Which Of The Following Statements Are True About Oxidative Phosphorylation

You know, I was recently tidying up my kitchen, a task that, let's be honest, often feels like wrestling an octopus in a phone booth. Amidst the overflowing spice rack and the mystery Tupperware, I stumbled upon a dusty old textbook. It was a biology behemoth from my college days, and flipping through it felt like a strange trip down memory lane. I landed on a chapter about cellular respiration, and my eyes immediately gravitated towards a particularly dense diagram labeled "Oxidative Phosphorylation."
Suddenly, I remembered the sheer ordeal of trying to wrap my head around this process. It felt like being dropped into a foreign country with no map and only a phrasebook of incredibly complicated words. But hey, if my past self could eventually make some sense of it (with the help of copious amounts of coffee, I'm sure), then maybe, just maybe, we can tackle it together now without needing a degree in biochemistry. Think of this as a friendly chat, not a pop quiz!
So, let's dive into this fascinating, and admittedly a bit intimidating, world of oxidative phosphorylation. It's basically how our cells, and the cells of most living things, generate the vast majority of their energy. You know that feeling when you’ve just had a really satisfying meal and you’re buzzing with energy? Well, a good chunk of that is thanks to this very process working away in your mitochondria. Pretty cool, right?
Now, the thing about understanding complex biological processes is that they often involve a series of interconnected steps. Oxidative phosphorylation is no exception. It’s not just one single event; it’s a whole cascade, a well-choreographed dance of molecules. And like any good dance, it has its key players and its specific moves.
We’re going to break down some statements about oxidative phosphorylation, and you and I will figure out together which ones are the real deal. No pressure, just good old-fashioned scientific detective work. Ready to put on our lab coats (metaphorically, of course)?
The Grand Finale: What IS Oxidative Phosphorylation, Anyway?
Before we get into the true/false statements, let's get a quick refresher on the main event. Oxidative phosphorylation is, at its core, the process where the energy released from the oxidation of nutrients is used to generate ATP. ATP, or adenosine triphosphate, is the energy currency of the cell. Think of it as the cash your cells use to pay for everything they need to do – muscle contraction, nerve impulses, building new molecules, you name it. Without ATP, life as we know it would grind to a halt. Sad trombone.
This process happens primarily in the mitochondria, those little powerhouses of the cell. It's a two-part show: the electron transport chain (ETC) and chemiosmosis. They're like the opening act and the headliner, each crucial for the overall success.
The electron transport chain involves a series of protein complexes embedded in the inner mitochondrial membrane. These complexes act like a series of buckets, passing high-energy electrons from one to another. As the electrons move, they release energy. This is where the "oxidative" part comes in – it's a series of oxidation-reduction reactions.
The energy released from these electron transfers is used to pump protons (H+) from the mitochondrial matrix to the intermembrane space. This creates a concentration gradient, a buildup of protons on one side of the membrane. Imagine filling a dam with water; the potential energy is stored.
Then comes the "phosphorylation" part, driven by chemiosmosis. The protons, wanting to move back down their concentration gradient to the matrix, flow through a special enzyme called ATP synthase. This enzyme is like a tiny turbine. As the protons rush through it, it spins, and this spinning motion provides the energy to attach a phosphate group to ADP (adenosine diphosphate), creating ATP.
It's a remarkably efficient process, generating far more ATP than glycolysis (the initial breakdown of glucose) or the Krebs cycle alone. So, when you're running a marathon or just trying to remember where you put your keys, your mitochondria are working overtime thanks to oxidative phosphorylation.

Let's Play: True or False?
Alright, enough preamble. Let's get to the juicy part. I've got a few statements here that are floating around about oxidative phosphorylation. Your job, should you choose to accept it, is to help me discern the truth from the… well, let's just say the less-than-accurate. Think of it as a mental workout, and who doesn't love a good mental workout?
Statement 1: Oxidative phosphorylation directly uses oxygen as a substrate to create ATP.
Hmm, this is an interesting one. Oxygen is definitely involved, right? We breathe it in, and it's absolutely essential for this whole shebang to work. But is it directly used in the ATP creation itself? Let’s ponder this. Think back to the electron transport chain. What’s the ultimate fate of those electrons? They need a final acceptor. And guess who that is?
Yes, it’s oxygen! Oxygen is the final electron acceptor in the electron transport chain. It combines with electrons and protons to form water (H₂O). This step is crucial because if there wasn't a final acceptor, the electrons would just keep building up, and the chain would grind to a halt. So, while oxygen isn't directly used to make the ATP molecule itself (like attaching a phosphate group), it's absolutely essential for the process that leads to ATP production. Without its role as the final acceptor, the entire ATP-generating machinery would seize up.
Therefore, I'd lean towards this statement being TRUE, albeit with a slight nuance. Oxygen's role is fundamental to enabling ATP synthesis by accepting electrons and allowing the electron transport chain to continue its work. It's not like we're popping oxygen molecules into ATP synthase, but without oxygen accepting those electrons, the proton gradient wouldn't be maintained, and ATP synthase wouldn't have the energy flow to work with.
Statement 2: The primary site of oxidative phosphorylation in eukaryotic cells is the cytoplasm.
Okay, deep breaths. Where did we say this whole ATP-generating party happens? Remember those little powerhouses we talked about? The ones with the folded inner membranes?
If you’re thinking “mitochondria,” then you’re absolutely on the right track! The mitochondrial matrix and the inner mitochondrial membrane are where all the action for oxidative phosphorylation takes place. The cytoplasm is where glycolysis happens – the initial breakdown of glucose. While glycolysis is important for getting the process started by producing pyruvate, the heavy lifting for ATP production via oxidative phosphorylation is firmly within the mitochondria.
So, this statement? Definitely FALSE. It's like saying the main stage of a rock concert is the parking lot. Important for getting there, maybe, but not where the music is actually made!
Statement 3: The electron transport chain pumps protons from the mitochondrial matrix to the intermembrane space.
Let's revisit our dam analogy. We talked about pumping protons from somewhere to somewhere else to build up that potential energy. Where were we building the "lake" behind the dam?
The electron transport chain complexes are like little proton pumps. As electrons move through them, they use the released energy to actively move protons (H+) from the mitochondrial matrix (the inner compartment of the mitochondrion) to the intermembrane space (the region between the inner and outer mitochondrial membranes). This creates that crucial proton gradient, with a higher concentration of protons in the intermembrane space.
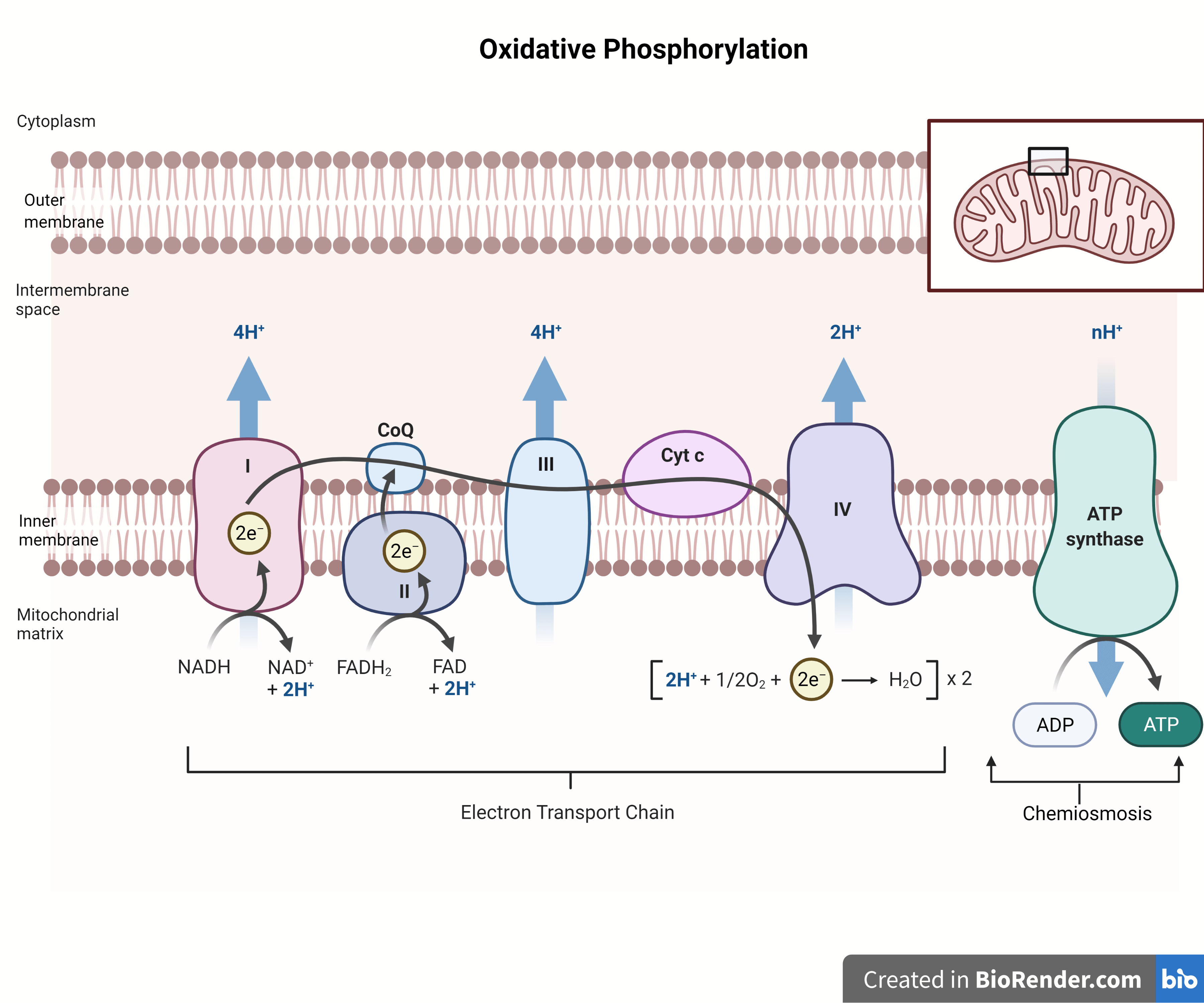
This proton gradient is the stored energy that will be used to drive ATP synthesis. So, if the statement says protons are pumped from the matrix to the intermembrane space, then that sounds spot on. It's the whole point of that part of the process!
Therefore, this statement is TRUE. It’s the engine that drives the subsequent ATP production.
Statement 4: ATP synthase directly uses the energy from breaking chemical bonds in glucose to produce ATP.
Now, let’s be careful here. We know that glucose is the starting point, and it gets broken down through a series of steps, releasing energy. But remember how we described ATP synthase? What was its direct energy source?
ATP synthase is powered by the proton motive force – the electrochemical gradient of protons across the inner mitochondrial membrane. It's the flow of these protons back into the matrix that spins the rotor of ATP synthase, allowing it to phosphorylate ADP into ATP. The energy from breaking glucose bonds is ultimately converted into the proton gradient, but ATP synthase itself doesn't directly interact with glucose molecules or the bonds within them.
It’s like saying a water wheel directly uses the energy of the falling tree that redirected the river. The falling tree was essential for getting the water there, but the water wheel’s immediate energy source is the moving water itself. Similarly, ATP synthase’s immediate energy source is the proton flow, not the original glucose bonds.
So, this statement is FALSE. It's an indirect connection, but not a direct one.
Statement 5: Fermentation and oxidative phosphorylation both produce ATP.
Ah, fermentation. The backup plan, the anaerobic hero! We've all heard of it, especially when our muscles are burning after a tough workout. Both fermentation and oxidative phosphorylation are ways cells generate ATP, but they’re quite different in their methods and efficiency.
Oxidative phosphorylation, as we’ve discussed, requires oxygen and is super efficient, churning out lots of ATP. Fermentation, on the other hand, happens in the absence of oxygen (anaerobically). It’s a less efficient process, producing only a small net gain of ATP (typically from glycolysis, which precedes fermentation). Its main job is to regenerate NAD+ so that glycolysis can continue to produce a little bit of ATP.

So, do they both produce ATP? Yes, they do. Fermentation produces ATP through glycolysis, and oxidative phosphorylation produces ATP through the electron transport chain and chemiosmosis. The amount and the mechanism are vastly different, but the outcome of ATP production is present in both.
Therefore, this statement is TRUE. It highlights that there are multiple ways cells can get their energy fix, even if one is the all-star performer.
Statement 6: The inner mitochondrial membrane is impermeable to protons.
This one’s a bit of a trick question if you’re not paying close attention to the specific way protons move. The inner mitochondrial membrane is indeed a barrier, but it’s not entirely impermeable to everything. We’ve been talking about how protons are pumped across it and then flow back.
The inner mitochondrial membrane is selectively permeable. While it effectively prevents the free diffusion of protons back into the matrix, it's not like a solid wall. The only way protons can get back into the matrix and release their potential energy is through the specialized channel provided by ATP synthase. This controlled flow is what makes chemiosmosis possible.
So, while it’s a barrier that maintains the proton gradient, it’s not completely impermeable in the sense that nothing can pass. It’s the regulated passage through ATP synthase that is key. If it were truly impermeable, the proton gradient wouldn't dissipate, and ATP synthesis would stop.
Because of the crucial role of ATP synthase in proton movement, stating it’s impermeable to protons is a bit of an oversimplification and could lead to confusion. The membrane impedes free diffusion, but it's not a total block. However, in the context of establishing and utilizing a proton gradient for ATP synthesis, the controlled flow is the operative word.
Given the strict definition of "impermeable" meaning nothing can pass, this statement is technically FALSE. The membrane is selectively permeable, and the controlled passage of protons through ATP synthase is essential.
Statement 7: NADH and FADH2 are electron carriers that donate electrons to the electron transport chain.
Okay, let's talk about the delivery drivers. Where do those high-energy electrons come from in the first place before they start their journey down the electron transport chain?
We know that cellular respiration involves breaking down fuel molecules like glucose. The processes of glycolysis and the Krebs cycle (also known as the citric acid cycle) generate reduced electron carriers. The two most important ones in this context are NADH and FADH2. These molecules are like little rechargeable batteries, carrying high-energy electrons harvested from the breakdown of food.

When NADH and FADH2 encounter the electron transport chain, they donate their electrons to the first few protein complexes in the chain. This donation of electrons is what gets the whole electron transport chain moving. So, yes, they are indeed the primary electron donors.
This statement is absolutely TRUE. They are the starting point for delivering those crucial electrons that will ultimately power ATP synthesis.
Statement 8: The entire process of oxidative phosphorylation occurs in a single step.
We’ve touched on this before, haven’t we? The idea that it’s a complex, multi-stage process.
As we discussed, oxidative phosphorylation is a coordinated sequence of events involving the electron transport chain and chemiosmosis. It's not a single magical moment where ATP pops into existence. There’s the electron transfer, the proton pumping, the establishment of the gradient, and then the ATP synthase using that gradient to make ATP. Each of these stages is vital.
So, if anyone tells you it’s a single step, I’d politely suggest they might be a little… misinformed. Or perhaps they’re just trying to simplify things a lot. But biologically speaking, it’s a journey, not a hop, skip, and a jump.
Therefore, this statement is FALSE. It’s a multi-step, highly regulated process.
Wrapping It Up
Phew! We made it through! I hope this wasn't as intimidating as that dusty textbook made it feel. Oxidative phosphorylation is a cornerstone of life, a testament to the incredible efficiency and elegance of our cellular machinery. It’s a bit like a complex engine; many parts have to work together perfectly for it to run smoothly and generate the power we need.
So, to recap our truth-tellers and myth-busters:
- Oxygen is indeed crucial as the final electron acceptor, making statement 1 TRUE.
- The cytoplasm is NOT the main site; it's all about the mitochondria, making statement 2 FALSE.
- Protons are indeed pumped from the matrix to the intermembrane space, making statement 3 TRUE.
- ATP synthase uses the proton gradient, not direct glucose bond energy, making statement 4 FALSE.
- Both fermentation and oxidative phosphorylation produce ATP, making statement 5 TRUE.
- The inner mitochondrial membrane is selectively permeable, not totally impermeable to protons, making statement 6 FALSE.
- NADH and FADH2 are the key electron donors, making statement 7 TRUE.
- Oxidative phosphorylation is a multi-step process, not a single one, making statement 8 FALSE.
It's amazing to think about all the intricate biological processes happening within us every second, without us even having to consciously think about them. Next time you feel that burst of energy, maybe give a little nod to your mitochondria and the marvelous dance of oxidative phosphorylation. Keep exploring, keep questioning, and never be afraid to dive into the wonderfully complex world of science!
