Which Of The Following Statements About Dehydration Synthesis Is False

Alright, science lovers and… well, everyone else! Gather ‘round. We’re about to dive into something that sounds super fancy but is actually happening inside you right now. We’re talking about dehydration synthesis. Sounds like a fancy way to get rid of wrinkles, doesn’t it? If only!
Basically, it’s how your body builds bigger things from smaller things. Think of it like LEGOs. You have tiny little LEGO bricks (monomers), and you snap them together to make a giant castle (polymers). Pretty cool, right? Your body is basically a super-powered LEGO builder.
Now, the “dehydration” part is where things get a little… moist. Or rather, not moist enough. When you link these little LEGO bricks together, your body chucks out a tiny water molecule. Yep, water. It’s like a little farewell gift from the bonding process. So, you’re sacrificing a bit of H₂O to make something bigger and better. Who knew water could be so… sacrificial?
It’s basically chemistry’s way of saying, “Let’s make some cool stuff, but first, let’s get rid of this pesky water!”
This whole dehydration synthesis thing is responsible for some pretty important jobs in your body. It’s how you build proteins, which are like the workers of your cells, doing all sorts of jobs. It’s how you build carbohydrates, which are your body’s favorite snacks for energy. And it’s how you build nucleic acids, like DNA, which is basically your body’s instruction manual. Pretty epic, if you ask me. Your body is constantly busy, like a tiny, well-oiled, molecule-building factory.
So, we have this amazing process. But, like any good story, there are always a few twists and turns. And sometimes, there’s that one statement that just… doesn’t quite fit. That statement that makes you scratch your head and say, “Wait a minute. Is that really true?”
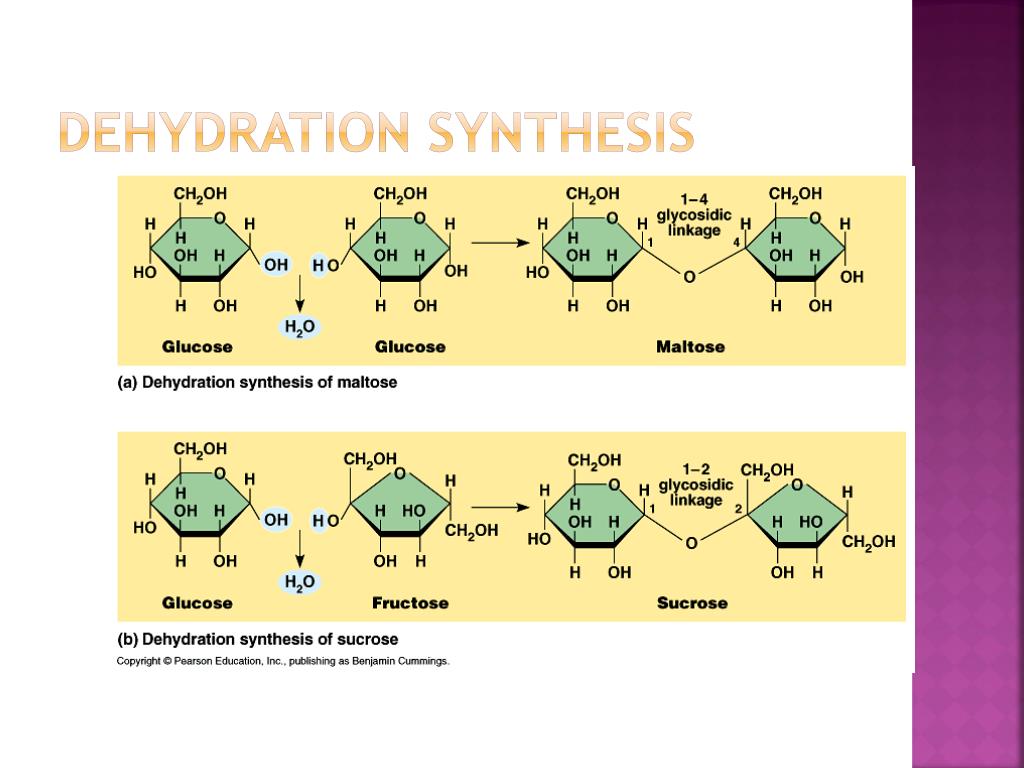
Let’s play a little game. We’re going to look at a few statements about dehydration synthesis. Your mission, should you choose to accept it (and you should, it’s fun!), is to spot the fib. The one that’s just plain wrong. The imposter in the molecule-building party.
Statement Number One:
“Dehydration synthesis is the primary way your body builds large carbohydrates like starch and glycogen.”
Hmm. Does that sound right? Carbohydrates are like those energy-packed cookies your body loves. And dehydration synthesis does build them. So, this one feels pretty legit. It’s like saying, “Pizza is good.” Most of us can agree with that. Seems plausible, doesn’t it?
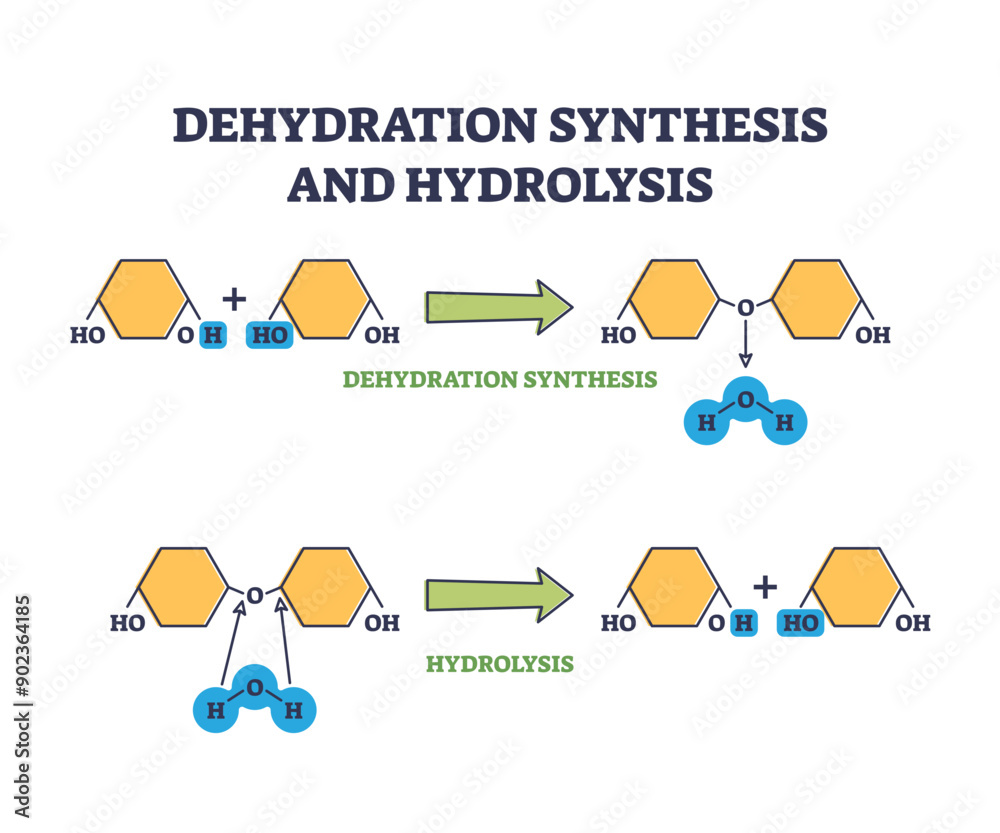
Statement Number Two:
“This process involves the joining of two or more monomers to form a larger polymer.”
Okay, remember our LEGO analogy? Monomers are the little bricks, polymers are the big structures. This statement is basically saying that we’re snapping those bricks together to make something bigger. That’s literally what dehydration synthesis is all about. This one also sounds like it’s telling the truth, the whole truth, and nothing but the truth. It’s the fundamental definition, like saying “Water is wet.”
Statement Number Three:
“A molecule of water is released every time two monomers are joined together.”

Ah, the “dehydration” part. We talked about this! It’s like a little byproduct, a tiny sacrifice for the greater good of building. This statement is essentially confirming that little bit of water going bye-bye. So, it seems to be another true statement. It's like saying, "You have to study to pass the test." Unfortunate, but true for many of us.
Statement Number Four:
“Dehydration synthesis requires the input of ATP energy but does not produce it.”
Now this one… this one makes me pause. Does it need energy? Yes, building things usually does. Like building that LEGO castle, it takes effort. But does it not produce energy? That feels a little… specific. Like saying, “This delicious cake tastes great but provides no joy.” It seems like there might be more to the story. Or perhaps, not more to the story in the way we might expect. This is where the little alarm bells in my head start to chime. It’s the statement that feels like it’s trying a little too hard to convince you of something.

Let’s think about it. Your body is a powerhouse of energy management. It’s always finding ways to store, use, and sometimes even create energy in different forms. To say something never produces energy when it's involved in building essential molecules… that feels like a potential oversimplification, or maybe even a straight-up falsehood. It’s like claiming a chef never tastes their own cooking. Unlikely!
So, out of these seemingly innocent statements, one of them is, shall we say, misleading. It’s the one that tries to paint a picture that isn't quite complete. The one that might lead you astray if you’re not paying close attention to the energy dynamics of your amazing biological machinery.
Which one is it? Which statement about this fascinating dehydration synthesis is the one that’s not quite telling the whole story? It's the one that makes you think, "Hmm, is that always the case?" Because in the grand, glorious, and sometimes confusing world of biology, there’s usually a bit more nuance than a simple “yes” or “no,” or in this case, a simple “doesn’t produce energy.”
