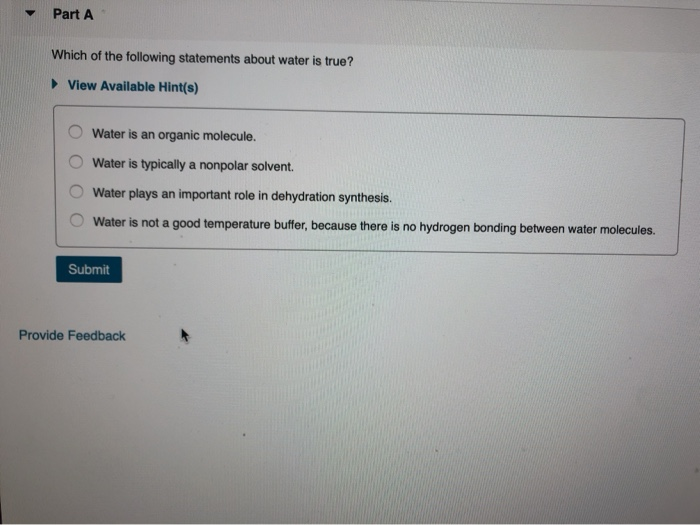
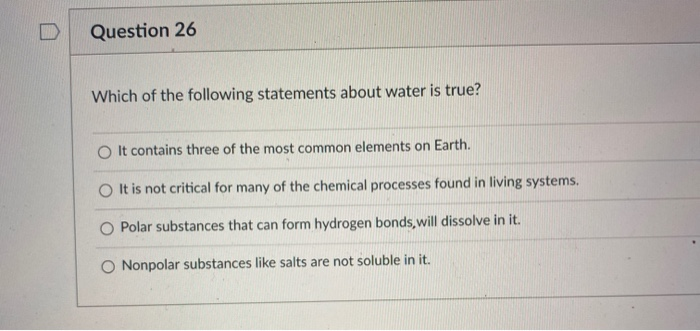
Which Of The Following Statements About Water Are True
Hey there, water lovers! Ever stopped to think about this amazing stuff that makes up, well, pretty much everything? It’s literally all around us, from the coffee you sipped this morning to the very tears you might shed watching a heartwarming movie. We’re talking about H₂O, the superstar molecule that keeps our planet humming. And guess what? It’s full of surprises, so many that trying to pick out the true statements about water can feel like a treasure hunt. But fear not, fellow adventurers, because we’re about to dive headfirst into some seriously cool facts about our favorite liquid. Get ready to have your mind… well, not blown, because water is too gentle for that, but definitely delightfully awakened!
Let’s get this party started with a classic. Imagine this: you're a tiny little water molecule. You’re chilling, just vibing with your buddies. Suddenly, BAM! You get super excited, you start wiggling and jiggling like you’ve had way too much caffeine. What happens? You turn into a gas, right? You float away, becoming steam or water vapor, ready to join the clouds for a sky-high adventure. This is the magic of evaporation! It's like water getting dressed up in its invisible superhero cape. And when it cools down, it's like it’s saying, "Okay, party's over, time to get back together!" and it forms droplets. This is how rain is born, folks! It’s the ultimate cosmic re-grouping session.
Now, picture yourself on a scorching hot day. You reach for that frosty glass of water. You take a big gulp, and oh, the relief! That refreshing coolness is your body thanking you for the good vibes. But here’s where things get a little mind-boggling: water actually expands when it freezes. Wait, what?! That’s right! Think about it. When you freeze water into ice, it takes up more space than the liquid water it came from. This is why ice floats! If ice sank, our oceans would be solid blocks of frozen misery, and where would the cheerful whales and playful dolphins hang out? Probably contemplating their frozen existence, which is a bit too grim for our fun facts, wouldn’t you agree? So, thank goodness for this peculiar property of water. It’s like the universe’s way of saying, “Let there be floating icebergs for us to admire (from a safe distance, of course)!”
This weird expansion thing isn't just for fun. It's actually super important for life on Earth!
Let's talk about what happens when water meets other stuff. You know how salt disappears when you stir it into water? That's because water is an incredibly good solvent. It’s like a social butterfly, always ready to mix and mingle. It can dissolve so many things! Sugar, for instance, does a disappearing act faster than a magician with a rabbit. Your own body is practically a giant, walking solution, thanks to water carrying all sorts of nutrients and minerals where they need to go. Imagine trying to get all those vital bits and bobs around your body without water. It would be like trying to deliver mail without roads – utterly impossible and incredibly messy!
And speaking of being all over the place, did you know that water can exist in three different states? We’ve already touched on liquid (your everyday drinking water), gas (that invisible steam from a boiling kettle), and solid (ice, your trusty ice cubes). But the way it transitions between these is pure poetry in motion. It’s a perpetual cycle of transformation, a never-ending dance of molecules. It’s like water has multiple personalities, and they’re all pretty awesome. One minute it’s a smooth, flowing river, the next it’s a wispy cloud, and then it’s a solid, crystalline sculpture of ice. Talk about versatility!
Now, here's a fun one that might surprise you. If you've ever been to the beach, you've experienced the sheer power of surface tension. That’s the thin, invisible "skin" on the surface of water that allows tiny insects, like those impossibly cute water striders, to walk on it without sinking. They’re basically tiny water surfers, catching waves that are invisible to us! It’s this same surface tension that allows you to fill a glass of water just a little bit over the brim, and the water will bulge upwards. It’s like the water is holding its breath, trying its best not to spill. Pretty neat, huh?

And let’s not forget about its incredible ability to absorb and release heat. This is a big deal for our planet. Oceans act like giant thermostats, soaking up heat during the day and releasing it at night, helping to keep temperatures more stable. Think of it like a giant, global air conditioner. Without this thermal regulation, some parts of the world would be boiling hot all the time, and others would be frozen solid. So, next time you’re enjoying a pleasant temperature, you can thank water for being such a diligent climate controller!
So, to recap our little aquatic adventure: water is a shape-shifter, a super-solvent, and a world-regulator. It expands when it freezes (how wild is that?!), can exist in three states, and has a surface tension that lets tiny creatures have their own water-walking parties. It’s involved in everything from the rain falling outside to the very processes that keep you alive. Pretty incredible, right? It’s easy to take it for granted, but water is truly one of nature’s most magnificent creations. Go ahead, pour yourself a glass. Give it a little swirl. Appreciate its simplicity, and its extraordinary power. You’re basically holding a tiny piece of magic in your hands!
