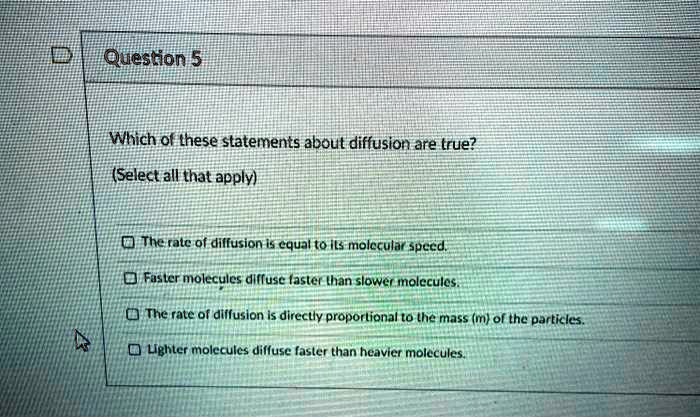
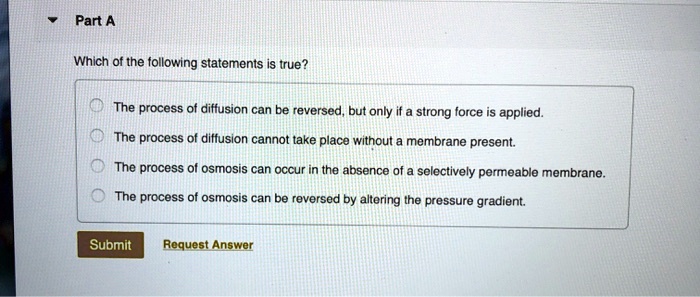
Which Of The Following Statements About Diffusion Is True
Ever get that feeling when you’re in the kitchen, chopping up some seriously fragrant garlic for dinner, and suddenly, the entire house smells like a vampire’s worst nightmare? Yeah, that’s diffusion in action, my friends. It’s basically nature’s way of saying, “Hey, I’m making a mess, and you’re all gonna know about it!”
Think of it like this: you’ve got a super popular kid at school, let’s call him Brad. Brad’s got all the best gossip. What happens? The gossip doesn’t stay with Brad. Nope. It diffuses through the hallways, passed from one person to another, until eventually, even Mrs. Higgins in the library knows that Kevin totally tripped in front of the whole cafeteria. It’s the same with molecules. They’re just tiny little gossipmongers, zipping around and spreading out until they’re everywhere.
So, when you’re faced with a question like, “Which of the following statements about diffusion is true?” and it feels like a pop quiz from your high school science teacher who always wore that questionable tweed jacket, don’t panic! We’re going to break it down in a way that’s as easy-breezy as a summer breeze, or as inevitable as that one relative who always calls on your birthday, no matter what timezone they’re in.
At its core, diffusion is all about things spreading out. It’s the universe’s universal law of “don’t be so selfish, share the space!” Imagine you have a really concentrated cup of coffee. You put a tiny drop of milk in it. At first, the milk is all huddled together, like a little beige blob contemplating its existence. But give it a minute, and that milk starts to mingle. It spreads out, diluting the coffee, and eventually, you have a uniformly creamy (or at least, less intensely black) beverage. That’s diffusion at its finest. No fancy equipment, no secret handshake, just molecules doing their thing.
Let’s dive into some of the nitty-gritty, but we’ll keep it light, I promise. We’re not trying to get you a Nobel Prize here, just a good chuckle and a solid understanding. So, what is diffusion, really? It’s the movement of particles from an area of higher concentration to an area of lower concentration. Think of a crowded concert. Everyone’s packed in like sardines. But as the crowd thins out towards the exits, people naturally spread out. They’re moving from the dense, packed area to the less crowded, more spacious areas. It’s a one-way ticket to mingling, folks.
One of the key players in diffusion is something called the concentration gradient. This is just a fancy way of saying there’s a difference in how many particles are in one spot compared to another. If you have a ton of perfume molecules in the bottle (high concentration) and hardly any in the air outside the bottle (low concentration), that gradient is what’s going to push those perfume molecules out into the world. And boom! Suddenly, your room smells like a fancy department store. You’re basically a walking, talking perfume diffuser.
Now, let’s consider some common misconceptions or statements that might be thrown around. For example, someone might say diffusion requires energy to happen. This is like saying you need to pay a bribe for the sun to rise. While some processes do need energy (like actively pumping something somewhere), diffusion is a passive process. It happens naturally, driven by the inherent random motion of molecules. They’re just bumping into each other and spreading out, like toddlers at a birthday party who’ve had too much sugar. They don't need a push; they're already going a mile a minute.
Another statement you might encounter is that diffusion stops when everything is evenly spread out. This is almost true, but not quite. When the concentration is the same everywhere, we say the system has reached equilibrium. At equilibrium, molecules are still moving, but they’re moving in all directions at the same rate. So, while there’s no net movement (meaning, no more spreading out happening), the party isn’t over for the individual molecules. They’re still dancing, just without any direction. It’s like a perfectly balanced dance floor; everyone’s grooving, but no one’s pushing anyone else to the edge.
What about temperature? Does it play a role? You betcha! Think about a frozen pizza versus a hot pizza. Which one are you going to smell first? The hot one, right? That’s because higher temperatures mean molecules move faster. Faster molecules mean more bumping, more spreading, and quicker diffusion. So, when you’re warming up leftovers, you’re not just warming up your food; you’re essentially fast-forwarding the diffusion process of all those delicious aromas. It’s like giving your food a turbo boost to share its deliciousness with the world.

Let’s get a bit more specific. Imagine you’re in a swimming pool. You accidentally spill a tiny bit of blue dye into a corner. What happens? That blue color doesn’t just sit there in a neat little blob, does it? It starts to spread out, slowly but surely, tinting the water. Eventually, the entire pool might have a faint blue hue. This is diffusion in action, moving from the concentrated area of dye to the less concentrated area of clear water. It takes time, especially in a big pool, but it will happen. It’s a marathon, not a sprint, for those dye molecules.
Another crucial aspect is the medium through which diffusion is occurring. Is it a gas, a liquid, or a solid? Diffusion happens fastest in gases because the molecules are already super far apart and zipping around at lightning speeds. Think of a whiff of smoke from a barbecue – it travels surprisingly far and fast. In liquids, it’s slower because the molecules are closer together and bumping into each other more frequently, like trying to navigate a crowded subway during rush hour. In solids, diffusion is extremely slow. It’s like trying to get a message across a wall made of concrete – it’s going to take a monumental effort and a whole lot of time. Most of the time, when we talk about diffusion in everyday life, we’re thinking about gases and liquids.
Now, let’s talk about what doesn’t happen during diffusion. Diffusion is not about active transport. Active transport is like hiring a moving truck to haul your stuff from one place to another, even if it’s uphill. It requires energy. Diffusion, on the other hand, is like letting gravity do the work of rolling a ball down a hill. It’s effortless, natural, and driven by the inherent tendency of things to spread out.
So, if you see a statement that says, "Diffusion requires energy input," you can probably wave goodbye to that one. It's like saying you need a special invitation for gravity to work. Nope, not how it rolls (or diffuses, in this case).

What about the size of the molecules? Does that matter? You bet. Smaller molecules tend to diffuse faster than larger ones. Think of it like trying to get a bunch of marbles and a bunch of bowling balls through a small opening. The marbles are going to zip through much quicker, right? They’re lighter, smaller, and can move more freely. The bowling balls, well, they’re going to take their sweet time, bumping and grinding their way through. So, in biological systems, tiny things like oxygen and carbon dioxide can diffuse across membranes pretty easily, while bigger molecules might need a little help.
Let’s revisit the scenario of a tea bag in hot water. The tea leaves are all concentrated inside the bag. The water outside is clear. What happens? The tea molecules, from high concentration inside the bag, start to move out into the lower concentration of the water. This is diffusion. It's why your tea gets its color and flavor. If you try to do this with cold water, it’ll take way longer, because, as we discussed, temperature is a biggie.
Consider this: you’re in a stuffy room, and someone opens a window. Suddenly, fresh air starts to come in, and the stale air goes out. That exchange, that mixing of air molecules, is diffusion at work. It’s nature’s way of airing things out, making sure no one suffocates on their own breath. It’s a beautiful, albeit invisible, process.

So, to recap, when you’re trying to figure out which statement about diffusion is true, keep these things in mind:
- It's about moving from high concentration to low concentration.
- It's a passive process – no energy needed, just the inherent jiggling of molecules.
- It happens faster at higher temperatures.
- It's generally faster in gases than in liquids, and super slow in solids.
- Smaller molecules tend to diffuse faster.
- It continues until equilibrium is reached (when concentrations are equal, but molecules are still moving).
Imagine you’re trying to get a secret message to someone across a crowded room. You could shout it (active transport, requires effort and might be embarrassing!). Or, you could whisper it to someone nearby, who then whispers it to someone else, and so on. That whispering chain is like diffusion. It’s a chain reaction of sharing information, spreading out until it reaches its destination.
One more analogy: think about a perfectly mixed batch of cookie dough. You’ve got chocolate chips, nuts, and dough all happily coexisting. If you were to, hypothetically, separate all the chocolate chips into one pile and all the dough into another, it would take a lot of effort to get them back to that perfectly mixed state. Diffusion, conversely, is the natural tendency of those ingredients to end up mixed together in the first place. It’s the universe saying, “Let’s just all hang out together and be one big delicious mixture!”
So, the next time you smell your neighbor’s amazing barbecue wafting over the fence, or you see the color from a tea bag spreading through your mug, you can smile and think, “Ah, yes. That’s diffusion. Just molecules doing their thing, spreading out like butter on toast.” It’s a fundamental principle, essential for life, and surprisingly relatable when you think about all the times you’ve experienced its invisible dance. It’s the unsung hero of smells, flavors, and all sorts of essential biological processes. It’s the universe’s ultimate chill-out zone for molecules, allowing them to spread their wings and explore the vastness of their surroundings. And that, my friends, is diffusion in a nutshell – easy-going, inevitable, and downright everywhere.
