Which Of The Following Is True For All Exergonic Reactions

Hey there, curious minds and life-lovers! Ever feel like your brain just loves a good puzzle, especially when it comes to figuring out how the world around us ticks? Well, today we're diving into a little something that might sound a bit science-y at first, but trust me, it's actually pretty darn cool and can even make understanding your own energy levels a whole lot more fun. We're talking about exergonic reactions. Sounds fancy, right? But don't let that scare you!
Think of your body like a super-efficient engine. It's constantly doing things – breathing, thinking, doing that epic dance move you invented in the shower. All these amazing feats require energy. And where does that energy come from? From chemical reactions! Specifically, from reactions that are kind enough to release energy. And that, my friends, is where our exergonic buddies come in.
So, let's play a little game. Imagine we've got a few statements about these energy-releasing reactions, and we need to pick the one that's always, without fail, true for every single one of them. It's like a cosmic trivia question! Ready to put on your thinking caps? Let's break it down, shall we?
The Great Exergonic Investigation!
Okay, so we're on a quest for the one true statement about exergonic reactions. It’s like finding the secret ingredient that makes a perfect cookie – you just know it when you find it!
Let's consider some possibilities. Maybe it's about how fast they happen? Or what kind of stuff they're made of? Or perhaps, and this is the big one, it's about the energy itself. After all, we did say they release energy, so that seems like a pretty good place to start.
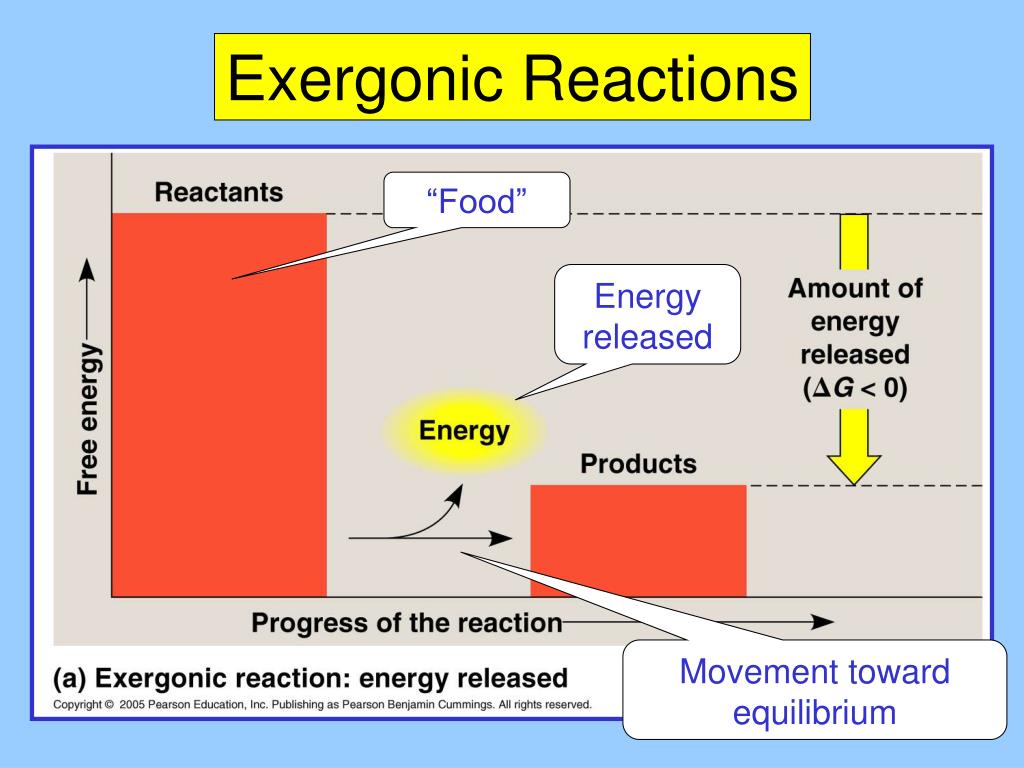
Here's the scoop: when we talk about energy in reactions, we often look at the difference in energy between the starting materials (we call these reactants) and the things you end up with (the products). It's like comparing the ingredients you start with to the delicious meal you create.
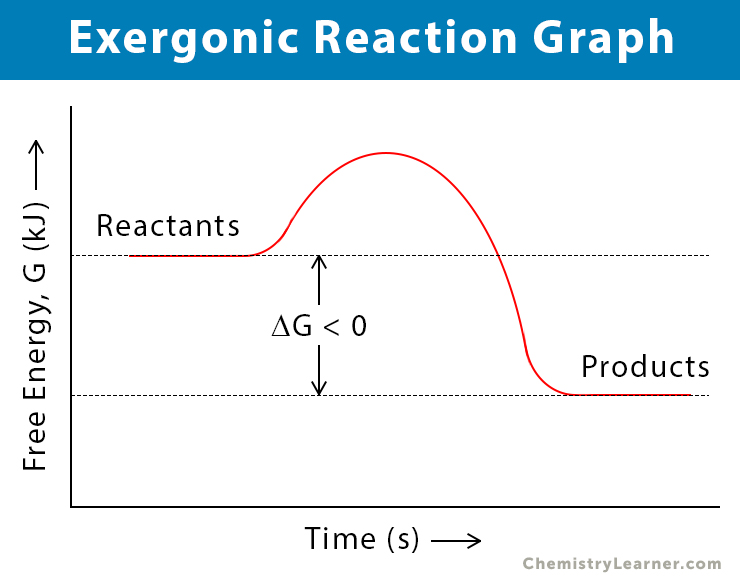
Now, for exergonic reactions, the magic happens because the reactants have a higher energy level than the products. Think of it like a ball sitting at the top of a hill. It has a lot of potential energy, right? When you let it go, it rolls down, releasing that energy as it moves. That's the essence of an exergonic reaction!
So, the true statement for all exergonic reactions is that they release free energy. Woah! What's "free energy," you ask? Well, in the world of chemistry, it’s the energy available to do work. So, these reactions are basically giving us a little energy boost, a little "oomph" to get things done. Isn't that neat?
Let's recap this super important point. The core characteristic of any exergonic reaction, no matter how big or small, how fast or slow, is this: it’s a generous giver of energy. It’s like the friend who always brings snacks to the party!
Why This Matters (Besides Being Super Smart!)
You might be thinking, "Okay, but how does knowing about 'free energy release' make my Tuesday any more exciting?" Ah, my friend, that's where the fun really begins!
Understanding exergonic reactions is like having a secret decoder ring for life's processes. Think about digestion. When you eat food, your body breaks it down through a series of reactions. Many of these are exergonic, releasing the energy your cells need to power everything from your heartbeat to your ability to laugh at a silly meme. Pretty cool, huh?
Even something as simple as rusting is an exergonic reaction! Though you might not want to intentionally rust your car, it's still a demonstration of energy being released into the environment. Nature is constantly buzzing with these energy exchanges.
And get this: even when we want to build things up – like muscle tissue or complex molecules – we often rely on exergonic reactions to provide the energy needed for those building processes. It's like needing fuel to power a construction crew. One set of reactions gives us the power, and another set uses that power to build.

So, the next time you feel a burst of energy, whether it's from a good meal or a great night's sleep, you can give a little nod to those unsung heroes: exergonic reactions, tirelessly working behind the scenes to keep you going!
Making Life More Fun with a Little Science!
Honestly, the more you understand about how things work, the more you can appreciate the intricate dance of the universe. It adds a layer of wonder to the everyday. You start seeing energy transformations everywhere!
Think about the sun! That magnificent ball of fire is a giant exergonic reaction (or rather, a whole bunch of them!). It's constantly releasing energy that warms our planet, helps plants grow, and basically makes life as we know it possible. Talk about an ultimate energy provider!

Even when you're feeling a bit sluggish, you can remember that your body is a marvel of chemical processes. And the ones that are giving you the energy to get up and move? Yep, you guessed it – exergonic reactions!
It's about seeing the world not just as a collection of objects, but as a dynamic system of energy flow. And that, my friends, is a truly inspiring perspective.
So, the next time you encounter the term "exergonic reaction," don't just glaze over. Instead, smile! You know its secret. You know it’s the generous one, the energy-giver, the essential spark that keeps so much of life humming. And that knowledge? That’s energy you can use to learn even more!
Keep that curiosity alive! The universe is full of amazing explanations waiting to be discovered, and each one can add a little more sparkle and understanding to your day. Go forth and be wonderfully, energetically curious!
