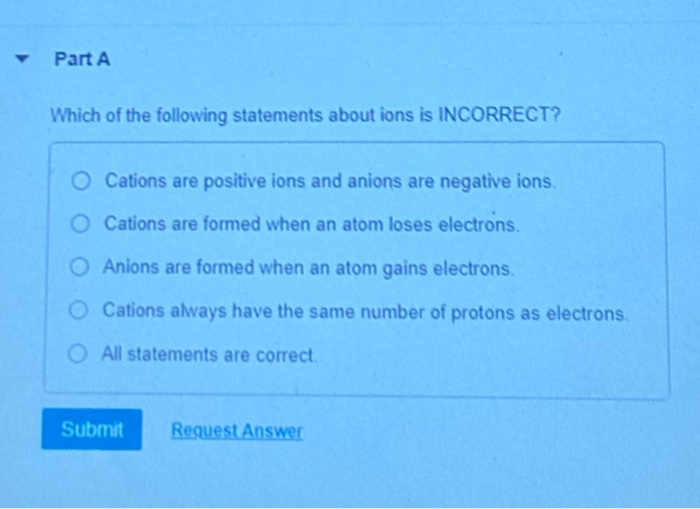
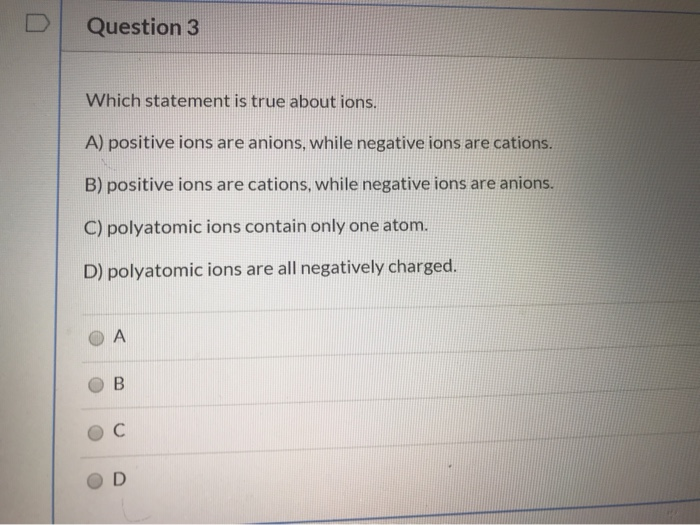
Which Of The Following Statements Is True About Ions

Imagine tiny, invisible builders constantly buzzing around, like a microscopic construction crew. These aren't just any builders, though; they’re called ions. And guess what? They’re everywhere, doing all sorts of fascinating work that keeps our world ticking.
Think of yourself. You’re made of billions and billions of these little guys. They’re not just sitting there; they’re on a mission, some positively charged, some negatively charged, all interacting like a big, energetic family reunion.
Some ions are like the life of the party, always ready to share their energy. Others are a bit more reserved, happily accepting what others offer. It’s a constant dance of give and take, a microscopic tango that’s happening in every breath you take and every bite you eat.
Let’s talk about electricity. Ever wonder how your phone magically powers up or how a lightbulb brightens your room? Yup, you guessed it: ions are the superstars behind the scenes. They’re the tiny couriers, zipping around and carrying electrical charges.
It’s like a tiny parade of charged particles, marching in organized lines to deliver messages and power. Without them, our modern lives would be a lot dimmer, both literally and figuratively. They’re the unsung heroes of our plugged-in world.
But ions aren't just about gadgets and gizmos. They’re also vital for your body to function. Your nerves, for instance, communicate using electrical signals, and who do you think is doing the heavy lifting there? You got it – our ever-present ions.
They’re like tiny messengers, sending signals from your brain to your toes, telling them to move, feel, and react. It’s a complex system, but at its core, it’s driven by these charged particles doing their essential jobs.
Have you ever had a juicy piece of fruit? Or maybe a salty pretzel? The taste you experience is often thanks to ions. They’re the flavor enhancers, interacting with your taste buds to create those delicious sensations.
Think of sodium and potassium ions. They’re like little flavor wizards, making things taste sweet, sour, or even wonderfully salty. So, the next time you savor a meal, remember the tiny, invisible chefs at work.
Water, the elixir of life, is also a stage for ionic activity. When salt dissolves in water, it breaks apart into ions. These charged little guys then spread out, making the water a better conductor of electricity.
It’s like adding glitter to water; it disperses and changes how the light (or in this case, electricity) travels through it. This seemingly simple act has huge implications for everything from weather patterns to the health of our oceans.
Interestingly, some ions are positively charged, meaning they’ve lost an electron, like a generous donor. Others are negatively charged, having gained an electron, acting like grateful recipients. This attraction between opposites is what drives many chemical reactions.
It’s a fundamental rule of the universe, mirrored in everyday life. Positive vibes attract negative vibes, and in the world of ions, this attraction creates the bonds that hold everything together.
When we talk about atoms, they're typically neutral, like perfectly balanced scales. But when an atom decides to gain or lose an electron, poof! It transforms into an ion, now with a definite charge and a whole new personality.

This transformation is key to chemistry. It’s like an atom deciding to wear a different hat, and suddenly, it interacts with the world in entirely new ways. These charged atoms are the building blocks of so many substances we encounter.
Consider the humble battery. It's essentially a controlled environment where ions are encouraged to move from one side to another. This flow of charged particles is what generates the electrical current we use every day.
It’s a miniature, self-contained power plant, all thanks to the controlled migration of ions. They're the tiny engines that make our portable devices roar to life.
Even in the ground beneath our feet, ions are hard at work. They play a crucial role in soil chemistry, influencing how plants absorb nutrients. Think of them as the soil’s personal trainers, helping the plants get their essential vitamins.
They unlock nutrients that would otherwise be stuck, making them available for plants to grow strong and healthy. So, the food on your plate often owes its existence to these hardworking soil ions.
Sometimes, ions can exist on their own, floating freely in solutions. Other times, they link up with other ions, forming compounds. It's like they're either enjoying some solo time or forming friendships and partnerships.
These partnerships create new materials with different properties. Salt, for example, is a compound formed from sodium and chloride ions. They come together to make something entirely new and essential for life.
The concept of ions might seem complex, but at its heart, it’s about charged particles. These charged particles are the unsung heroes of chemistry, biology, and physics, quietly influencing our world in countless ways.
They're the tiny magnets that pull things together, the electrical couriers, and the flavor creators. Without them, our world would be a very different, and much less interesting, place.
So, the next time you hear about ions, don’t think of abstract scientific terms. Think of them as tiny, energetic workers, participating in a grand, ongoing cosmic dance that makes everything possible.
They're the sparks that light up our lives, the carriers of information, and the silent architects of our physical reality. They're truly remarkable, these invisible entities that shape our world.
Remember that a key characteristic of ions is their electrical charge. This charge is what makes them so reactive and useful in so many different processes, from powering your phone to making your muscles contract.

This electrical charge is the defining feature. It’s what sets them apart from neutral atoms and allows them to participate in the fascinating world of chemical and biological interactions that we rely on.
Consider the notion that some statements about ions might be incorrect. It’s easy to get confused with all the talk of charges and electrons. But there’s a fundamental truth that holds steady.
Understanding that they possess these charges is like having the master key to unlock their behavior and their importance. It’s the most fundamental aspect of what makes them so special and so integral to our existence.
Essentially, ions are atoms or molecules that have gained or lost electrons. This action directly results in them having a net electrical charge. This is the core concept that underpins their entire existence and function.
They are not just plain old atoms anymore; they are atoms that have taken on a new electrical identity. This change is what allows them to interact and bond in ways that neutral atoms cannot, leading to the formation of the diverse substances around us.
So, when you’re asked about ions, remember this simple, powerful truth: they are charged entities. This fundamental property is the bedrock of all their amazing abilities and the reason they are so indispensable to life as we know it.
