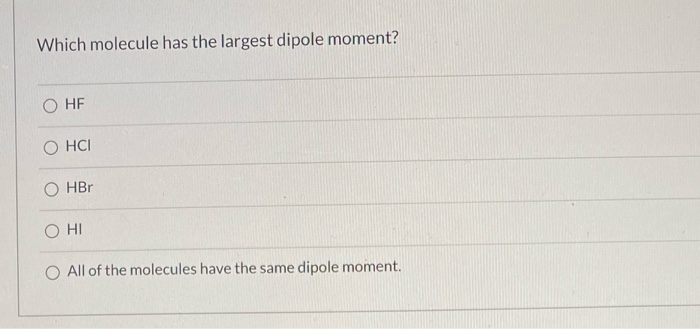
Which Of The Following Molecules Has The Largest Dipole Moment

Ever wondered which molecule throws the biggest "party" when it comes to electricity? It's like picking the star of a molecular dance-off, and we're here to spill the tea on who's got that electrifying charisma! Get ready to dive into the quirky world of dipole moments, where tiny particles get surprisingly charged up!
Imagine molecules as little teams of atoms holding hands. Sometimes, these atoms share their toys (electrons) equally, like a perfect team. But other times, one atom is a bit of a hog and pulls the shared toys closer. This uneven sharing creates a sort of tiny imbalance, like a little tug-of-war.
That imbalance is what we call a dipole moment. It's like a mini-battery within the molecule, with one end slightly positive and the other end slightly negative. Some molecules are just chill and share their toys perfectly, meaning they have no dipole moment. Others are like super energetic dancers, constantly shifting their charges around!
So, the big question is: which molecule wins the award for the biggest, most spectacular dipole moment? It's a real nail-biter, a scientific mystery that has chemists buzzing with excitement. Think of it like guessing who's going to win the Nobel Prize, but way more down-to-earth and, dare we say, cuter!
We're talking about a competition where the "heaviest" dipole moment doesn't necessarily mean the biggest molecule. It’s all about how the atoms are arranged and how willing they are to share (or not share!) their precious electrons. It's a delicate dance of attraction and repulsion, a molecular ballet that determines who's got the most oomph.
Now, when we throw around terms like "dipole moment," it might sound a little intimidating. But trust us, it's more fun than a barrel of monkeys. It’s the secret sauce that makes some molecules behave in totally unexpected ways. They can stick to each other, dissolve in water, or even conduct electricity – all thanks to their little internal battery!
The contenders in this thrilling molecular showdown are a diverse bunch. We've got simple ones that are like the underdogs, and then there are the complex giants that might surprise you. Each one brings its own unique personality to the table, making this a truly captivating spectacle.
Think about it: we’re not just looking at abstract numbers here. We’re talking about real-world magic! The dipole moments of molecules are the reason why water can dissolve sugar, why your soap lathers up, and why your phone screen is so responsive. It’s the invisible force that shapes so much of our daily lives.
So, who do you think has the biggest dipole moment? Is it a molecule that’s super symmetrical, or one that looks like it’s constantly being pulled in different directions? The suspense is killing us, and we’re sure it’s killing you too!
Let's get a little closer to the action. We're not going to give away the grand prize just yet. But we can tell you it involves a molecule that’s a bit of a diva. It’s got a strong personality and isn't afraid to show it. It's the kind of molecule that commands attention.
Imagine a molecule with atoms that have very different appetites for electrons. One atom is like a vacuum cleaner, sucking up all the electron "food." The other atom is left feeling a bit empty. This creates a significant charge difference, a really pronounced tug-of-war.
This extreme sharing imbalance is what leads to a large dipole moment. It’s like the molecule has a really strong internal magnet, pulling things towards its negative side and pushing things away from its positive side. This makes it very interactive with other charged or polar molecules.
The race for the largest dipole moment is not just about individual atoms. It’s also about how those atoms are arranged in space. A molecule can have polar bonds, but if they are arranged in a perfectly symmetrical way, their effects can cancel each other out. Think of it as two people pulling on a rope in opposite directions with equal force – no movement!
But if those polar bonds are arranged in a way that they all add up, you get a substantial net dipole moment. It’s like having a team of people all pulling on the rope in the same direction. That’s when you see some serious molecular power!
So, the winner is a molecule that cleverly combines both a significant difference in electron-holding power between its atoms and an asymmetrical arrangement of these attractions. It’s a molecular masterpiece of unevenness!
The chemistry behind this is fascinating, and the implications are mind-blowing. Understanding these dipole moments helps scientists design new materials, develop medicines, and even create cleaner energy sources. It’s the little things that often have the biggest impact, right?
We’re talking about molecules that are practically celebrities in the scientific world. They’re the ones that grab headlines (in chemistry journals, at least!) for their impressive electrical personalities. They are the MVPs of molecular interactions.

So, when you look at a chemical formula, do you see just letters and numbers? Or do you start to imagine the tiny, energetic dance happening within each molecule? The world of chemistry is full of these hidden marvels, and dipole moments are definitely one of the most exciting.
It’s like having a secret code to understand how the world works at its most fundamental level. And this particular molecule, with its colossal dipole moment, is a prime example of that hidden power. It’s a true testament to the ingenious design of nature.
The journey to discover which molecule holds the title of "Largest Dipole Moment" is a thrilling adventure. It's a testament to the curiosity and ingenuity of scientists who delve into the microscopic world to uncover its secrets. They’re like detectives, piecing together clues to understand the invisible forces at play.
And the prize? Not a shiny medal, but a deeper understanding of the universe. It's the satisfaction of unraveling a complex puzzle and the knowledge that this understanding can lead to incredible advancements. It's a win-win for science and for humanity!
So, if you ever find yourself looking at a chemical structure and wondering, "What's the deal with this one?", remember the concept of the dipole moment. It's the key to unlocking the molecule's personality and its potential to interact with the world around it.
This particular molecule we're hinting at isn't just a winner; it's a showstopper. It demonstrates the power of asymmetry and electron-attracting differences in a spectacular way. It’s the kind of molecule that makes you say, "Wow!"
It’s a constant reminder that even the smallest things can have a monumental impact. And in the grand scheme of molecular interactions, a large dipole moment is a very big deal indeed. It dictates how molecules behave and, by extension, how our world functions.

So, keep your eyes peeled for this electrifying molecule. It’s out there, making its mark, and proving that in the world of chemistry, sometimes the biggest moments come in the smallest packages. It’s a true chemical superstar, and its story is just beginning!
The exploration of such molecular properties is what drives scientific discovery. It's the passion for understanding the fundamental building blocks of matter that leads to breakthroughs. And the pursuit of knowing which molecule reigns supreme in the dipole moment arena is a testament to that ongoing quest.
It’s a journey filled with calculation, observation, and often, a healthy dose of surprise. Because in the world of molecules, things are rarely as straightforward as they seem. There are always hidden nuances and unexpected twists that keep things exciting.
And that, dear reader, is why the question of "Which Of The Following Molecules Has The Largest Dipole Moment" is so darn entertaining. It’s a gateway to a universe of invisible forces, a peek into the secret lives of atoms, and a reminder that science can be incredibly, wonderfully, and sometimes hilariously, complex!
So, go forth and ponder the dipole moments! Who knows what other molecular marvels you might discover. The universe is waiting to be explored, one charged particle at a time!
The beauty of this topic is that it’s not just for the lab coat crowd. Anyone can appreciate the idea of tiny electrical charges and how they influence everything. It’s a fundamental concept that underlies so much of what we experience.
And the molecule with the largest dipole moment? It's a fascinating study in electron distribution and molecular geometry. It’s a prime example of how subtle differences at the atomic level can lead to dramatic macroscopic properties. It's a true marvel of chemical engineering, albeit nature's own.

The next time you're curious about how things work, remember that the answer might just lie in the invisible dance of electrons within molecules. It’s a world of wonder, and we’ve only just scratched the surface!
This quest to identify the molecule with the largest dipole moment is a fantastic way to get people excited about chemistry. It’s relatable, it’s intriguing, and it has real-world implications. It’s the perfect recipe for scientific curiosity!
So, let the molecular guessing game continue. The thrill of discovery is in the journey, and the world of chemistry is full of exciting destinations. And who knows, you might just find yourself captivated by the electrifying personalities of molecules!
The pursuit of knowledge is a grand adventure, and understanding dipole moments is a captivating chapter. It’s a testament to human ingenuity and our insatiable desire to unravel the mysteries of the universe. And this particular molecular competition is a brilliant illustration of that spirit!
So, keep exploring, keep questioning, and keep marveling at the incredible complexity and elegance of the molecular world. The molecule with the largest dipole moment is waiting to be fully appreciated for its electrifying contributions!
It’s a topic that sparks the imagination and reminds us that even the smallest components of matter hold immense power and fascinating stories. The world of chemistry is a playground of discovery, and dipole moments are a key to unlocking its wonders!
So, the next time you think about molecules, remember this exciting challenge. It’s a fun way to explore the unseen forces that shape our reality and a reminder of the endless fascinating discoveries waiting to be made in the realm of chemistry.
