Which Of The Following Reactions Would Be Classified As Oxidation-reduction
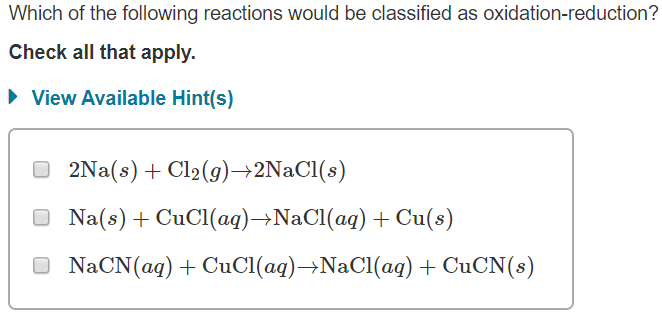
So, I was watching this nature documentary the other day, right? And they were talking about how a caterpillar turns into a butterfly. Absolutely mind-blowing metamorphosis. They described this whole complex biological process, all these intricate chemical changes happening inside. And I was sitting there, munching on some questionable leftover pizza, thinking, "Wow, that's a whole lot of 'stuff' rearranging itself. Is that like… chemistry happening?" And then it hit me, like a rogue ping pong ball to the forehead: that's kind of what we're diving into today. Not butterflies, sadly, but some serious chemical transformations. We're talking about reactions, people! Specifically, the ones that make you go, "Whoa, something got gained and something got lost here."
Remember those days in chemistry class, staring blankly at the whiteboard, trying to decipher the cryptic scribbles of your teacher? If you’re anything like me, there were probably a few moments where you just nodded along, hoping for the best, and secretly plotting your escape route to the nearest vending machine. Well, buckle up, buttercups, because we’re about to demystify a particularly spicy corner of chemistry: oxidation-reduction reactions, also known as redox reactions. And trust me, they’re way cooler than they sound. Think of it as chemistry with a bit of a dramatic flair, where electrons are the ultimate celebrity, being passed around like hot potatoes.
So, the big question, the headline that probably had you clicking: "Which of the following reactions would be classified as oxidation-reduction?" It’s a classic question, isn’t it? Like asking "Which came first, the chicken or the egg?" except way less philosophical and with a much higher chance of appearing on a pop quiz. The answer, my friends, hinges on a fundamental concept: the transfer of electrons. If electrons are getting a bit of a joyride, moving from one atom or molecule to another, then congratulations, you’ve got yourself a redox reaction on your hands!
The Nuts and Bolts of Electron Shifting
Alright, let’s get down to the nitty-gritty. In chemistry, we have these things called oxidation states, or oxidation numbers. Think of them as a sort of “ownership score” for electrons. A positive oxidation state means an atom or ion is losing electrons, or more accurately, it has a greater pull on the shared electrons than it would if it were neutral. A negative oxidation state means it's gaining electrons, or has a stronger hold on them.
Now, here’s the golden rule, the secret handshake of redox reactions: oxidation is the loss of electrons, and reduction is the gain of electrons. See the rhyme? Lose electrons? You’re oxidized. Gain electrons? You’re reduced. It’s like a game of musical chairs, but with subatomic particles. And the crucial part is that these two processes always happen together. You can’t have one without the other. It’s a chemical tag team!
So, when we look at a reaction, we’re essentially scanning for changes in these oxidation states. If an element’s oxidation state goes up during the reaction, it’s been oxidized. If it goes down, it’s been reduced. Simple, right? Well, sometimes. There are a few sneaky exceptions and special cases, but for the most part, this is your cheat sheet.
Spotting the Redox Stars
How do we actually do this? How do we look at a chemical equation and say, "Yup, that's a redox!"? It comes down to assigning oxidation states to each element in the reactants and then doing the same for the products. Then, you just compare. If you see a change for any element, bingo!
Let’s take a classic example, something you might have seen in a textbook or a dimly lit laboratory. The formation of rust, which is basically iron reacting with oxygen. The simplified equation looks something like:
4Fe (s) + 3O2 (g) → 2Fe2O3 (s)
Now, let’s play detective with oxidation states. * On the left side, we have pure iron (Fe). In its elemental form, its oxidation state is 0. It hasn’t lost or gained any electrons relative to itself. * Also on the left, we have oxygen (O2). In its elemental form, its oxidation state is also 0. * Now, let’s look at the product, iron(III) oxide (Fe2O3). This is where it gets interesting. Oxygen in most oxides has an oxidation state of -2. Since we have three oxygen atoms, that’s a total of -6. For the compound to be neutral, the iron must have an oxidation state that balances this out. With two iron atoms, each must have an oxidation state of +3. So, 2(+3) + 3(-2) = +6 - 6 = 0. Makes sense!
So, comparing the reactants and products: * Iron (Fe) goes from an oxidation state of 0 to +3. Its oxidation state increased. That means iron lost electrons. Therefore, iron is oxidized.
* Oxygen (O) goes from an oxidation state of 0 to -2. Its oxidation state decreased. That means oxygen gained electrons. Therefore, oxygen is reduced.
See? Because both oxidation (loss of electrons by iron) and reduction (gain of electrons by oxygen) occurred, this reaction is definitely a redox reaction. It’s like a chemical ballet, with electrons pirouetting from one partner to another.
What About the Non-Redox Reactions?
Okay, so we know what makes a reaction a redox reaction. But what about the ones that aren't? These are often called non-redox reactions or sometimes metathesis reactions, which sounds fancy but basically means "reaction of changing places." Think of them as chemical rearrange-ments where no one’s really losing or gaining electrons in the grand scheme of things. The oxidation states of the elements involved tend to stay the same.
A prime example is a simple acid-base neutralization reaction. Like when you mix hydrochloric acid (HCl) with sodium hydroxide (NaOH):
HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l)
Let’s do our oxidation state check-up:
* Reactants: * In HCl, H is +1, Cl is -1. * In NaOH, Na is +1, O is -2, H is +1.
* Products: * In NaCl, Na is +1, Cl is -1. * In H2O, H is +1, O is -2.
Now, let’s see if anything changed:
* Hydrogen (H): +1 in HCl, +1 in NaOH, +1 in H2O. No change. * Chlorine (Cl): -1 in HCl, -1 in NaCl. No change. * Sodium (Na): +1 in NaOH, +1 in NaCl. No change. * Oxygen (O): -2 in NaOH, -2 in H2O. No change.
Since none of the elements changed their oxidation states, this is not a redox reaction. It's a classic example of ions swapping partners. You could even write it as a net ionic equation, where you see that essentially, H+ ions and OH- ions combine to form water, and Na+ and Cl- ions just hang out in solution. No electron transfer drama here.
Another common type of non-redox reaction is a precipitation reaction, where two soluble ionic compounds react to form an insoluble solid (a precipitate). For instance:
AgNO3 (aq) + NaCl (aq) → AgCl (s) + NaNO3 (aq)
Let’s do the oxidation state tango again:
* Reactants: * AgNO3: Ag (+1), N (+5), O (-2) * NaCl: Na (+1), Cl (-1)
* Products: * AgCl: Ag (+1), Cl (-1) * NaNO3: Na (+1), N (+5), O (-2)
* Silver (Ag): +1 → +1. No change. * Nitrogen (N): +5 → +5. No change. * Oxygen (O): -2 → -2. No change. * Sodium (Na): +1 → +1. No change. * Chlorine (Cl): -1 → -1. No change.
Once again, no oxidation state shifts mean no redox reaction. The silver ions and chloride ions just decided to get together and form a solid, while the sodium and nitrate ions decided to stay dissolved. It’s like a social gathering where some people pair off and leave the party, while others just mingle.
The “Which Of The Following” Scenario
So, when you’re presented with a question like "Which of the following reactions would be classified as oxidation-reduction?", here's your game plan:

- Look for elements in their elemental form: If you see a reaction where an element starts out by itself (like Fe, O2, H2, Cl2) and ends up in a compound, or vice versa, that’s a HUGE red flag. Elemental forms usually have an oxidation state of 0, so if they end up in a compound with a non-zero oxidation state, a change has definitely occurred.
- Identify changes in oxidation states: This is your bread and butter. Assign oxidation states to every atom in the reactants and products. If you find at least one element whose oxidation state increased and at least one element whose oxidation state decreased, then you’ve got your redox reaction.
- Beware of simple ion exchange: Reactions that look like simple swapping of partners, like most acid-base reactions and precipitation reactions, are usually not redox reactions. The key is that the oxidation states remain constant for all elements.
Let’s imagine some hypothetical reaction options:
* Reaction A: 2Na (s) + Cl2 (g) → 2NaCl (s) * Na: 0 → +1 (oxidized) * Cl: 0 → -1 (reduced) * Verdict: REDOX! (Because both Na and Cl changed oxidation states.)
* Reaction B: H2SO4 (aq) + 2NaOH (aq) → Na2SO4 (aq) + 2H2O (l) * Let’s check: H (+1), S (+6), O (-2) in H2SO4. Na (+1), O (-2), H (+1) in NaOH. Na (+1), S (+6), O (-2) in Na2SO4. H (+1), O (-2) in H2O. * Verdict: NOT REDOX! (All oxidation states remain the same. This is an acid-base neutralization.)
* Reaction C: 2H2O2 (aq) → 2H2O (l) + O2 (g) * Hydrogen peroxide (H2O2): H is +1, but O is tricky. In peroxides, oxygen is usually -1. * Water (H2O): H is +1, O is -2. * Oxygen gas (O2): 0. * Oxygen: goes from -1 in H2O2 to -2 in H2O (reduced) AND from -1 in H2O2 to 0 in O2 (oxidized). * Hydrogen: +1 → +1. No change. * Verdict: REDOX! (Even though only oxygen changes, it’s both oxidized and reduced in a process called disproportionation. Pretty neat, huh?)
So, when you see a question like that, it's all about playing the oxidation state game. It's not about memorizing every single reaction; it's about understanding the underlying principle of electron transfer.
Why Should We Care About Redox?
Honestly, the fact that we’re talking about electrons being tossed around is cool enough, but redox reactions are everywhere and they are super important. Think about it:
- Energy production: How do we get electricity? Often through burning fuels (combustion is a redox reaction!). How do our bodies get energy from food? Cellular respiration, a complex series of redox reactions.
- Corrosion: That rust we talked about? It’s a constant battle against redox reactions destroying our metal structures.
- Batteries: Ever used a phone, a remote, or a car? You're using redox reactions to store and release electrical energy.
- Life itself: Photosynthesis, the process plants use to create energy from sunlight, is a redox reaction.
So, the next time you see a chemical equation, take a second glance. Are electrons being transferred? Is someone gaining and someone losing? If the answer is yes, then you’re looking at a redox reaction, a fundamental process that powers our world. It’s not just abstract chemistry; it’s the engine behind so much of what we see and do every day. And that, my friends, is pretty darn fascinating.
