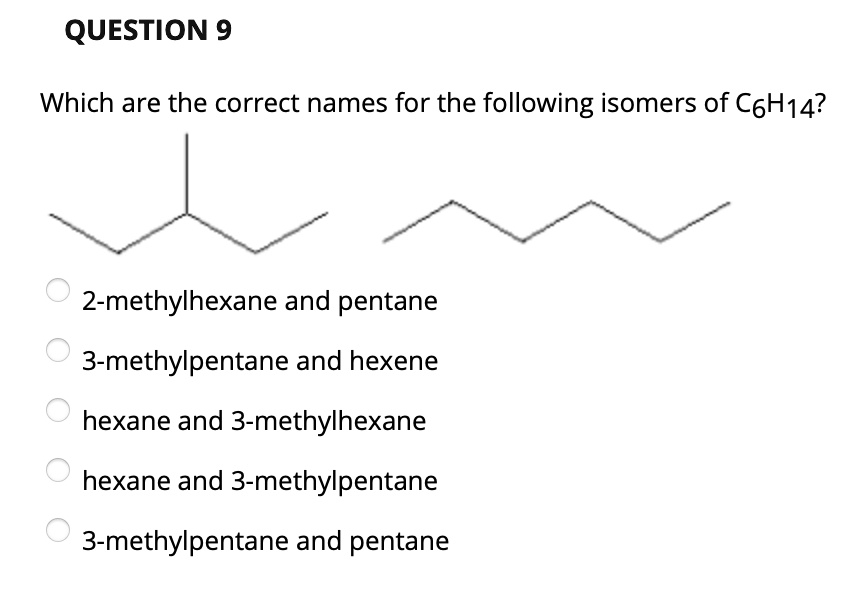
Which Of The Following Is The Correct Model Of C6h14
Hey there, curious minds! Ever find yourself staring at a string of letters and numbers like "C6H14" and wondering, "What in the world is that?" It looks a bit like a secret code, right? Well, today, we're going to decode one of these chemical mysteries together, specifically the one that goes by C6H14. You know, the one that's got chemists scratching their heads and asking: Which of the following is the correct model of C6H14? Don't worry, no pop quiz here, just a chill exploration into the fascinating world of molecular shapes!
So, C6H14. What does that even mean? Think of it like a recipe. The 'C' stands for carbon atoms, and the 'H' stands for hydrogen atoms. The little numbers? Those tell us how many of each atom we've got. So, C6H14 means we have 6 carbon atoms and 14 hydrogen atoms. Easy peasy so far, right? It's like saying you've got six big building blocks (carbon) and fourteen smaller ones (hydrogen) to work with.
But here's where it gets really interesting. Just because we have the same ingredients, does it mean we'll always end up with the same cake? Nope! In the world of chemistry, having the same number and type of atoms doesn't automatically mean you have the same molecule. This is where the concept of isomers comes in, and it's like having different LEGO creations made from the exact same set of bricks. Mind-bending, isn't it?
Imagine you have six carbon atoms and fourteen hydrogen atoms. You could, in theory, connect them in a straight line. Like a tiny, molecular train. Or, you could start bending that train, making it a bit wiggly. Maybe you could even branch off from the main line, creating little side trips. Each of these different arrangements, these different ways of connecting our carbon and hydrogen pals, results in a different molecule, even though they all have that same C6H14 recipe.
And that's precisely why the question "Which of the following is the correct model of C6H14?" is so crucial. It implies there isn't just one way to arrange those atoms. There are actually multiple possibilities, and each one is a unique molecule with its own unique properties. It's like having a whole family of molecules all sharing the same basic DNA, but each one looks and acts a little differently.

So, what are these different C6H14 shapes like?
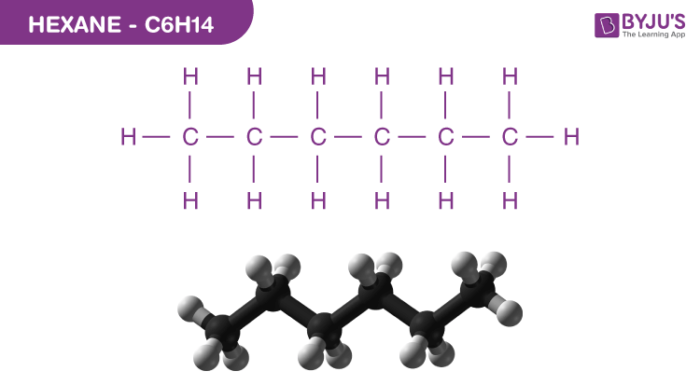
Let's dive into some of the cooler possibilities. The simplest way to arrange six carbons in a chain is just that: a straight, unbroken line. This molecule is called hexane. Think of it as the most straightforward, no-fuss version of C6H14. It’s like a perfectly straight road. This is often the first one people think of when they hear C6H14, and for good reason – it’s the foundational structure.
But what if we take that six-carbon chain and start bending it? We could make it a bit shorter and then attach a carbon atom to a side branch. This leads us to molecules like 2-methylpentane. Now, instead of a straight road, we have a main road with a little exit ramp. The carbon atoms are still all connected, but the arrangement is different. It's like taking that train and adding a caboose that’s attached a little further up the line. Still the same number of cars, but the overall shape is altered.
We can get even more creative! We could have a shorter main chain and more branches. Consider 3-methylpentane. This is like having a main road and a side street that's a bit longer. The atoms are still holding hands, just in a different dance formation. It’s like a slightly different branching pattern on a tree – still a tree, but with a unique silhouette.

And it doesn't stop there! We can have even more compact structures. What about 2,2-dimethylbutane? This one’s like a small, sturdy little cluster. We’ve got a four-carbon chain, and then two carbon branches coming off the second carbon. Imagine a central hub with two spokes. It’s a much more branched and compact arrangement compared to the straight hexane.
There's also 2,3-dimethylbutane. This one's like a symmetrical little structure, with two branches coming off adjacent carbons on a four-carbon chain. Think of a short main road with two side roads that are close together. It’s another variation on the branched theme, showcasing how subtle changes in connections lead to different molecular personalities.

So, you see, C6H14 isn't just one thing. It's a family! These different arrangements are called constitutional isomers, which is a fancy way of saying they have the same molecular formula but a different connectivity of atoms. It’s like how a sedan and a truck are both "cars" in a broad sense, but they have very different designs and functions. Hexane and 2,2-dimethylbutane are both C6H14, but they’re not interchangeable. They have different boiling points, different melting points, and can even behave differently in chemical reactions.
Why is this so cool?
Well, for starters, it highlights the incredible diversity that can arise from a simple set of building blocks. It's a testament to the intricate dance of atoms and bonds that make up our world. Think about it: just by rearranging the same pieces, we can get molecules that might be used as fuels, solvents, or even as parts of more complex biological molecules.
This understanding is absolutely vital in chemistry. When scientists are trying to synthesize new medicines, design better fuels, or understand how life works, they need to know exactly which molecule they're dealing with. If you're trying to build a specific LEGO spaceship, you need the right instructions and the right arrangement of bricks, not just a pile of them. The same goes for chemistry!
So, the next time you see something like C6H14, don't just see a jumble of letters and numbers. See the potential! See the possibilities! It's a little puzzle, a molecular riddle, and understanding the different ways these atoms can connect is what makes chemistry so endlessly fascinating. It's like being a detective, figuring out the unique identity of each molecule based on its structure. Pretty neat, huh?
The "correct model" of C6H14 depends entirely on which specific isomer you're talking about. Each of those named molecules (hexane, 2-methylpentane, etc.) is a correct model for a specific type of C6H14. Without more information, you can't just pick one. It’s like asking, "Which of these drawings is the correct drawing of a dog?" Well, it depends on whether you're talking about a poodle, a bulldog, or a chihuahua! Each is a correct representation of a dog, but they look very different.
So, the next time you encounter C6H14, remember it's not a single entity, but a family. And exploring that family tree is a fantastic way to appreciate the subtle yet significant beauty of molecular structure. Keep that curiosity alive, folks!
