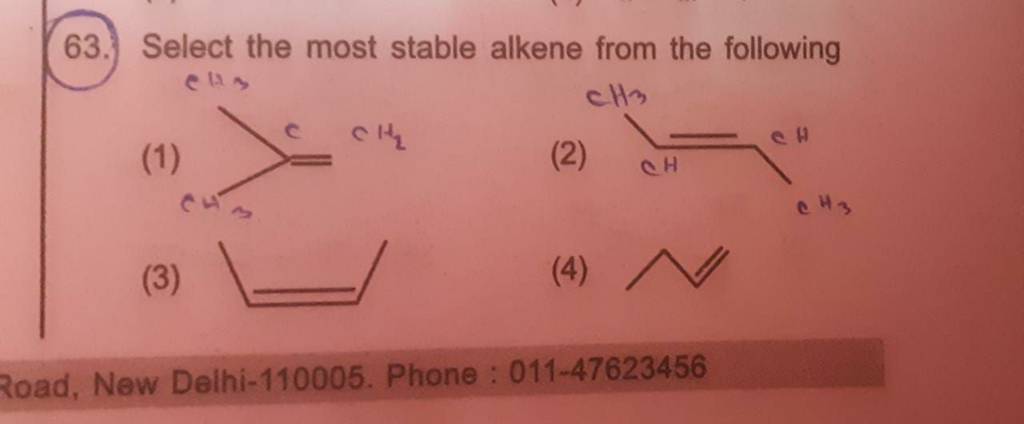
Which Of The Following Is The Most Stable Alkene
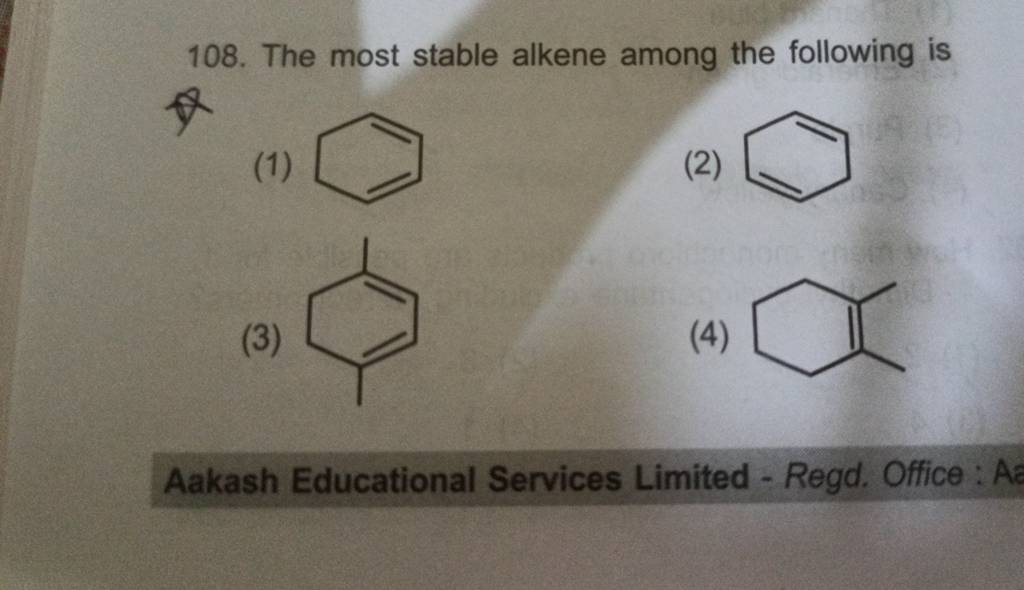
Ever wondered about the secret lives of molecules? Specifically, the ones that are super chill and don't get ruffled easily? Today, we're diving into the fascinating world of alkenes! Think of them as little chemical rockstars, and we're about to crown the ultimate king of stability.
So, what exactly is an alkene? It's a type of molecule that has a special "double bond" between two carbon atoms. This double bond is like a fancy handshake in the molecular world, and it gives alkenes their unique personality. Some are a bit flighty, while others are built to last.
We're going to look at a few contenders. Imagine them lining up for a "Most Stable Alkene" competition. There's a whole lineup of amazing molecules, each with its own strengths. But only one can take home the gold medal!
Let's meet our contestants! We have ethene, the simplest alkene, like the little starter car of the bunch. Then there's propene, a step up, a bit more robust. And the plot thickens with butene isomers, like twins with slightly different vibes.
But the real stars of our show are the bigger, bolder alkenes. Think of molecules with more carbon atoms involved. The more "friends" the double bond has supporting it, the happier and more relaxed it tends to be. It’s like a party where everyone is having a good time!
One of the most exciting aspects is how chemists figure all this out. They have special tools and experiments that reveal which molecules are as sturdy as a brick house and which ones might crumble under pressure. It's like a scientific detective story, and we get to peek behind the scenes.
The key to our alkene champion lies in something called "substitution". Don't let the fancy word scare you! It just means that other atoms or groups are attached to the carbons that are part of the double bond. The more "stuff" hanging off these carbons, the more stable the alkene.
Think of it like building with LEGOs. A single brick is okay, but adding more bricks around it makes the whole structure stronger. Similarly, when more groups are attached to the double bond carbons, the double bond becomes more secure and less likely to get into trouble.
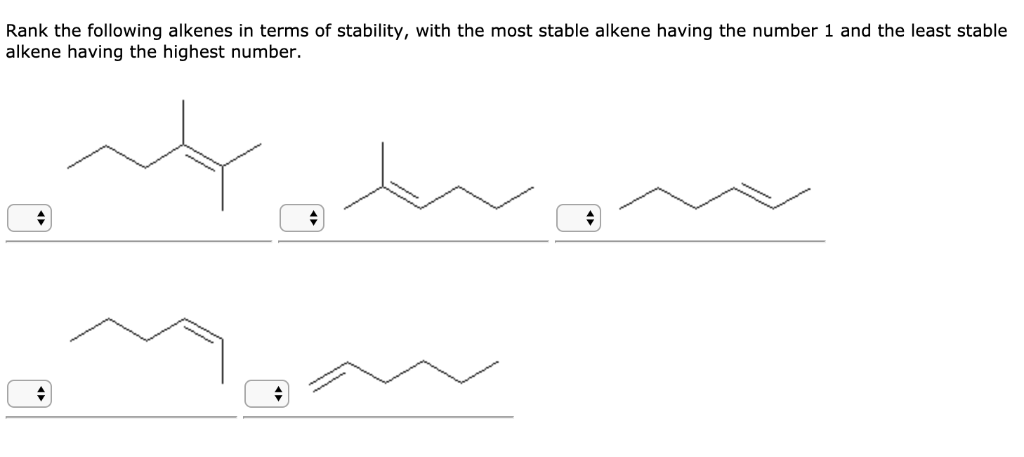
So, when we ask, "Which of the following is the most stable alkene?", we're essentially looking for the alkene with the most friends hanging out with its double bond. It's a friendly competition, but there's definitely a winner!
Let’s get a little more specific without getting too technical. Imagine we have a list of options. We’d be looking at the number of alkyl groups attached to the double bond. Alkyl groups are just fancy terms for chains of carbon and hydrogen atoms.
For example, ethene has no alkyl groups attached to its double bond carbons. It’s like a single, unadorned line. It’s the baseline, the starting point of our stability journey.
Propene has one alkyl group attached. It’s got a little bit more going on, a small addition to its structure. This makes it a tiny bit more stable than ethene. We’re seeing progress!
Then we have the butenes. These come in different flavors! Some have one alkyl group, and some have two alkyl groups attached to the double bond carbons. The ones with two groups are definitely feeling more solid.
But the real champions, the ones that truly shine in the stability contest, are the alkenes with three and even four alkyl groups attached to the double bond carbons. These are the superheroes of the alkene world!
Let's call the alkene with three alkyl groups "trisubstituted". It’s like a well-attended party around the double bond. Lots of support, lots of stability. It’s feeling pretty good about itself.
And the ultimate winner, the undisputed champion, is the "tetrasubstituted" alkene. This is where it's at! Imagine four groups all cozying up to the double bond carbons. It’s a molecular fortress, incredibly stable and unflustered by most things.
So, if you were presented with a list of alkenes, and you saw one with the most alkyl groups attached to its double bond, that’s your winner! It’s the most stable of the bunch. It’s the one that’s going to weather any chemical storm.
Why is this so cool? Because these stability differences matter! They affect how molecules react, how they behave in chemical reactions, and even how they're used in everyday products. It's like knowing which of your friends is the most reliable – you can count on them!
This whole concept of alkene stability is a fundamental building block in understanding organic chemistry. It's like learning your ABCs before you can write a novel. And this particular concept is one of the first exciting discoveries that gets students hooked.
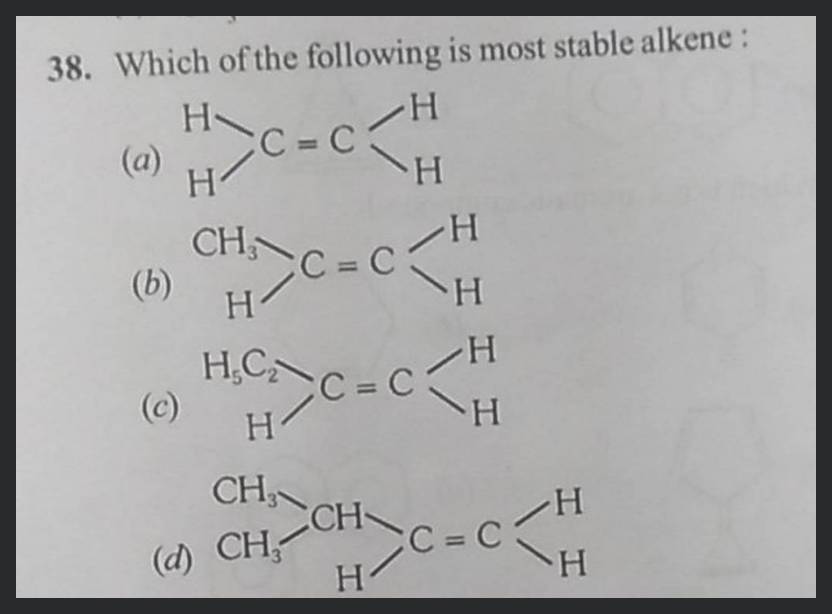
It’s a little like picking your favorite ice cream flavor. Some are good, some are better, but there’s usually one that just hits the spot perfectly. In the world of alkenes, the "perfect hit" for stability comes from those well-supported double bonds.
You might be thinking, "But how do they know?" Scientists use clever techniques. One way is by measuring the heat released when an alkene is hydrogenated (which means adding hydrogen to break the double bond). The more stable the alkene, the less heat is released. It’s like comparing how much energy it takes to break down something solid versus something flimsy.
Think of it like this: a truly stable alkene is like a well-built sandcastle that can withstand a gentle tide. A less stable alkene might be like a pile of loose sand that gets washed away easily. We're looking for the sandcastle that stands tall!
The most stable alkene is generally the one with the most substituents on the double bond carbons. It’s a simple rule with huge implications!
So, when you encounter a question like "Which of the following is the most stable alkene?", you now have the secret weapon! Just count those attached groups. More groups mean more stability. It’s as straightforward as that!
It’s a bit of a treasure hunt in the molecular world. You're hunting for the alkene that has the most "bling" around its double bond, in terms of those helpful alkyl groups. The more bling, the more stable it is. Simple, right?
This principle pops up in so many areas of chemistry. From designing new medicines to creating advanced materials, understanding stability is crucial. It's the foundation upon which many scientific breakthroughs are built.
It's exciting to think that a simple concept like "more is better" when it comes to attached groups can unlock so much understanding. It’s a testament to the elegant simplicity that can exist in complex science.
So, next time you hear about alkenes, you can impress your friends with your newfound knowledge. You can be the alkene guru, pointing out the most stable ones with confidence. It’s a fun party trick, chemical-style!
Remember, it's all about that double bond and its entourage. The bigger and more supportive the entourage, the more chill and stable the alkene. It’s the ultimate chemical popularity contest!
And that, my friends, is the delightful secret behind identifying the most stable alkene. It’s not just about memorizing facts; it’s about understanding the "why" behind the "what". And the "why" here is a beautiful dance of electron clouds and supportive groups.
So go forth and conquer the world of alkene stability! You're now equipped to identify the rockstars of the alkene universe. They’re the ones with the most friends, the most support, and the most unshakeable composure. They are, truly, the most stable alkenes!
