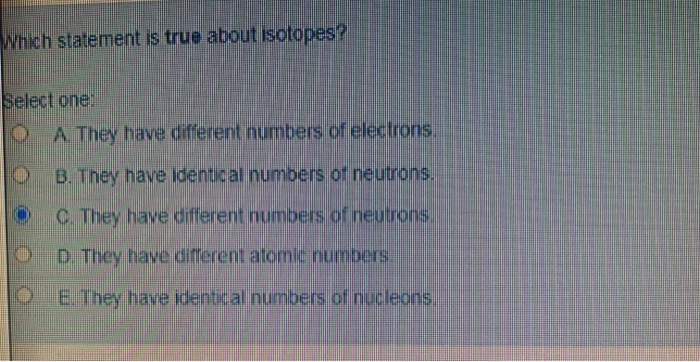
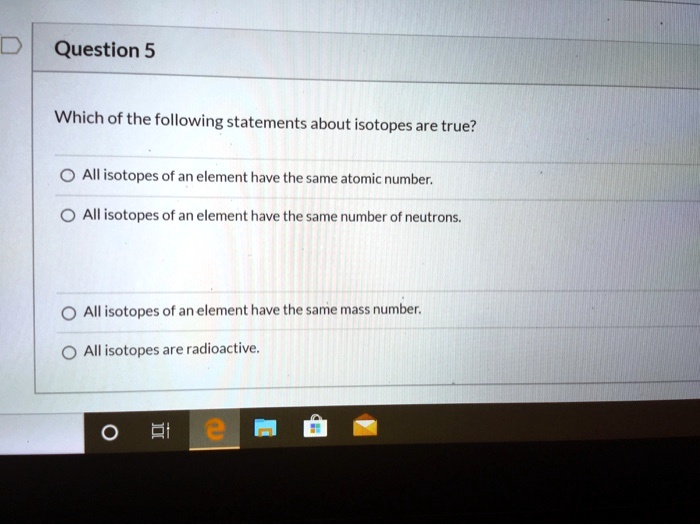
Which Of The Following Statements About Isotopes Is True

Hey there, science enthusiasts and curious cats alike! Ever feel like you're the only one who gets that inside joke with your best friend? You know, the one that makes absolutely no sense to anyone else, but to you guys, it's pure gold? Well, buckle up, because we're about to dive into a little corner of chemistry that's kinda like that: isotopes.
Think of it this way: atoms are like people, and they've got their basic building blocks. You've got your protons (the positive guys, always chirping loudly), neutrons (the neutral folks, just chilling in the background, keeping things steady), and electrons (the negative ones, zipping around like hyperactive puppies). Now, usually, an atom’s identity is pretty much locked down by its protons. If you’ve got 6 protons, you’re carbon. If you’ve got 8, you’re oxygen. Simple, right?
But here’s where it gets interesting, and frankly, a little bit quirky. Sometimes, atoms of the same element can have a slightly different number of neutrons. It's like having two siblings who look almost identical – same hair color, same nose shape – but one's a little bit taller, or has a slightly different laugh. They're still family, still the same core person, but with a subtle, sometimes significant, variation. These are our isotopes!
So, imagine you’ve got a big family reunion. Everyone’s there, the usual suspects. Then, Uncle Bob walks in. He’s definitely a Bob, no doubt about it. But maybe this year, he’s decided to sport a handlebar mustache, or he’s suddenly obsessed with knitting tiny sweaters for squirrels. He’s still Uncle Bob, but he’s got a different vibe, a slightly altered configuration. That’s an isotope. Same element (Uncle Bob), different number of neutrons (handlebar mustache or squirrel sweaters).
The Nitty-Gritty: What Makes Them Different?
The key player here, the real difference-maker, is the neutron. Protons are non-negotiable; they define the element. Electrons? They can get swapped around a bit, leading to ions, but that’s a whole other chat. Neutrons, however, are the wild cards in the atomic deck. They hang out in the nucleus with the protons, acting like a bunch of well-meaning but slightly disruptive roommates.
An atom’s atomic number (the number of protons) is its official ID. It’s like your social security number – always the same for you. But its mass number (protons + neutrons) can change. If an atom has the same number of protons but a different number of neutrons, it’s an isotope of that element.
Let’s take carbon, for example. Everyone knows carbon, right? It's in all of us, it's in your charcoal grill, it's in that fancy diamond ring. The most common form of carbon has 6 protons and 6 neutrons. We call this carbon-12. It’s the plain vanilla, no-frills carbon. It’s like your average Tuesday – predictable and gets the job done.
Then we have carbon-13. This guy also has 6 protons (because it’s still carbon!), but it’s got 7 neutrons. So, it’s a little bit heavier. Think of it as carbon-12’s slightly stockier cousin. He’s still a carbon, but he’s got a bit more heft. He’s like the friend who always carries a bigger backpack, just in case.
And then there’s the celebrity of the carbon family, carbon-14. This one also has 6 protons, but it’s rocking 8 neutrons. Carbon-14 is famous because it's radioactive. It's unstable and likes to… well, decay. More on that later, because it’s pretty darn cool.
So, Which Statement Rings True?
Now that we've got our isotope families straight, let's look at some common statements about them and see which ones are the real deal. It’s like a little pop quiz, but with much less pressure and way more interesting atomic families!
Statement 1: Isotopes have different numbers of protons.
Okay, let’s think about our Uncle Bob analogy. If Uncle Bob suddenly started having a different number of fingers, he wouldn’t be Uncle Bob anymore, would he? He’d be… someone else. Similarly, if an atom has a different number of protons, it’s a different element. So, this statement is a big, fat NO.

Isotopes are about variations within the same element. It's like saying, "These two people are both Smiths, but one has curly hair and the other has straight hair." They're both Smiths. If one was a Jones, they wouldn't be a Smith anymore. Protons are the element's name tag, and it stays the same for isotopes of that element.
Statement 2: Isotopes have different numbers of electrons.
Electrons are like the party guests. They can come and go, and sometimes they get a little bit lost, leading to an atom having a positive or negative charge (that's an ion, remember?). But generally, for a neutral atom, the number of electrons matches the number of protons. While it's possible for isotopes to be in an ionic state (meaning they’ve gained or lost electrons), the defining characteristic of an isotope isn't about electron count. It's about what's going on in the nucleus, the atomic VIP lounge.
So, while an isotope can have a different number of electrons (if it's an ion), that's not what makes it an isotope. The number of protons is still the boss. This statement is also a no, because it’s not the defining difference. It’s like saying "People with red shoes are different from people without red shoes." Well, sure, but that’s not the fundamental difference between, say, a doctor and a lawyer.
Statement 3: Isotopes have different numbers of neutrons.
Ah, we’re back to our star of the show! Remember carbon-12, carbon-13, and carbon-14? They all have 6 protons. But carbon-12 has 6 neutrons, carbon-13 has 7, and carbon-14 has 8. That’s the difference! It’s the neutron count that’s doing the tango here.
This is like the difference between a regular donut and a donut with sprinkles. It's still a donut, same delicious dough, same basic shape, but that sprinkle variation makes it a little bit special, a little bit different. The donut itself is defined by its "donut-ness" (protons), but the sprinkles (neutrons) add that extra layer of variation. So, yes, this statement is absolutely true!
Why Should We Even Care About These Quirky Atoms?
You might be thinking, "Okay, so some atoms are a bit heavier. Big deal." But these subtle differences have some seriously cool real-world applications. It’s not just theoretical mumbo-jumbo for lab coats.
Remember carbon-14? That radioactive isotope we talked about? It’s like nature’s very own stopwatch. Plants take in carbon-14 from the atmosphere as they grow. When an organism dies, it stops taking in new carbon-14, but the carbon-14 it has starts to decay at a predictable rate. Scientists can measure how much carbon-14 is left in ancient artifacts or fossils and use that to figure out how old they are. It’s called radiocarbon dating, and it’s how we know how old that dinosaur bone or that Egyptian mummy really is. Pretty neat, huh? It’s like finding a hidden timestamp on history!
Then there are other isotopes. Uranium isotopes, for example, are used in nuclear power plants. It’s a bit like having a super-powered battery that can keep the lights on for entire cities. It’s all thanks to the different ways these uranium atoms behave.
And what about medical imaging? Certain radioactive isotopes are used to help doctors see inside your body without having to, you know, perform surgery like in the olden days. They can track how certain substances move around your body, or pinpoint areas that need attention. It's like having tiny, invisible guides helping out the medical team. All thanks to the specific properties of certain isotopes!

Even in everyday life, things like hydrogen come in different isotopic forms. We’ve got regular hydrogen (protium), which has no neutrons. Then there’s deuterium, which has one neutron – it’s like hydrogen’s slightly more robust sibling. And then there’s tritium, with two neutrons – the heavy-duty version. Heavy water, which is water made with deuterium instead of regular hydrogen, is even used in some nuclear reactors. So, while you might not be directly playing with isotopes on a daily basis, their influence is pretty widespread.
In a Nutshell (or Atom Shell!)
So, to recap our little atomic journey: Isotopes are like members of the same family, but with a slightly different number of neutrons. They have the same number of protons (which defines the element) but a different mass number. Think of them as different versions of the same character in a story, each with their own little quirks.
The statement that is unequivocally true about isotopes is: Isotopes have different numbers of neutrons.
It’s a simple concept, but it unlocks a world of fascinating science, from dating ancient artifacts to powering our world and helping us stay healthy. So next time you hear the word "isotope," don't let it sound like a foreign language. Just think of Uncle Bob’s new mustache, or that sprinkle-covered donut. It’s just a slightly different version, and that difference can be pretty darn amazing!
Keep on exploring, keep on questioning, and remember, even the smallest variations can lead to the biggest discoveries. Now go forth and share your newfound isotope wisdom with the world (or at least your closest science-loving friend!). You've earned it!
