Which Of The Following Statements Is True Regarding Protein Structure

So, I was having this epic battle with a particularly stubborn jar of pickles the other day. You know the one. It's like it's personally offended by the idea of being opened. I was twisting, I was banging, I was practically doing the Macarena on it (don't ask). My biceps were screaming, my knuckles were turning white, and that jar just sat there, smugly sealed. Then, my roommate, bless her speedy soul, walks in, gives it a quick, almost casual twist, and pop! Pickles for everyone. I swear, it was like watching a superhero flex their superpowers.
And it got me thinking. How did she do that? Was it a secret ninja move? A special pickle-opening incantation? Nope. It was all about the shape. She just happened to grab it at the perfect angle, applying just the right amount of pressure in the right spot. And that, my friends, is surprisingly similar to how we talk about… well, pretty much everything in science. Especially when it comes to the tiny, intricate world of protein structure.
You see, proteins are the workhorses of our bodies, and frankly, the universe. They build muscles, they fight off infections, they help digest that questionable burrito you had last night. But they can't do any of that if they're just a jumbled mess of amino acids. They need to fold up, to contort themselves into these incredibly specific, three-dimensional shapes. It’s like they have to achieve their own personal pickle jar opening technique to be useful. Pretty wild, right?
This whole idea of protein structure is a HUGE topic in biology and biochemistry. You can literally get entire degrees dedicated to it. And if you've ever stumbled upon a question like "Which of the following statements is true regarding protein structure?" you might feel a little like I did with that pickle jar – a bit lost and overwhelmed. But don't worry, we're going to break it down. Think of this as your friendly, non-intimidating guide to the fascinating world of protein folding. No intimidating jargon, just good old-fashioned curiosity.
The Folding Frenzy: It's Not Just About the Sequence!
Okay, so let’s start with the absolute basics. Proteins are made up of long chains of smaller units called amino acids. Think of them like beads on a string. The sequence of these amino acids – which bead comes after which – is super important. It's dictated by our DNA, after all. It’s like the blueprint for the protein.
But here’s the kicker: that sequence alone doesn't tell the whole story. It’s the folding of that chain into a complex 3D shape that actually gives the protein its function. It's like having a really long piece of string. You can arrange it in a million different ways, but only one or two specific configurations will actually do anything useful. That specific, folded shape is what we call the protein's native conformation.
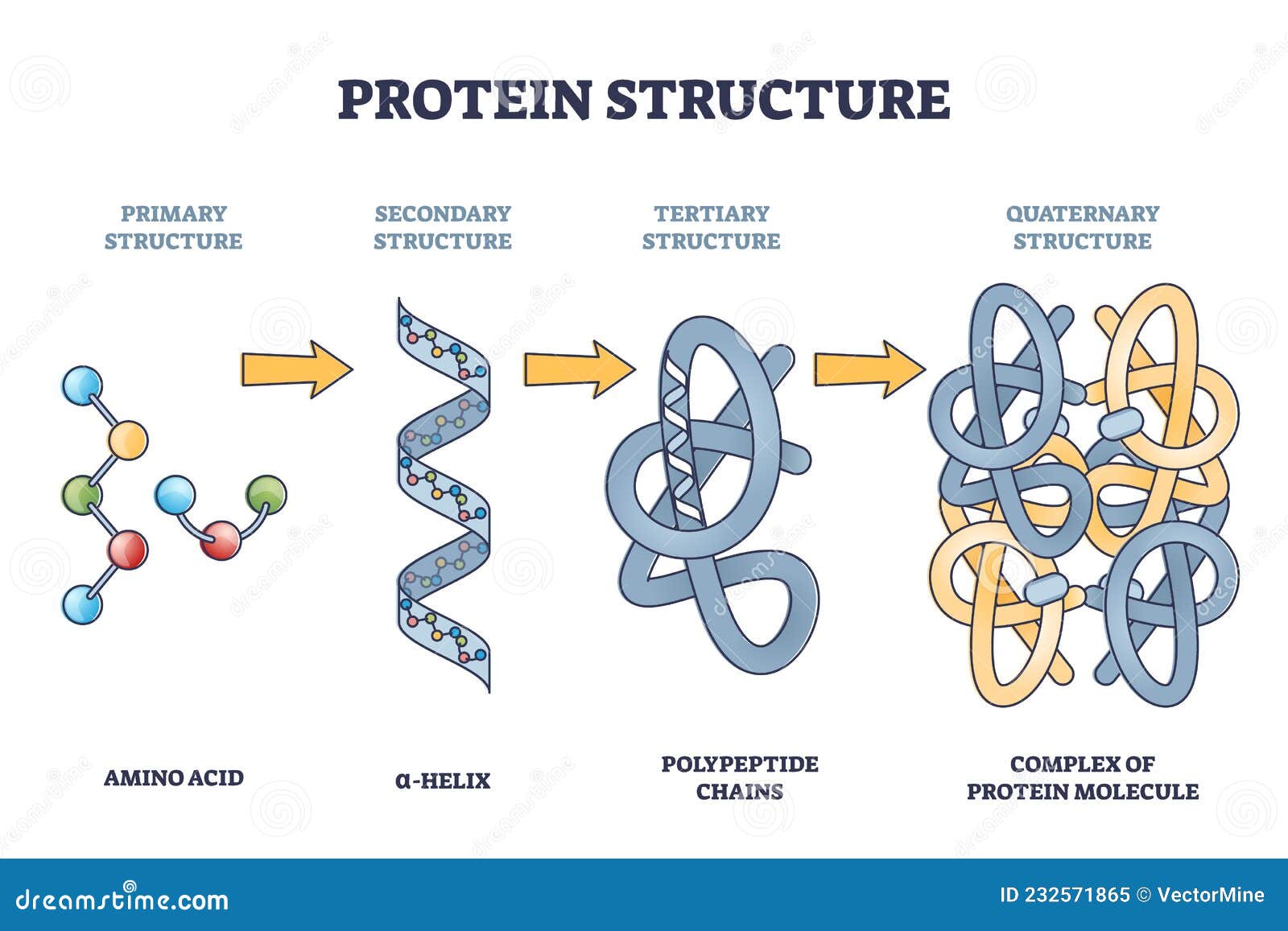
So, while the amino acid sequence is the primary structure (that’s just the order of the beads), the way those beads start to interact with each other, forming local coils and folds, is the secondary structure. Think of alpha-helices (like a spring) and beta-sheets (like a pleated skirt). These are the first hints of 3D architecture. It's like the string starting to twist and bend in specific ways.
Then comes the tertiary structure. This is where the whole chain gets its overall 3D shape. All those helices and sheets fold and pack together in a very precise, intricate way. This is like taking that pleated skirt and spring and bunching them all up into a compact, functional unit. This is where the magic really happens, where the protein develops its active sites and binding pockets. It’s the pickle jar, perfectly shaped to grip.
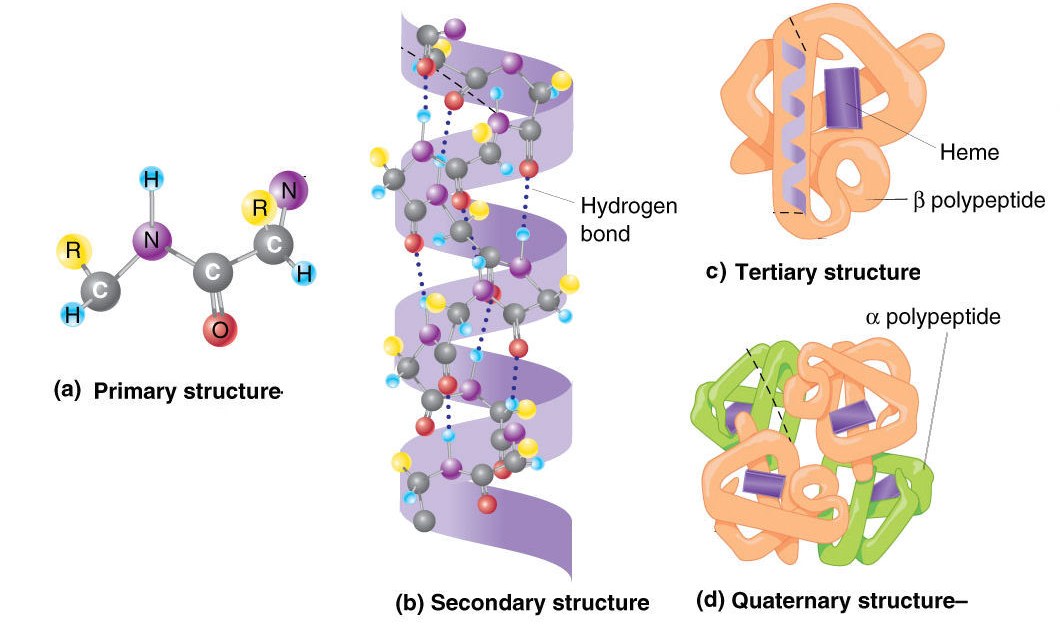
And for some proteins, there's even a quaternary structure. This happens when multiple folded protein chains (called subunits) come together to form a larger, functional complex. Think of it like multiple, perfectly shaped pickle jars coming together to form a whole crate. Hemoglobin, the protein that carries oxygen in your blood, is a classic example of a protein with quaternary structure.
Debunking Some Myths (Because Science Loves a Good Myth-Buster)
Now, let’s talk about what’s not necessarily true. This is where those tricky multiple-choice questions love to play. Here are a few things people sometimes get wrong about protein structure:
Myth 1: The sequence is the structure.
As we just discussed, this is a big NOPE. The sequence is the primary structure, the starting point. But the tertiary and quaternary structures are determined by how that sequence folds. Think of it this way: you can have two people with the same ingredients (amino acids) for a cake, but one might make a lopsided mess, and the other a perfectly baked masterpiece. The ingredients are the same, but the final form (structure) is different.
Myth 2: Proteins are always perfectly stable.
This is another one that gets people. Proteins are not rigid statues. They are dynamic molecules. They move, they wiggle, they flex. This flexibility is often crucial for their function. For instance, an enzyme might need to change its shape slightly to bind to its substrate or to catalyze a reaction. It’s more like a highly skilled dancer than a brick wall.
However, there’s a delicate balance. Too much movement, or movement in the wrong places, and the protein can lose its function. And sometimes, proteins can fold incorrectly. This is where things get a little bit scary, as misfolded proteins can clump together and cause diseases like Alzheimer's or Parkinson's. It’s like a dancer tripping over their own feet and falling down, but on a cellular level, and with much more serious consequences.

Myth 3: All proteins have the same basic shape.
Absolutely not! This is like saying all tools have the same shape. A hammer isn't shaped like a screwdriver, and a wrench isn't shaped like a saw. Each protein has evolved a unique 3D structure that is perfectly suited to its specific job. Some are globular (ball-shaped), others are fibrous (long and thin), and some are a bit of everything. It’s a magnificent diversity, reflecting the incredible diversity of biological functions.
Myth 4: Denaturation is reversible.
This one is a bit more nuanced. Denaturation is the process where a protein loses its native structure, often due to heat, extreme pH, or chemicals. Think of cooking an egg. The clear, gooey albumin protein turns white and solid. It’s denatured. While sometimes, under very specific and mild conditions, a protein can refold (this is called renaturation), often, once denatured, especially by heat or harsh chemicals, the unfolding is irreversible. The damage is done, and the protein is, for all intents and purposes, useless.
It’s like trying to un-cook that egg. You can't get that clear goo back. The chemical bonds that held the structure together have been broken, and it's not a simple matter of just putting them back in the right order. So, while we can sometimes coax a protein back into shape, it's not a guaranteed or easy process.
So, What IS True Then? Let's Get to the Core Ideas!
Alright, enough with the myths! Let's focus on what we know is true about protein structure. This is where you can really nail those questions. Here are some fundamental truths:
Statement 1: The amino acid sequence determines the protein’s three-dimensional structure.
This is the big one, the bedrock of protein science. While the sequence doesn't instantly become the structure, it provides all the information needed for the protein to fold into its correct shape. Think of it as the instructions for folding. The amino acids have different chemical properties (some like water, some hate it, some are electrically charged), and these properties dictate how the chain will fold upon itself. This is known as the Anfinsen’s dogma, named after the scientist Christian Anfinsen who did groundbreaking work in this area.
It’s a bit like a complex origami instruction manual. The paper (amino acid sequence) has all the folds pre-determined by the creases and lines. You just need to follow the steps, and voilà, you get your crane (native protein structure).
Statement 2: Protein structure is hierarchical, with primary, secondary, tertiary, and sometimes quaternary levels.
We touched on this earlier, but it's worth repeating. This organizational principle is key. It’s not just one big fold; it's layers of complexity building upon each other. The primary sequence leads to local secondary structures, which then fold into the overall tertiary structure. And if multiple chains are involved, they assemble into the quaternary structure.
This layered approach makes the folding process more manageable and efficient for the cell. It’s like building a complex LEGO model. You don't just jam all the pieces together at once. You build smaller sections (secondary structures), then combine those sections (tertiary structure), and maybe even connect multiple completed models (quaternary structure).
Statement 3: Protein function is directly dependent on its specific three-dimensional structure.
This is the “why bother with all this folding stuff?” question. Because without the right shape, a protein simply can’t do its job. An enzyme with a misfolded active site won't be able to bind its target molecule. An antibody with a distorted binding region won't recognize the foreign invader. It’s like trying to use a key that’s been bent out of shape – it just won't fit the lock.
This is why even small changes in amino acid sequence (mutations) can sometimes have drastic effects on protein function and, consequently, on an organism's health. A single wrong “bead” in the sequence can subtly alter the folding, leading to a protein that’s just… not quite right. A bit like a slightly off-key note in a beautiful melody.

Statement 4: Proteins can undergo conformational changes, and these changes are often essential for their function.
Remember how we said proteins aren't rigid? This is that concept in action. Many proteins are designed to change their shape in response to signals or interactions. This is called conformational change. For example, ion channels in your cell membranes open and close by changing their shape to allow or block ions from passing through. Muscle proteins contract and relax by altering their configuration.
It’s this dynamic nature that makes proteins so versatile. They can adapt and respond to their environment. It's the difference between a statue and a living, breathing organism. The statue is fixed; the organism is constantly in motion, adapting. This ability to flex and shift is a hallmark of many functional proteins.
The Pickle Jar Analogy Revisited (Because Why Not?)
So, thinking back to my pickle jar ordeal, the reason my roommate was successful was that she understood the structure of the lid and its connection to the jar. She didn’t just randomly twist. She applied force in a way that exploited the threads and the seal. That’s a physical analogy, of course, but it mirrors the precision required for protein function.
Proteins are like that perfectly designed pickle jar lid. The sequence is the metal, the manufacturing process. The secondary and tertiary structures are the specific grooves and ridges that allow it to fit and seal perfectly. And the function? Well, that’s keeping those pickles fresh and delicious. If the lid (protein) is damaged or the wrong shape, the contents (cellular processes) are compromised.
Understanding protein structure is fundamental to understanding how life works. It’s a cornerstone of biology and medicine. When you encounter a question about it, remember these core truths: the sequence is the blueprint, folding creates the functional shape, and that shape dictates everything the protein does. It’s a complex, beautiful, and endlessly fascinating area of science.
So, the next time you’re struggling with a stubborn jar, or marveling at how your body performs its daily miracles, give a little nod to the humble protein and its incredible journey of folding. It’s a journey that, much like a perfectly opened pickle jar, is all about getting the shape just right.
