Which Of The Following Statements Correctly Describes Osmosis

Okay, gather 'round, you lovely caffeine-fueled souls! Let's talk science. But before your eyes glaze over like a forgotten donut, I promise this isn't going to be your high school biology teacher droning on about cell walls. Nope. We're diving into the glorious, slightly bizarre world of osmosis. Think of it as nature's most dramatic water cooler gossip session. Seriously.
So, the big question, the one that keeps scientists up at night (or at least fueled by copious amounts of coffee): Which of the following statements correctly describes osmosis? I'm going to break it down, with a few detours into the absurd, because that's how we roll at this café, right? Grab your latte, settle in.
The Great Water Migration: What's the Deal?
First things first, what is osmosis? Imagine you've got a bunch of tiny, super-picky bouncers at a club door. These bouncers are what we call a semipermeable membrane. They let some things in (mostly water, bless its simple heart), but they're super strict about others. Think of them as the bouncers at a VIP party where only water molecules are on the guest list, and even then, they're only allowed in if things are a bit unbalanced.
Now, imagine you have two rooms separated by this bouncer-filled wall. In one room, you've got a chill party with just a few water molecules hanging out. In the other room, it's a full-blown rave – tons of water molecules, plus some other stuff dissolved in there, like salt or sugar. These dissolved things are called solutes. They're basically the party animals that the membrane bouncers don't want anywhere near the VIP water-only section.
So, what happens? The water molecules, being the social butterflies they are, want to even things out. They see the packed party room and think, "Whoa, too crowded in there! And hey, the chill room looks a little lonely." So, they start migrating. But here's the kicker: they don't just wander aimlessly. They move from the area where they are in high concentration (the chill room, where there's more water relative to solutes) to the area where they are in low concentration (the rave room, where there's less water relative to solutes because all the solutes are hogging the space).
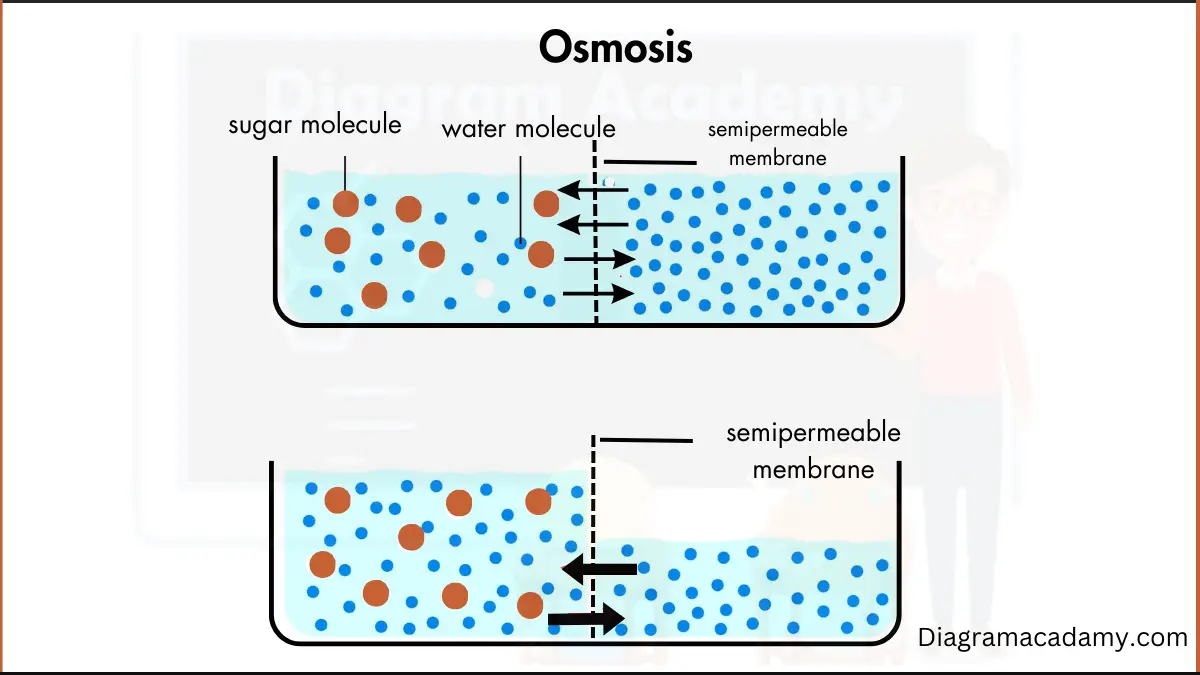
It's all about balance, people! The water is trying to dilute the party in the rave room so it's not so ridiculously crowded with solutes. It's like the ultimate cosmic game of "redistribute the snacks."
Debunking the Myths: What Osmosis Isn't
Now, let's clear up some common misconceptions, because the internet is a wild place, and sometimes osmosis gets a bad rap.
Myth 1: Osmosis is when solutes move. Nope! Remember those picky bouncer membranes? They're usually pretty good at keeping those solute party animals out. So, it's the water that moves, not the dissolved stuff. Think of it like this: the salt in your pretzel can't escape into your bloodstream easily because your cell membranes are, in a way, semipermeable. Your body's got its own osmosis game going on to keep things in check!
Myth 2: Osmosis is just diffusion. Close, but no cigar! Diffusion is like when your perfume wafts across the room – the molecules spread out from high concentration to low concentration. Osmosis is a specific type of diffusion, but it only involves the movement of water across a semipermeable membrane. So, while all osmosis is diffusion, not all diffusion is osmosis. It's like saying all squares are rectangles, but not all rectangles are squares. Mind. Blown.
Myth 3: Osmosis is always a slow, boring process. Okay, this one isn't strictly false, but it's surprisingly fast and impactful! Ever plucked a raisin and dropped it in water? Poof! Within a few hours, it's a plump little grape again. That's osmosis at work, nature's tiny hydration specialists bringing shriveled things back to life. Or consider when you've eaten way too much salty popcorn – you get thirsty, right? That's your body trying to balance out the salt concentration by drawing water into your bloodstream. Your cells are basically sending out a distress signal: "We're getting dehydrated here!"

The Verdict: Putting it All Together
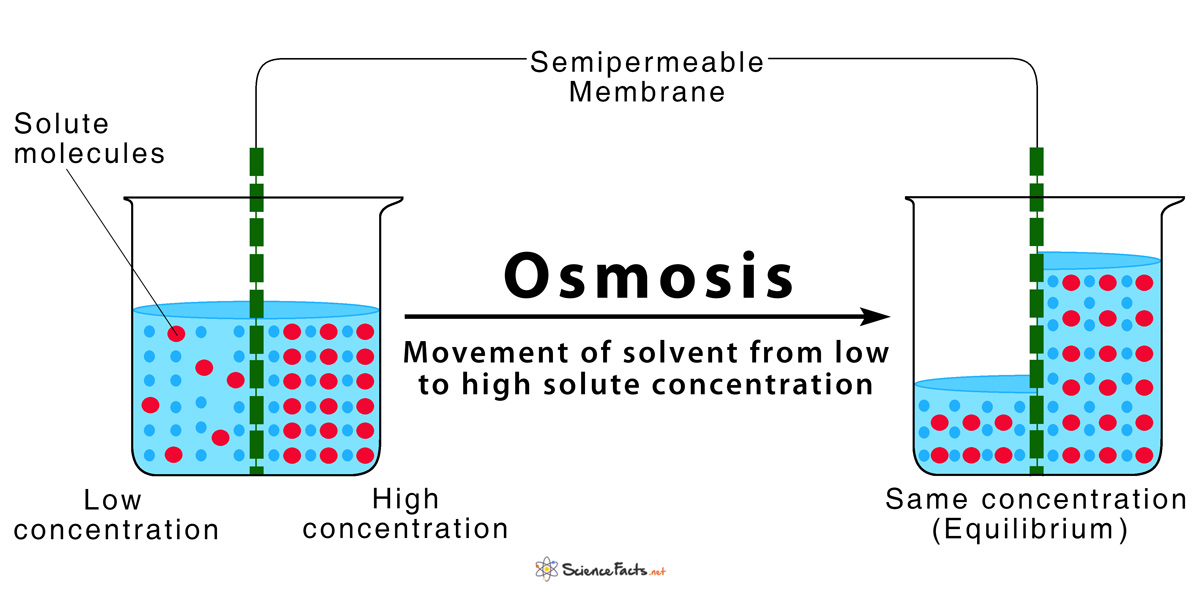
So, let's circle back to our main event. Which statement correctly describes osmosis? It’s the one that talks about the movement of water molecules across a semipermeable membrane from an area of higher water concentration to an area of lower water concentration. Or, to put it in simpler terms:
Water moves from where it's more abundant (relative to solutes) to where it's less abundant (relative to solutes), passing through a barrier that's selective about what else can go through.
It's a fundamental process that keeps plants alive (their roots soak up water!), helps our kidneys filter waste, and generally makes life on Earth possible. Pretty cool for something that sounds like a fancy word for "water wiggling."
A Surprising Splash: Unveiling the Unexpected
And here's a fun fact that might make you spill your coffee: Osmosis is so powerful, it can generate significant pressure! This is called osmotic pressure. Imagine trying to cram a ton of water into a space that's already pretty full because of the solutes. The water pushes back, and that push can be surprisingly strong. It's like a tiny, molecular tug-of-war where water is determined to win!
Think about a pickle. A cucumber starts off soft and squishy. When you soak it in brine (salty water), the water leaves the cucumber cells (because the salt concentration outside is higher) and makes it firm and puckery. That's osmosis at work, making your favorite crunchy snack! Who knew a pickle was a masterclass in fluid dynamics?
So, next time you see a wilted plant perk up after a watering, or enjoy a salty snack, give a little nod to osmosis. It's the silent, often invisible, but always crucial force that keeps our world hydrated and in balance. And honestly, that's way more exciting than just saying "water moved." Now, who wants another croissant?
