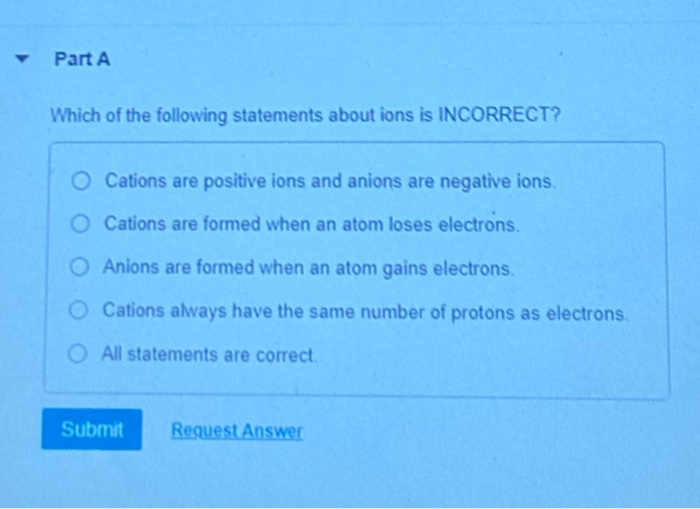
Which Of The Following Statements About Ions Is Incorrect
Hey there, science buddies! Let's dive into the wonderfully weird world of ions. Don't worry, this isn't going to be some stuffy textbook lecture. Think of this more like us grabbing a coffee (or, you know, imaginary science-themed donuts) and chatting about these charged-up little guys. We're going to tackle a classic quiz question: "Which of the following statements about ions is incorrect?" It's like a mini scavenger hunt for a misconception!
So, what exactly are ions? Imagine atoms are like people. Most of the time, they're pretty chill, hanging out with an equal number of positive (protons) and negative (electrons) charges. They're neutral, like someone who's just calmly scrolling through social media. But sometimes, these atoms get a little… energetic. They either decide to gain an electron or lose an electron. And when that happens, BAM! They become an ion. They've gone from neutral to having a definite electrical charge.
Think of it like this: if an atom is a perfectly balanced scale, gaining an electron tips it to the negative side, and losing one tips it to the positive side. It's all about that electron count, my friends. Pretty neat, huh? This whole process is fundamental to how so many things work, from the electricity that powers your phone to the way your body sends signals.
Now, let's get to our quiz question. You'll often see a list of statements, and your mission, should you choose to accept it (and you totally should, because it's fun!), is to spot the one that's just… not right. It's the odd one out, the imposter in the ion club. Let's break down some common statements you might encounter and see why some are totally true and one is… well, not so much.
Understanding the Players: Cations and Anions
Before we start hunting for the incorrect statement, we need to know our main ion characters. We've got two main types, and their names are pretty descriptive if you think about it. First up, we have cations. These are the ions that have a positive charge. How do they get that way? Remember our atom analogy? They lost one or more electrons. Since electrons are negatively charged, losing them leaves the atom with more positive protons than negative electrons. It's like getting rid of your annoying little siblings – suddenly, everything feels a bit more peaceful and positive!
Think of common cations like sodium (Na+) or calcium (Ca2+). They're positively charged and ready to mingle. The 't' in cation can also be a helpful reminder – think of it as a little plus sign: cation = positive.
On the other side of the charge coin, we have anions. These are the ions that have a negative charge. And how do they achieve this state of negativity? By gaining one or more electrons. So, they have more negative electrons than positive protons. It's like adopting a whole litter of puppies – suddenly your house is filled with more chaos and undeniable cuteness (and in this case, negative charge!).

Examples of anions include chloride (Cl-) or oxide (O2-). They're the yin to the cation's yang. And a little trick for remembering: the 'n' in anion could stand for 'negative' or just 'negatively charged'. See? Science can be full of little helpful nudges.
Common Statements and Why They're (Mostly) True
Let's look at some typical statements you might find in our "find the incorrect one" game. Most of these are actually spot-on!
Statement 1: Ions are formed when an atom gains or loses electrons.
This is our foundational truth! As we just discussed, it's all about that electron shuffle. Atoms are stable when they have a balanced number of protons and electrons. When they gain or lose electrons, they upset that balance and become charged. This statement is 100% correct. It's the very definition of ion formation. No funny business here!
Statement 2: Cations are positively charged ions.
Yep, we covered this! Cations are formed when an atom loses negatively charged electrons, leaving it with an excess of positive protons. So, a positive charge is what defines a cation. This is another absolutely correct statement. It's like saying "dogs bark." It's a fundamental characteristic.
Statement 3: Anions are negatively charged ions.
And the flip side of that coin! Anions are formed when an atom gains negatively charged electrons, giving it more negative charge than positive charge. So, if you see an ion with a negative sign (like F-), it's an anion. This statement is also perfectly accurate. It’s the rule, not the exception!
Statement 4: All atoms readily form ions.
Now this is where things start to get a little… nuanced. While many atoms can form ions, it's not quite accurate to say they all readily do. Some elements are really stable as neutral atoms. Think of the noble gases like Helium (He) or Neon (Ne). They're already perfectly happy with their electron configuration and don't have much inclination to gain or lose electrons. They're like the chill hermits of the periodic table – content in their solitude!
Other elements, like the alkali metals (Group 1) or halogens (Group 17), are very reactive and readily form ions. They're practically itching to either lose an electron or gain one to achieve that stable, noble gas configuration. So, while the potential to form an ion exists for most elements (under the right conditions), the readiness varies hugely. This statement is starting to sound a bit shaky, isn't it?
Statement 5: Ions are formed from the nucleus of an atom.
Hold on a minute! This is where our scavenger hunt might be leading us. The nucleus of an atom is where the protons (positively charged) and neutrons (no charge) hang out. It's the dense, central core. While the nucleus does contain the protons that contribute to the atom's identity and its positive charge when it's neutral, the formation of an ion involves the gain or loss of electrons, which orbit the nucleus.
The nucleus itself doesn't gain or lose protons or neutrons when an ion is formed. The number of protons defines the element! If an atom gained or lost protons, it would literally turn into a different element. Imagine your car suddenly turning into a bicycle just because you put more gas in it – not how it works!

So, this statement is fundamentally incorrect. Ions are formed by changing the number of electrons, not by altering the nucleus. The nucleus stays put, happily containing its protons and neutrons, while the electrons come and go. This is a classic misconception, so be on the lookout for it!
Spotting the Incorrect Statement: The Final Showdown
Alright, let's put it all together. If you were presented with a multiple-choice question like:
- Ions are formed when an atom gains or loses electrons.
- Cations are positively charged ions.
- Anions are negatively charged ions.
- Ions are formed from the nucleus of an atom.
Which one would you pick as the incorrect statement? Drumroll, please… it's statement number 4!
Why? Because as we discussed, ions are formed by the gain or loss of electrons, which orbit the nucleus. The nucleus itself, containing protons and neutrons, remains unchanged during ion formation. Changing the nucleus would mean changing the element, not creating an ion of that element. It's the electrons that are the mobile players in the ion game!
It’s like saying a baker is incorrect when they say they make cakes by adding flour and sugar. What if the statement said they make cakes by changing the oven's temperature? That's part of the process, sure, but it's not the fundamental ingredient change that defines the cake's creation. Similarly, electrons are the key ingredients changing for ion formation, not the nucleus.

This is a super common point of confusion, so it's a favorite for quiz makers trying to catch you out. Always remember: electrons move, nuclei stay put (when we're talking about forming ions of a specific element).
Why Does This Matter, Anyway?
You might be thinking, "Okay, so electrons move. Big deal." But it is a big deal! This simple gain or loss of electrons is the bedrock of so many cool chemical and biological processes. It's how salt (sodium chloride, NaCl) forms – the sodium atom gives an electron to the chlorine atom, creating Na+ and Cl- ions that are attracted to each other.
It's how batteries work – ions moving from one side to another creates an electrical current. It's how your nerves send signals – the movement of ions across cell membranes is what carries those messages. So, understanding ions isn't just about acing a test; it's about understanding how the world around you (and inside you!) functions.
A Sweet Little Send-Off
So there you have it! Navigating the world of ions doesn't have to be a chore. Think of it as unlocking a little secret code of the universe. And the next time you see a question asking about incorrect statements regarding ions, you'll know to look for the one that messes with the nucleus. Because the real magic happens with those orbiting electrons!
Keep exploring, keep asking questions, and never be afraid to get a little charged up about science. After all, understanding these tiny charged particles is a step towards understanding the big, amazing world we live in. You've got this, and who knows what other electrifying discoveries you'll make! Happy experimenting (or at least, happy thinking about experimenting)! You're doing great!
