Which Of The Following Is The Most Stable Diene
Hey there, science fans! Ever wondered what makes some molecules just… hang out better than others? It’s like choosing your favorite snack – some are just more satisfying, right? Well, in the wild world of chemistry, there’s a similar kind of "best" when it comes to things called dienes. These are molecules with a couple of double bonds, and they’re pretty cool. Today, we’re going to dive into a little game of “Who’s the chillest molecule around?” when it comes to dienes.
Imagine you have a bunch of friends at a party. Some friends are super high-energy, always bouncing around and ready for anything. Others are more laid-back, happy to just chill in a corner and observe. In the world of dienes, we have a similar spectrum of "chillness," or as the fancy scientists call it, stability. It’s all about how much energy it takes to get them to do something, or how likely they are to, well, stay as they are.
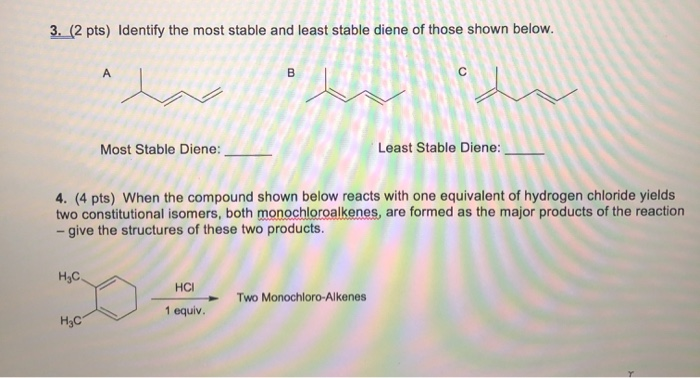
So, the big question we’re pondering is: Which of the following is the most stable diene? Now, I can’t actually give you the list to choose from, but I can tell you what makes one diene a real superstar of stability compared to its buddies. Think of it like picking a champion in a friendly competition. We’re looking for the one that’s just… the most content. The one that’s not itching to change or react.
One of the main reasons a diene might be super stable is because of something called conjugation. Don't let the big word scare you! Conjugation is basically when those double bonds are perfectly spaced out. Imagine them doing a little synchronized dance, with just one single bond in between. This special arrangement lets the electrons – those zippy little particles – spread out and relax. It's like giving them more room to breathe, and when electrons can spread out, the molecule becomes much happier and less likely to get into any kind of chemical kerfuffle.
When double bonds are conjugated, they can share their electron-loving vibes. This sharing, or delocalization, is a huge deal in making a molecule stable. It's like a group of friends all chipping in to buy a comfy couch. Instead of everyone trying to sit on their own lumpy chair, they pool their resources, and poof – a super comfy couch for everyone! In a conjugated diene, the electrons aren't stuck in one place; they can roam around the whole system, and this freedom makes the whole molecule more at ease.
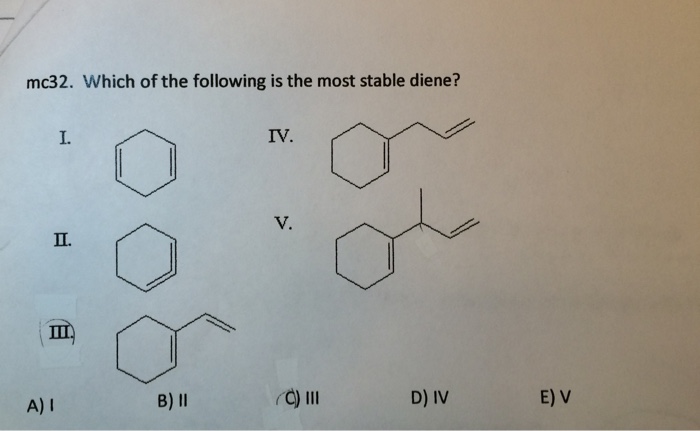
Now, some dienes have their double bonds sitting right next to each other, like a jumbled mess of friends all trying to talk at once. These are called cumulated dienes, and they're usually not as chill. They’re a bit more eager to get involved in reactions. Then you have dienes where the double bonds are far apart, separated by a bunch of single bonds. These are called isolated dienes. They behave pretty much like two separate double bonds, and while they're okay, they don't get that extra boost of stability that conjugation provides.
So, if you were to see a list of dienes, the one that’s likely to be the most stable is the one with its double bonds separated by exactly one single bond. This is the hallmark of a conjugated diene. It’s like the molecule has found its perfect rhythm, its sweet spot. This perfect spacing allows for that magical electron delocalization, making it the calmest and most content on the list.
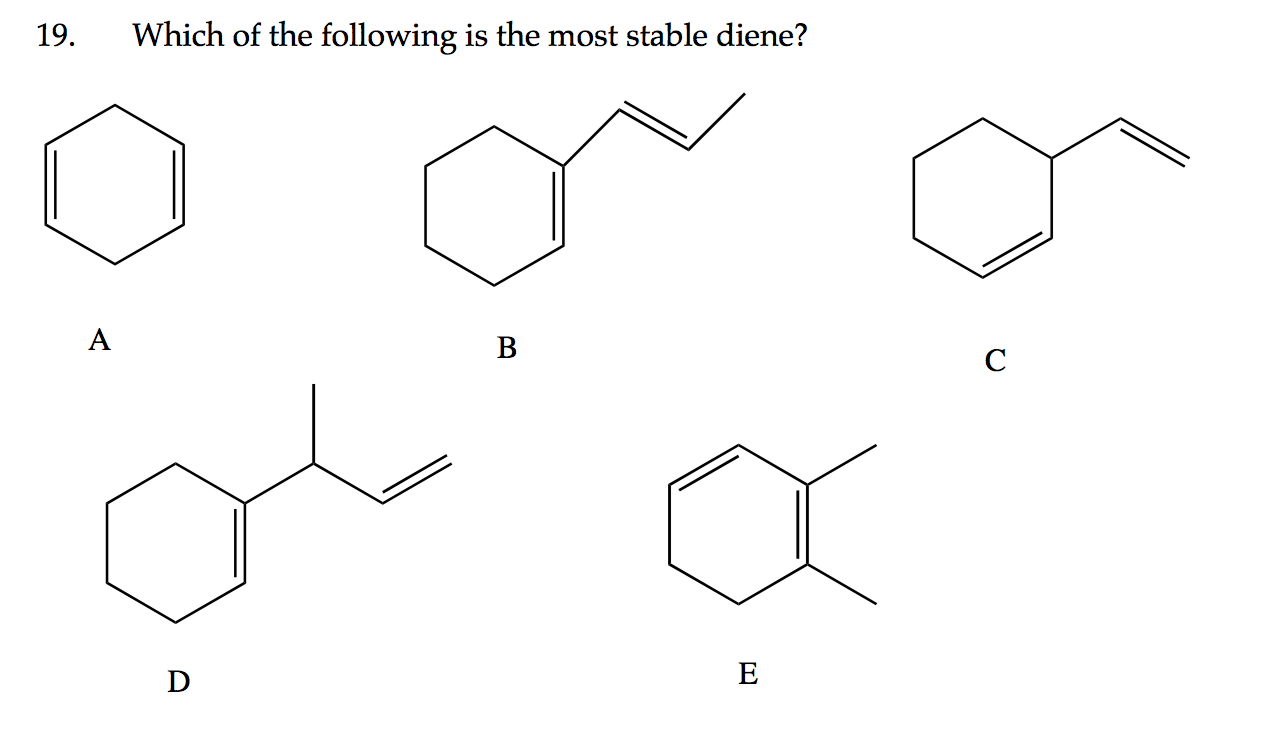
Think about it like this: You’re building with LEGOs. If you have two sets of colorful bricks (your double bonds) and you place them with just a single, plain brick in between, they look organized, right? They have a nice flow. But if you try to cram all the colorful bricks together, or scatter them too far apart, it just doesn't look as neat or feel as solid. The conjugated diene is the neatly built LEGO masterpiece!
This stability isn't just some abstract idea; it actually affects how these molecules behave. A more stable diene will be less reactive. It’s like a person who’s perfectly happy and content – they’re not looking for drama or a reason to change. They’re just… there, in their stable state. This makes them important in many chemical reactions, where chemists want to control what happens and when.

For example, when scientists are trying to build complex molecules, they often use dienes. Knowing which diene is the most stable helps them predict how the molecule will act and how easily it can be manipulated. It’s like having a reliable tool in your toolbox – you know it’s going to do its job without any unexpected surprises.
So, when you're faced with the question of which diene is the most stable, keep your eyes peeled for that sweet, sweet conjugation. The one with the double bonds dancing with a single bond in between is usually the star of the show. It’s the one that’s mastered the art of chemical chill, thanks to its electrons spreading out and finding their happy place. It's a little piece of molecular harmony, and that's pretty darn special!
"The magic of conjugation is what truly sets the most stable dienes apart. It's all about that perfect spacing!"
Isn't it fascinating how a tiny detail like the spacing of double bonds can make such a big difference? It’s these little molecular secrets that make chemistry so endlessly interesting. So next time you hear about a diene, remember the power of perfect spacing and the incredible stability it brings. It’s a testament to how elegant and efficient nature can be, even at the molecular level.
