Length of Fatty Acids: The Longer, The Stiffer?
We talked about the kinks in fatty acid tails, but the actual length of those tails also plays a role. Imagine those spaghetti strands again. If you have really long strands of spaghetti, they're going to be more likely to get tangled up and hold onto each other.
Longer fatty acid tails have more surface area for those van der Waals interactions (fancy term for weak attractions between molecules) to occur. These interactions help to hold the phospholipids together more tightly. This increased “stickiness” makes the membrane more rigid and therefore less fluid.
On the flip side, shorter fatty acid tails have less surface area for these interactions. They can’t hold onto each other as strongly, allowing for more movement and thus, increased membrane fluidity. Think of it like having shorter strands of yarn – they’re less likely to get all knotted up.
So, if we’re looking to boost fluidity, we’d want to see membranes with shorter fatty acid tails. It’s like trading in those super long, tangled phone cords for nice, short, manageable earbuds – much less hassle!
Putting It All Together: Which Would Tend To Increase Membrane Fluidity?
Alright, let's get to the juicy part! Based on our little chat, which of these factors would tend to make your cell membranes do the funky chicken and become more fluid?
Here’s the grand finale, the answer you’ve been waiting for! If you want to crank up the fluidity dial, you're looking for:
- More unsaturated fatty acids: Those beautiful kinks are your best friends for creating space and encouraging movement.
- Higher temperatures: More energy means more wiggles and jiggles!
- Shorter fatty acid chains: Less surface area for interactions means less rigidity.
- Cholesterol at lower temperatures: Cholesterol's a bit of a wildcard, but at chilly temps, it helps keep things moving.
Conversely, if something makes the membrane more rigid, that would be:
- More saturated fatty acids: Straight tails pack in tight, making for a stiff party.
- Lower temperatures: Everything slows down, and the membrane gets a bit frozen.
- Longer fatty acid chains: More opportunities for those intermolecular attractions to lock things down.
- Cholesterol at higher temperatures: It acts as a stabilizer, preventing excessive fluidity.
The Big Picture: Why Does This Even Matter?
You might be thinking, "Okay, so my cell membrane can be a little more or a little less fluid. So what?" Well, my friends, this fluidity is absolutely critical for life as we know it!
A fluid membrane is essential for all sorts of cellular processes. Think about transport. Things need to be able to move in and out of the cell, and a fluid membrane makes this much easier. It's like having a well-oiled door that opens and closes smoothly.
Then there’s cell signaling. Your cells are constantly communicating with each other, and many of the receptors involved in this communication are embedded within the membrane. If the membrane is too stiff, these receptors might not be able to function properly, and the whole communication network could break down. It’s like trying to have a phone conversation when the phone line is all tangled and bent!
Even things like cell division and movement rely on membrane fluidity. The membrane needs to be able to change shape and stretch, and that requires a certain degree of flexibility.
So, the next time you think about your cells, remember the incredible dance party happening within their membranes. It’s a delicate balance, constantly adjusting to keep everything running smoothly. And that, my friends, is pretty darn amazing!
Remember, life is all about finding that perfect balance – not too rigid, not too loose, but just right. And just like your cell membranes, you have the incredible ability to adapt and flow. So go out there, embrace the wiggles, and let your own unique brand of fluidity shine! Keep those membranes groovy, and keep living your best, most vibrant life! You’ve got this! ✨
