Which Of The Following Statements About Monosaccharide Structure Is True

Hey there, ever feel like life could use a little extra sweetness? Well, guess what? It already has! We're talking about the tiny building blocks of all things sweet and energy-giving: monosaccharides! Don't let the fancy science-y name scare you off; these little guys are the rockstars of the sugar world, and understanding them can actually be pretty fun. Think of it as unlocking a secret code to deliciousness and how our bodies work!
Now, I know what you're thinking. "Monosaccharides? Is this a pop quiz?" Nope! We're diving into a little bit of science, but in the most delightful way possible. Imagine you're at a fun trivia night, and a question pops up: "Which of the following statements about monosaccharide structure is true?" Sounds a bit intimidating, right? But trust me, it’s less about memorizing a textbook and more about appreciating the clever design of nature.
The Wonderful World of Monosaccharides
So, what exactly are these monosaccharides? Think of them as the single, indivisible units of carbohydrates. They're the simplest sugars, like the fundamental LEGO bricks of sweetness. You can't break them down into anything smaller when it comes to sugars. Pretty neat, huh?
They're the foundation for more complex sugars like sucrose (that's table sugar, folks!) and lactose (the sugar in milk). Without these basic building blocks, we wouldn't have the energy to dance, laugh, or even binge-watch our favorite shows. They are, in essence, nature's energy bars.
A Little Structural Detective Work
Now, for the juicy part: their structure! This is where the magic happens, and understanding it can help us nail that hypothetical trivia question. Monosaccharides are characterized by a few key features. They all have a specific number of carbon atoms in their chain, and they all possess a certain functional group that makes them, well, sweet and reactive.
Think about it like building with those LEGOs. The shape and the number of studs matter, right? For monosaccharides, the key is their carbon backbone and the presence of hydroxyl groups (-OH). These are the little arms that stick out and play a huge role in how these sugars behave.
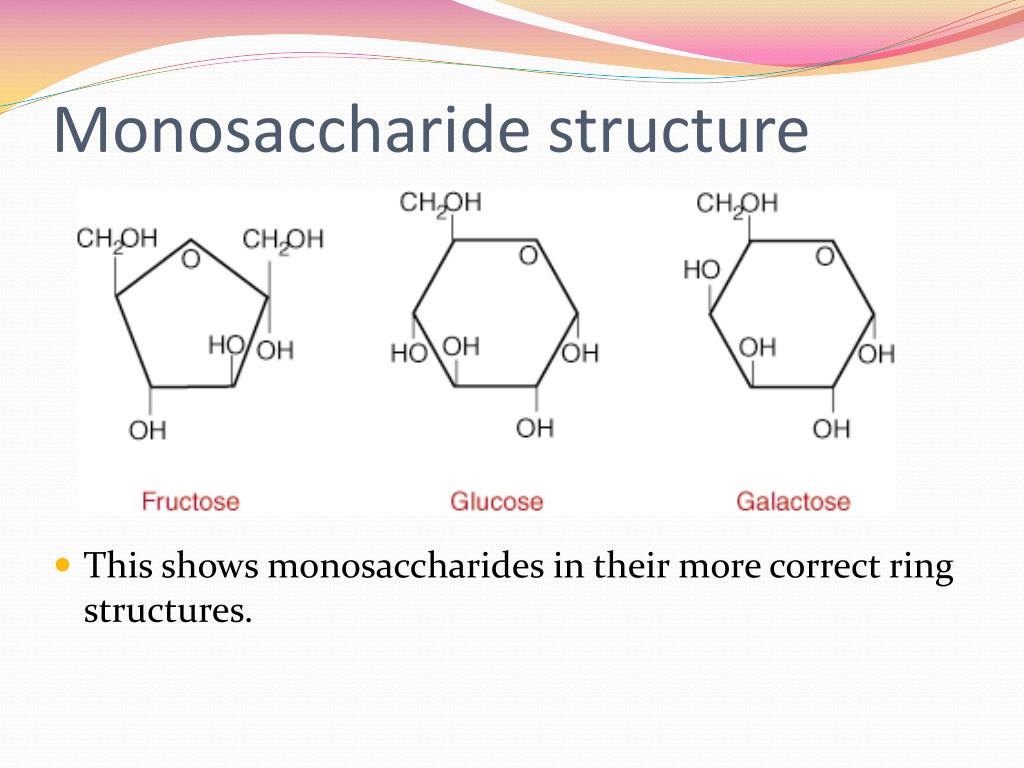
The All-Important Functional Groups
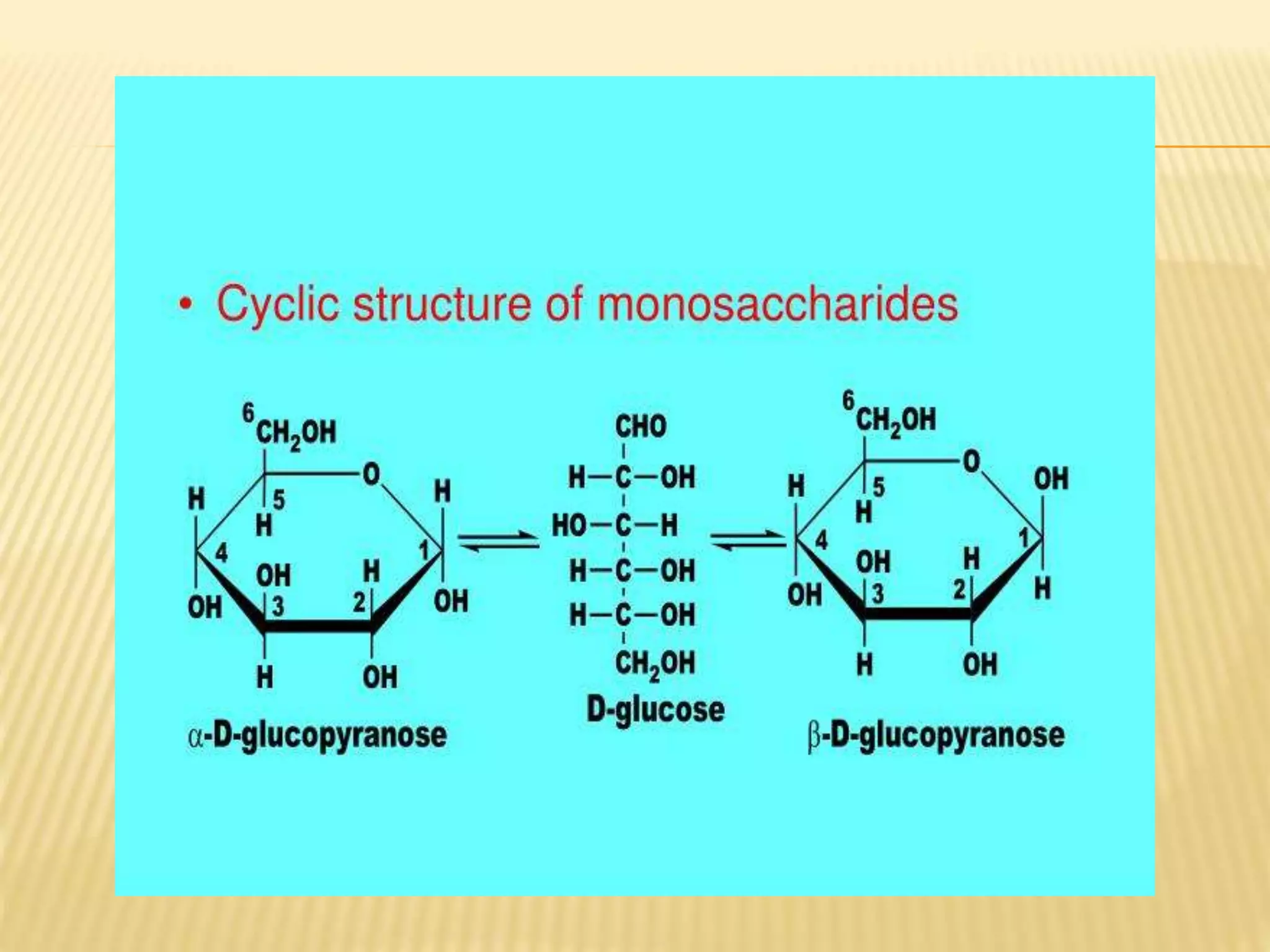
Here's where we can get a little specific, but in a good way! Monosaccharides are either aldoses or ketoses. What's the difference, you ask? It all comes down to the location of a very important functional group: the carbonyl group (C=O).
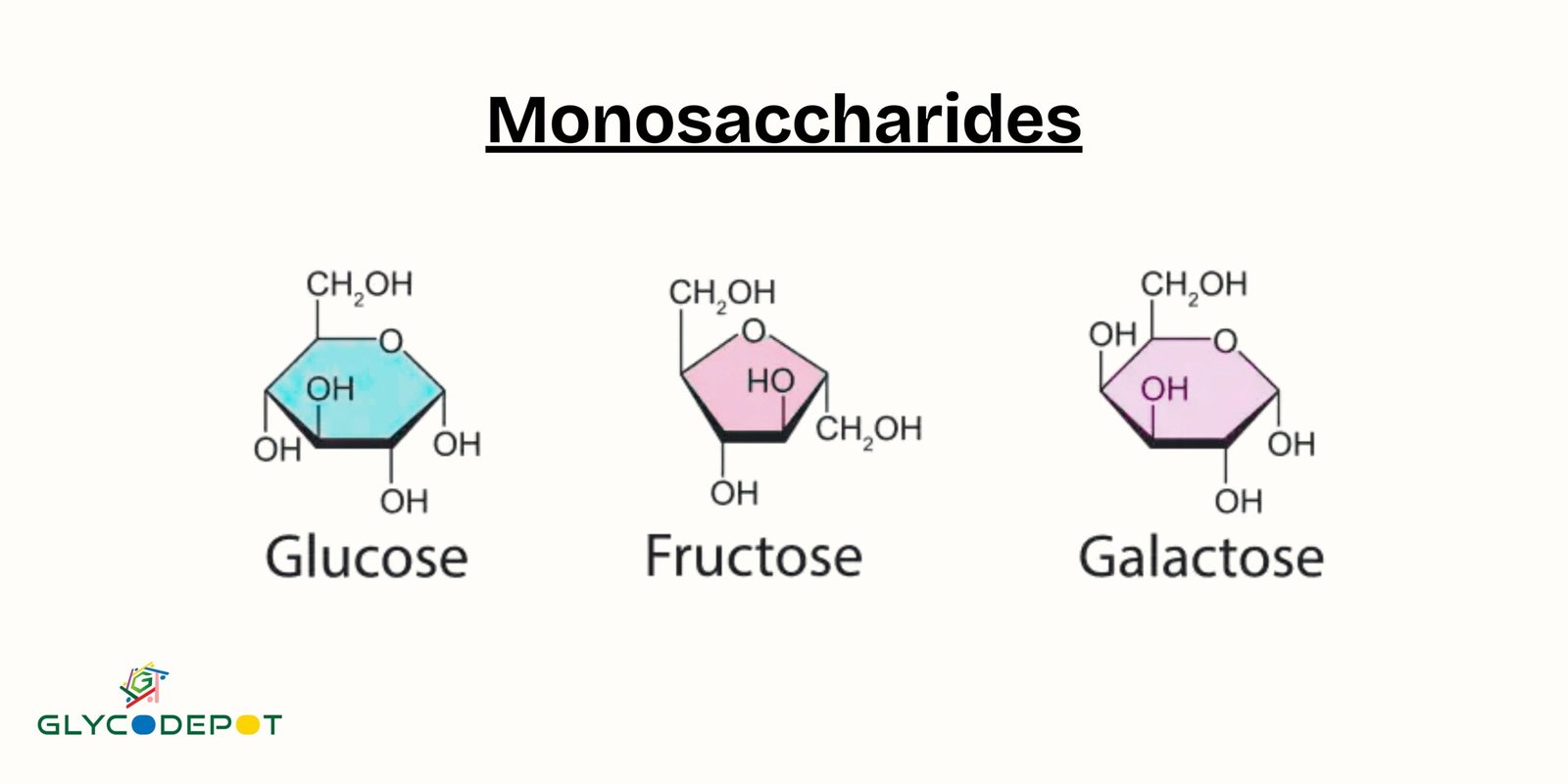
If the carbonyl group is at the end of the carbon chain, forming an aldehyde, then we're dealing with an aldose. Think of glucose and galactose – these are prime examples of aldoses. They’re like the straightforward, no-nonsense members of the monosaccharide family.
On the other hand, if the carbonyl group is somewhere in the middle of the carbon chain, forming a ketone, then we have a ketose. The most famous ketose? You guessed it – fructose! That’s the super-sweet sugar found in fruits. See? Even the names hint at their properties!

So, a true statement about monosaccharide structure would likely revolve around these fundamental characteristics. For example, a correct statement might be: "Monosaccharides contain a carbonyl group and multiple hydroxyl groups." This is a pretty solid, universally true statement about these sugar molecules. It's like saying, "All dogs have four legs and fur" – it's a defining characteristic.
Another accurate statement could be something along the lines of: "Monosaccharides can be classified as either aldoses or ketoses based on the position of the carbonyl group." This highlights the fundamental way scientists categorize these simple sugars. It’s a key piece of the structural puzzle!
It’s important to remember that while they all share these basic features, the exact number of carbon atoms and the specific arrangement of the hydroxyl groups can lead to different types of monosaccharides. This is what gives us the variety we see, from the glucose that fuels our muscles to the fructose that makes fruits so delicious.
Why Does This Even Matter? (Spoiler: It's Fun!)
Okay, so we've touched on aldehydes and ketones. Does this really make life more fun? Absolutely! Think about it: understanding these fundamental structures helps us appreciate why certain foods give us energy, why some are sweeter than others, and how our bodies process them.
It’s like understanding the basic ingredients and techniques that go into making your favorite cake. You don't need to be a master baker to enjoy a delicious slice, but knowing a little about how flour, sugar, and eggs interact can deepen your appreciation for the final product.
And for those of you who love to cook or bake, this knowledge can be a game-changer! Want to achieve a certain texture or sweetness? Understanding the properties of monosaccharides like glucose (which caramelizes beautifully) and fructose (which is sweeter and doesn't crystallize as easily) can help you become a kitchen wizard. It’s empowering!
Even outside the kitchen, this knowledge is part of the amazing tapestry of life. From the energy plants use to grow (thanks, photosynthesis!) to how our bodies build and repair themselves, monosaccharides are working tirelessly behind the scenes. It’s a tiny, sweet reminder of the complex and beautiful processes happening all around us and within us.
So, next time you enjoy a piece of fruit or a sweet treat, take a moment to think about the incredible monosaccharides that are making it all possible. They are the unsung heroes of energy and flavor, and their simple yet elegant structures are a testament to the wonders of chemistry and biology.
Don't you feel a little more inspired? The world of science is full of these fascinating little details that, once you peek behind the curtain, can make everyday life so much more interesting. Keep asking questions, keep exploring, and you’ll find that even the most scientific-sounding topics can be a source of delight and wonder. The journey of learning is, after all, the sweetest adventure of all!
