Which Of The Following Is The Correct Model Of C7h16
Ever found yourself staring at a string of letters and numbers, like C7H16, and wondered what on earth it represents? It’s a question that might pop up in a chemistry class, or perhaps when you're casually scrolling through something online and stumble upon it. Far from being a dull academic puzzle, understanding these molecular formulas is actually pretty fun and surprisingly relevant to our everyday lives. Think of it as a secret code that unlocks the building blocks of everything around us, from the air we breathe to the food we eat and the fuel that powers our cars.
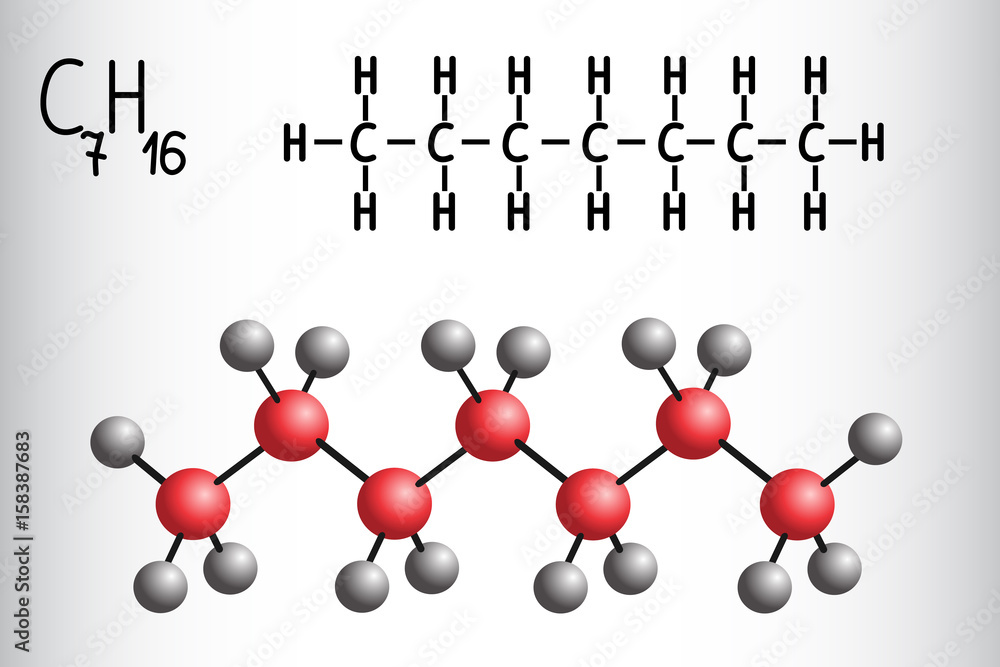
So, what is C7H16 all about? This particular formula belongs to a group of molecules called alkanes, which are simple hydrocarbons – meaning they are made up only of carbon (C) and hydrogen (H) atoms. The '7' tells us there are seven carbon atoms, and the '16' tells us there are sixteen hydrogen atoms. But here's where it gets interesting: the same formula, C7H16, can represent several different molecules! This phenomenon is called isomerism. Imagine having the same set of LEGO bricks, but being able to build different structures with them. That’s essentially what isomers are. These different structures, or models, of C7H16 have distinct shapes and properties, even though they share the same chemical formula. The question, "Which of the following is the correct model of C7H16?" implies that there isn't just one single correct representation, but rather multiple possibilities that fit the formula.
The purpose of understanding these different models is to accurately predict and explain the behavior of chemical substances. Knowing whether you’re dealing with heptane (a straight chain of carbons) or one of its branched isomers (like 2-methylhexane or 3-methylhexane) can make a huge difference in understanding things like boiling point, flammability, and how it interacts with other chemicals. For instance, different isomers of octane (another alkane, C8H18) have varying performance characteristics as fuel in our cars. Octane rating, something we often see at the gas pump, is directly related to the properties of these different isomers!
In education, exploring isomers like those of C7H16 is a fundamental part of learning organic chemistry. It teaches students about molecular structure, spatial arrangement of atoms, and how subtle differences in shape can lead to significant differences in function. In daily life, even if you’re not actively identifying isomers, you’re benefiting from the science that understands them. From the plastics in your gadgets to the medicines that keep us healthy, the precise structure of molecules matters immensely. So, the next time you see a chemical formula, remember it's just the beginning of a fascinating story about how matter is put together.
Want to explore this a bit yourself? It’s easier than you might think! You can find online molecular modeling kits that allow you to build and manipulate 3D representations of molecules. Just search for "build C7H16 isomers online" and you'll find interactive tools. You can also look up images of the different heptane isomers to see their distinct shapes. It's a visual and interactive way to grasp the concept of isomerism. Think of it as a digital puzzle, where you're figuring out how to arrange those seven carbon atoms and sixteen hydrogen atoms in all the possible ways!
