Which Of The Following Statements Concerning Osmosis Is False

Alright, settle in, grab your cuppa, and let's dive into something that sounds way more intimidating than it is: osmosis. You know, the stuff that makes raisins wrinkly and explains why your salad wilts faster than a politician's promise on election night? Yeah, that. We're going to play a little game today, a "spot the fib" challenge, about statements concerning this aquatic marvel. Think of me as your friendly neighborhood science storyteller, armed with a teacup and a healthy dose of silliness.
So, we've got a few statements about osmosis, and one of them is a colossal, honking fib. Our mission, should we choose to accept it (and we totally do, because coffee breaks are sacred), is to unmask the imposter. Imagine we're at a fancy party, and everyone's dropping these "facts" about osmosis, but one person is clearly making it all up. We're the sharp-eyed detectives of the molecular world.
The Usual Suspects: What Osmosis Actually Does
Before we get to the impostor, let's get our facts straight. Osmosis, at its heart, is about water wanting to chill. Like, it's always trying to find its happy medium. Think of it as a party guest who really hates being the center of attention. Water molecules, bless their tiny hearts, are a bit on the indecisive side. They’ll happily bobble around anywhere, but they really don't like being crammed into one place while there’s a more spacious area nearby.
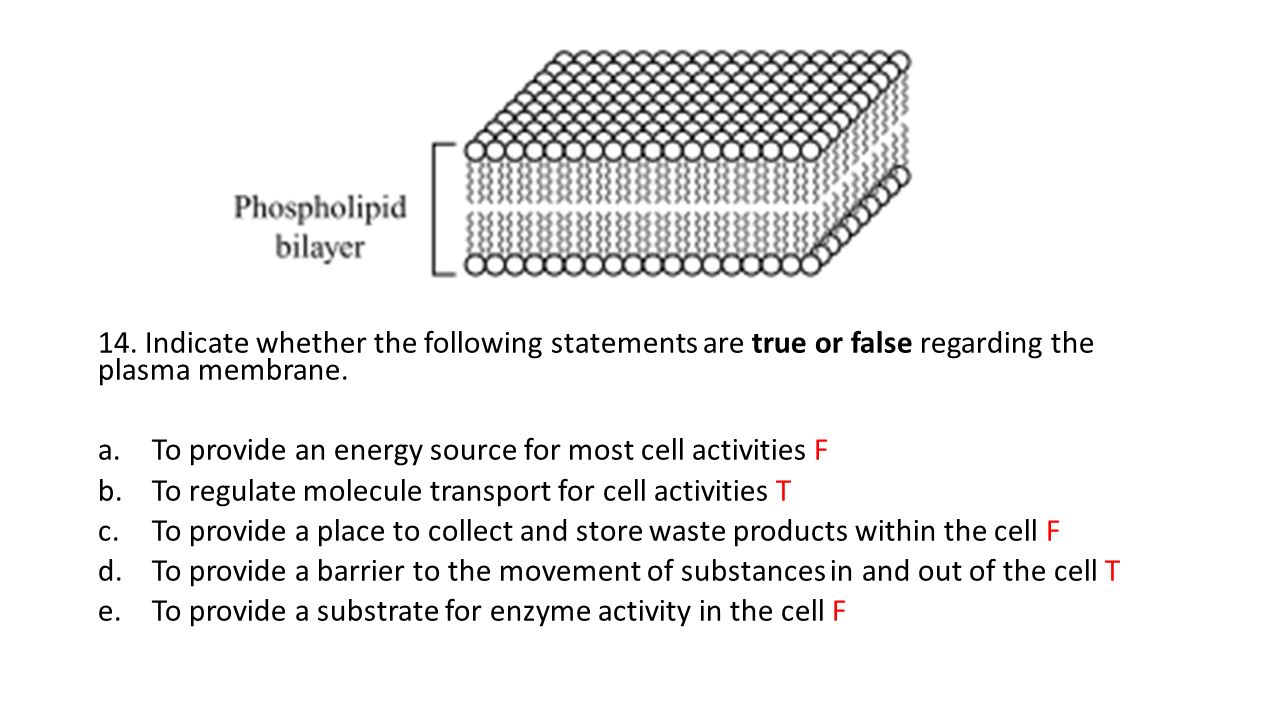
So, imagine you have a semi-permeable membrane. Fancy word, I know. Basically, it's like a bouncer at a club with a very specific guest list. It'll let small things through, like water molecules, but it'll keep bigger things out, like, say, sugar molecules or those pesky salt crystals that get everywhere. Now, if you put a solution with a high concentration of "stuff" (like salt or sugar) on one side, and pure water on the other, the water goes, "Whoa, that side looks way more crowded with other molecules! I need to go over there and dilute things down so everyone can have their personal space!"
And that, my friends, is osmosis in a nutshell. Water moves from an area of low solute concentration (lots of water, not much dissolved stuff) to an area of high solute concentration (less water, a whole lot of dissolved stuff). It's all about achieving equilibrium, that sweet spot where everything is balanced. It’s like a molecular peace treaty.
Let's Meet Our "Experts" (and One Liar)
Now, let's look at the statements. We’ve got our potential truths, and one sneaky falsehood lurking in the shadows. Get ready for some science-y pronouncements, some of which might sound plausible, but only one is a complete load of… well, you know.
Statement 1: "Osmosis is the movement of water across a semi-permeable membrane from an area of higher water concentration to an area of lower water concentration."
This one sounds pretty legit, right? It’s basically our definition. High water concentration means there's less stuff dissolved in it, so water has plenty of room to move. Low water concentration means there's a ton of dissolved stuff, making it a more crowded party for the water. So, water heads towards the party. This statement is a TRUE statement. No fib here!
Statement 2: "In a hypertonic solution, cells will lose water and shrink."
Okay, let’s break down "hypertonic." Think of it as "hyper" – over-the-top, extra stuff. So, a hypertonic solution is one with a really high concentration of solutes. Imagine putting a cell into a super-salty bath. The bath is hypertonic. Inside the cell, there’s less salt. So, following the osmosis rule, water will say, "Phew, this bath is crazy salty! I gotta get out of this cell and try to dilute that madness!" And poof! The cell loses water and shrinks. This is what happens to your fingertips after a long soak in the tub, by the way. They get pruney because the water in your skin moves out into the less salty (relatively speaking) bathwater. So, this statement is also TRUE.
Statement 3: "Osmosis requires energy input to occur."
Now, this one… this one tickles my suspicion. Think about our party analogy. Does the water need a personal invitation and a taxi to get to the more crowded side? No! It’s just going where the molecular vibe is right. Osmosis is a passive process. It doesn't require the cell to expend any energy, like a little molecular self-starter. It just… happens. It's like gravity; you don't have to try to fall down. If you trip, gravity’s got you. So, if this statement claims it needs energy, it’s probably our fibber. Let's hold onto this one. It’s smelling fishy, and not in a good, omega-3 way.
Statement 4: "Plant cells, when placed in a hypotonic solution, become turgid due to the influx of water."
Let's tackle "hypotonic" first. This is the opposite of hypertonic. Think "hypo" – like "hip-hop" needing less energy? No, that’s a terrible analogy. Think "hypo" like "hypodermic needle" – it goes under. So, hypotonic means less solute concentration. If you put a plant cell in pure water, the water outside the cell has a lower concentration of solutes than inside the cell. So, water rushes into the plant cell. Now, plant cells have a sturdy cell wall. This wall prevents the cell from bursting. Instead, the influx of water pushes the cell membrane against the cell wall, making the cell firm and stiff. This is called turgor pressure, and it's what keeps your salad crisp and your houseplants from looking like sad, droopy noodles. So, this statement is also TRUE.
Statement 5: "Active transport is the primary mechanism by which water moves across cell membranes during osmosis."
Wait a minute! We just established that osmosis is a passive process. Active transport, on the other hand, is like the VIP section of the molecular club. It requires energy (usually in the form of ATP) to move substances against their concentration gradient. Osmosis is all about water following its gradient, no energy ticket required. So, if this statement says active transport is the primary mechanism for osmosis, it's like saying a bicycle uses jet fuel to get around. It's fundamentally misunderstanding the engine! This statement is also shouting "LIAR!" along with Statement 3. Let's re-examine Statement 3.

Let's go back to Statement 3: "Osmosis requires energy input to occur." And Statement 5: "Active transport is the primary mechanism by which water moves across cell membranes during osmosis."
Ah, here's the crucial distinction! Statement 3 says osmosis itself requires energy. Statement 5 says active transport is the mechanism. Osmosis is the phenomenon of water movement. Active transport is a process that uses energy. While water can also move via aquaporins (special water channels), the fundamental drive of osmosis is passive. However, some might argue that the overall cell function that facilitates osmosis can involve active processes indirectly. But the core movement of water due to concentration gradients in osmosis is passive.
Let's look closely at Statement 3 again: "Osmosis requires energy input to occur." This is definitively false. The driving force is the concentration gradient, not an expenditure of cellular energy. It's a natural tendency.
Now, Statement 5: "Active transport is the primary mechanism by which water moves across cell membranes during osmosis." This is also false because, as we said, osmosis is passive movement. However, the question asks for *which of the following statements concerning osmosis is false. If there's only one, we need to be super precise.
Let's re-evaluate. Statement 3 states that osmosis *requires energy input. This is a direct contradiction of its passive nature. Statement 5 incorrectly identifies the mechanism of water movement during osmosis. While osmosis itself is passive, water can be moved by active transport in other contexts, and aquaporins are channels, not active transporters in the ATP-burning sense. But the primary mechanism for osmosis is not active transport.

Here's the trick: the question implies one false statement. Let's consider which statement is the most fundamentally incorrect about the core concept of osmosis itself.
Statement 3 is an absolute declaration that osmosis requires energy input. This is a direct lie about the process. Osmosis is defined by its lack of energy requirement for the water movement itself.
Statement 5 is also false because active transport is energy-dependent and moves things against gradients, whereas osmosis is water moving down a gradient passively. However, the phrasing "mechanism by which water moves across cell membranes during osmosis" is a bit more nuanced. While active transport isn't the driving force of osmosis, water movement across membranes is the subject. But again, osmosis itself is passive.
The most blatant, undeniable, "you are wrong, sir/madam" statement is number 3. Osmosis is a passive process. It doesn't need cellular energy to happen. It's the ultimate laid-back molecular migration.
So, the false statement is: "Osmosis requires energy input to occur." This is the one that's just plain wrong. It’s like saying sunshine needs a flashlight to be bright. Nope. It’s a natural phenomenon, a beautiful dance of water molecules seeking their equilibrium. Case closed!
