Which Of The Following Is True For Exergonic Reactions
:max_bytes(150000):strip_icc()/endergonic-vs-exergonic-609258_final-2904b2c359574dfcb65a9fca2d54179a.png)
Hey there! Grab your mug, settle in. We're gonna chat about something super cool in the science world, a little something called exergonic reactions. Sounds fancy, right? But honestly, it's not as intimidating as it might seem. Think of it like this: sometimes in life, things just... happen and release a bunch of energy. Like when you finally finish a huge project, and you just feel this whoosh of relief and energy, you know? That's kinda what we're talking about here, but on a molecular level. It’s where the magic, or at least the science, really happens!
So, what’s the big deal with these exergonic reactions? Basically, they're the ones that are happy to go. They're like the extroverts of the chemical world, always ready to mingle and release some good vibes – in the form of energy, of course. We're talking about reactions that are, shall we say, enthusiastic about happening. They've got this built-in motivation, a little internal push that makes them want to get on with it. It’s like they’ve got their little molecular bags packed and are ready to hit the road. No coaxing required, which is pretty neat, right?
Let's break it down, shall we? The core idea is all about energy. You know, that stuff that makes things go, makes your phone work, makes you dance (or at least tap your foot). In chemistry, we talk about this energy in terms of reactants (the stuff you start with) and products (what you end up with). With exergonic reactions, it’s a special kind of energy trade. It’s a net release, like a sweet little gift from the universe. The reactants have more energy stored up than the products do. Imagine you've got a super bouncy ball at the top of a hill. When you let it go, it rolls down, right? And as it rolls, it’s releasing that stored gravitational potential energy. That's a pretty good analogy for what's going on here.
So, what's the key takeaway? The most important thing to remember about exergonic reactions is that they release free energy. Think of "free energy" as the energy that's actually available to do work. It's not like all the energy in the reactants is just gone; some of it is still there, but the amount that's usable for doing stuff decreases as the reaction proceeds. And that decrease is precisely the energy that gets kicked out. It's like having a perfectly ripe apple. You can eat it, and it provides you with energy. The apple itself doesn't have as much potential energy to sit on a shelf anymore; it's given that energy over to you. You get the energy, and the apple... well, it becomes part of you! Pretty cool trade, if you ask me.
One of the coolest aspects is that these reactions are often spontaneous. Now, "spontaneous" in science doesn't necessarily mean it happens instantly with a poof. It just means that, thermodynamically speaking, it's favorable. It doesn't require a constant input of energy from the outside to keep it going. It’s like, if you set up dominoes, and you knock over the first one, the rest will fall. That first nudge is the push, but the subsequent falling is spontaneous, driven by the energy released from each domino toppling. Exergonic reactions have that built-in domino-effect potential. They're ready to roll!
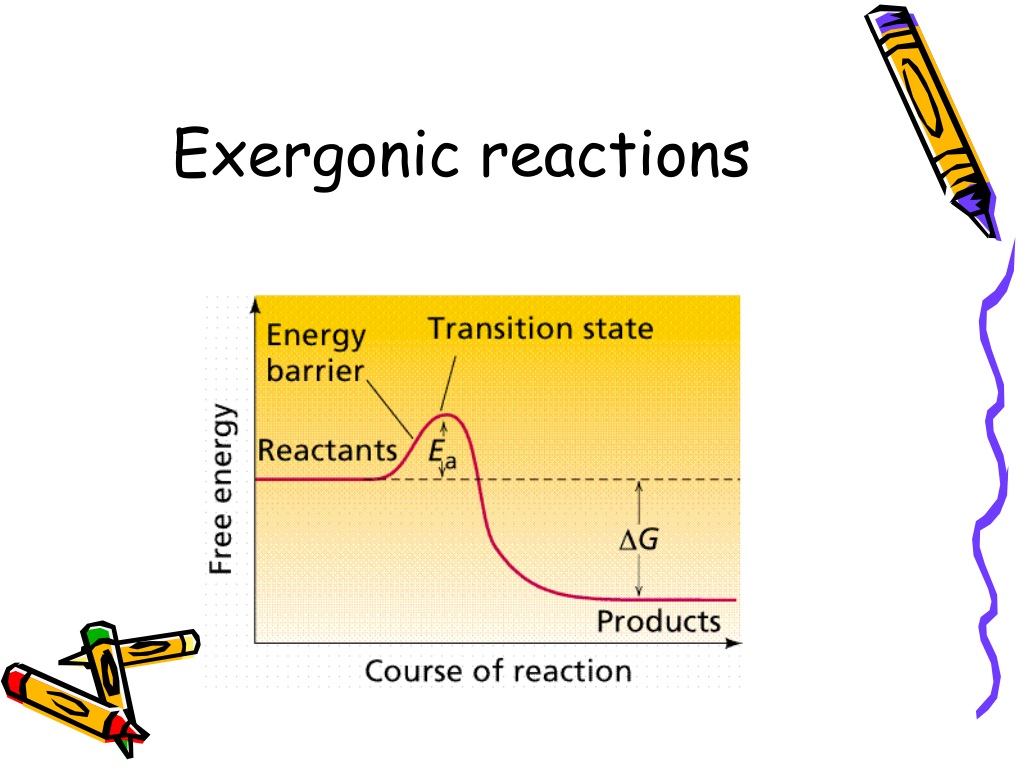
So, if we had to pick a true statement about exergonic reactions from a list, what would be on it? Let’s brainstorm some possibilities, shall we? This is where we get to be detectives of delicious chemistry. What must be true for these energetic little guys?
Possible True Statements (Let's See What Sticks!)
Statement 1: They absorb free energy. Hmm, absorb? That sounds like the opposite of releasing, doesn't it? If they're absorbing, they're taking in. Exergonic reactions are all about giving, not getting. So, nope, this one's a definite no-go. It's like trying to fill a leaky bucket – it's just not gonna hold. This is more like what endergonic reactions do, bless their energy-hoarding hearts.
Statement 2: They have a positive change in free energy (ΔG > 0). Now, this one's interesting. Remember how we said they release free energy? A positive change in free energy would mean they're gaining energy overall. That's not our exergonic friends. They're all about that negative delta G. Think of it like a bank account. If your balance goes up (positive change), you've gained money. If it goes down (negative change), you've spent money. Exergonic reactions are the spenders, the ones that end up with less energy in their system.

Statement 3: They release free energy (ΔG < 0). Ah, there it is! Bingo! This is the golden ticket. A negative change in free energy, often represented as ΔG (delta G), is the hallmark of an exergonic reaction. It means that the system, as it transitions from reactants to products, has lost energy to its surroundings. That energy that was lost? That's the free energy released. It’s like when you finish a delicious meal and feel all warm and content – that’s energy released (and used!). It's the universe’s way of saying, "Here you go, have some of this!"
Statement 4: They require a constant input of energy to proceed. Again, not quite. While some reactions might need a little nudge to get started (we call that activation energy, a whole other fun topic!), exergonic reactions, once initiated, don't need that constant energy supply. They are self-perpetuating to a degree because they're releasing energy as they go. Think of a snowball rolling down a hill. It picks up more snow as it goes, getting bigger and faster. It’s not being pushed the whole way; it's using its own momentum and the surrounding snow. Exergonic reactions have that kind of inherent drive.
Statement 5: The products have more free energy than the reactants. Let’s do a quick check. If the products have more energy, where did that extra energy come from? It must have been absorbed by the system. And if energy is absorbed, that means we're not dealing with an exergonic reaction. We're back to our endergonic friends. So, for exergonic reactions, it’s definitely the other way around: the reactants are the energy-rich ones, and the products are the less energy-rich ones, having gifted some of that energy away. It’s like a wealthy person giving away some of their fortune.
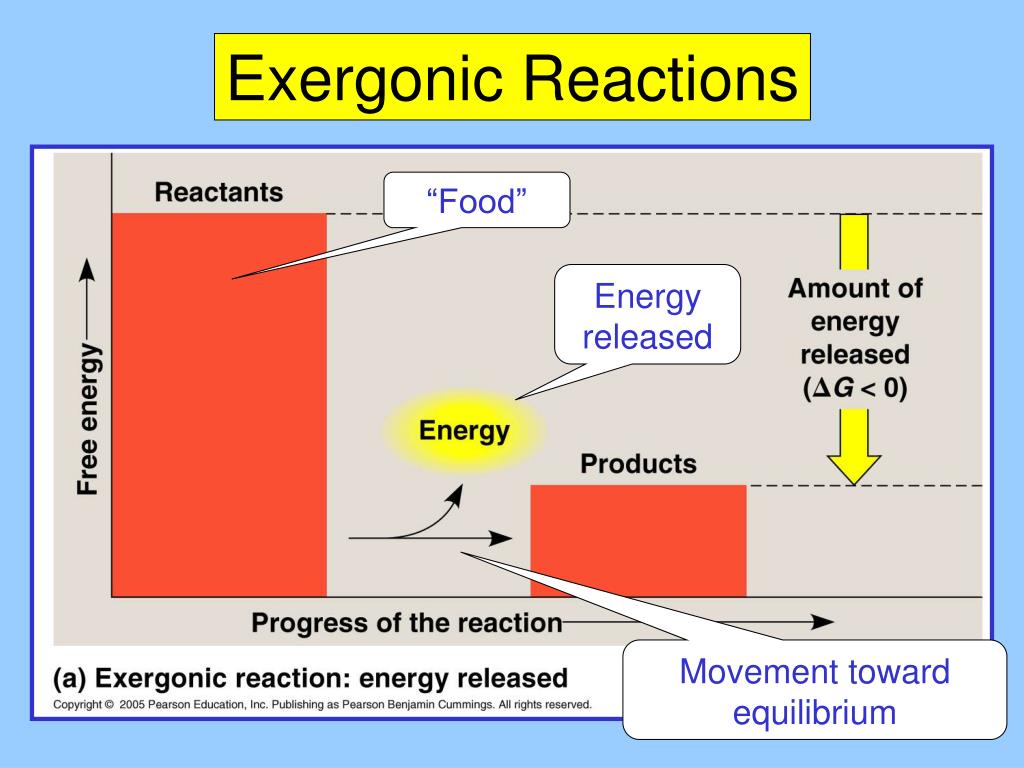
So, when you see those options, you’re looking for the one that screams "energy released!" or "negative ΔG!" It’s all about the net flow of energy out of the system. It’s the difference between something that’s energetically favorable and something that’s energetically unfavorable. Think about burning wood. That gives off a ton of heat and light, right? That’s a classic exergonic reaction. The wood (reactants) had a lot of stored chemical energy, and when it burns (reaction), it releases that energy as heat and light (products). The leftovers (ash) have way less energy than the original wood.
Another way to think about it is the stability of the molecules. Molecules that have a lot of stored energy are generally less stable. They’re like a tightly wound spring, ready to release that tension. When they react and become more stable products, they release that stored energy. So, exergonic reactions often lead to more stable products. Stability is a good thing in the molecular world, kind of like being calm and collected. And reaching that calm state often involves releasing some pent-up energy.
Let's consider some everyday examples. When you digest your food, that's a whole series of exergonic reactions happening in your body! Your body breaks down complex molecules into simpler ones, and in the process, it releases energy that you use to, well, live! Breathing is another great one. When you inhale oxygen and exhale carbon dioxide, there's a whole lot of energy being managed within your cells. Cellular respiration, the process of creating ATP (the energy currency of your cells), is a prime example of exergonic reactions at play. It’s a continuous cycle of energy release and utilization, keeping you going.
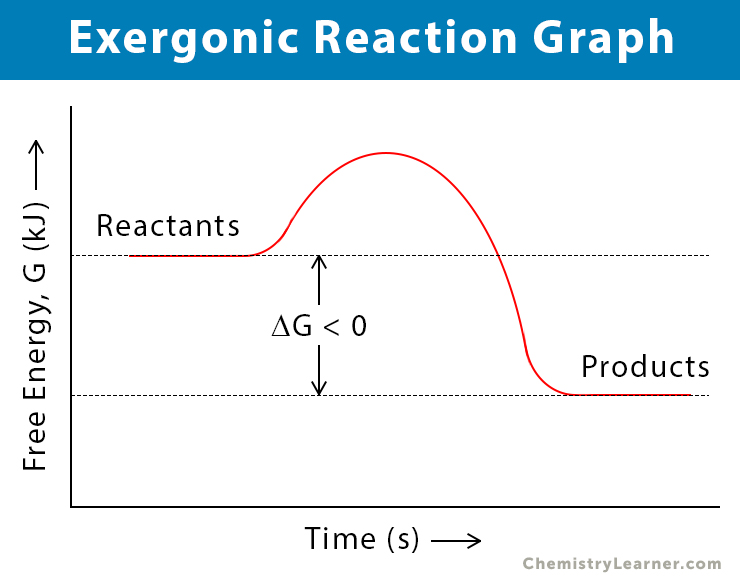
Even simple things like rusting metal are exergonic reactions. Iron reacts with oxygen to form iron oxide (rust). This reaction releases energy, although it might be slow and spread out, so you don’t necessarily see a dramatic flash of light. But it’s happening! It’s the universe’s way of finding a more stable, lower-energy state for the iron. It's like the iron is saying, "You know what? This whole reactive state is exhausting. I’d rather be rust, thank you very much. And I'll give you some energy as I make this transition!"
So, when you're faced with a question about exergonic reactions, just remember the core concept: energy out. It's about a net decrease in free energy. The system is giving something back to the universe. It's the opposite of needing to push hard to make something happen. These are the reactions that are happy to happen, that provide the energy for all sorts of processes, from the grand scale of stars burning to the tiny, intricate workings of your own cells. It's fundamental to life and the world around us. Pretty powerful stuff, wouldn't you agree?
It’s like the difference between trying to inflate a balloon (that requires energy input, an endergonic process) versus letting a fully inflated balloon go (it shoots off, releasing energy, an exergonic process). One is about putting energy in, the other is about energy coming out. And the one that’s all about energy coming out is our exergonic friend. Always remember that key distinction, and you'll be a pro at spotting these energetic reactions. It’s all about the energy balance, the give and take of the molecular world. And exergonic reactions are definitely on the "give" side, which is pretty essential for everything we do. So, next time you feel a burst of energy, you can think of those amazing exergonic reactions working their magic!
