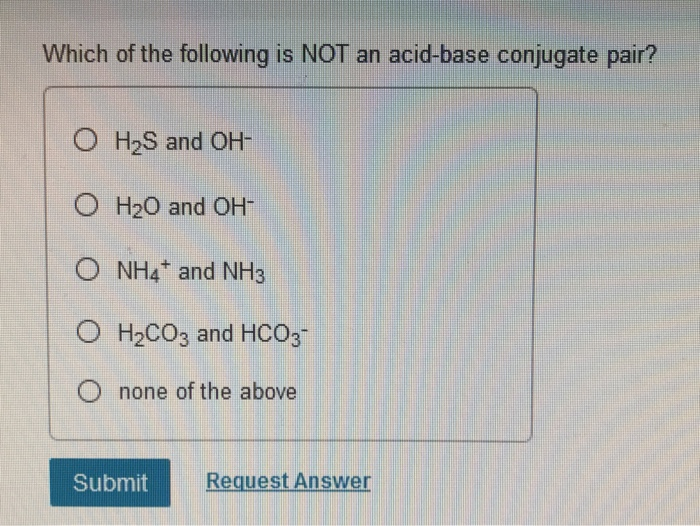
Which Of The Following Is Not An Acid-base Conjugate Pair
Hey there, science adventurers! Ever found yourself staring at a list of chemical critters and wondering, "Who's related to whom in this whole acid-base family reunion?" Well, get ready to have your mind tickled, because we're about to dive headfirst into the wonderfully wacky world of acid-base conjugate pairs. Think of it like a super-powered game of "Who's Your Daddy?" but with molecules!
So, what in the fizzy-pop heck is an acid-base conjugate pair, you ask? Imagine a molecule that's a bit of a drama queen. It loves to donate a proton – that's just a fancy word for a tiny little hydrogen ion, like a mini hydrogen superhero with a positive charge. When it does this, it transforms into its conjugate base. This conjugate base is like the molecule's chill, laid-back twin, just hanging out, ready to accept a proton back if the mood strikes. Conversely, a molecule that's more of a borrower, eager to snag a proton, is an acid. When it grabs that proton, it becomes its conjugate acid. It's like the proton-borrowing molecule suddenly grew a hydrogen accessory!
Let's get down to the nitty-gritty, the juicy stuff, the moment of truth! We're going to look at some contenders, some pairs that look like they're related, but are they truly blood brothers and sisters in the acid-base realm? It’s like trying to spot a fake designer handbag at a flea market – some things just don't add up!
Picture this: You're at a party, and you see two people who look suspiciously alike. They have the same nose, the same slightly quirky smile. You might think, "Aha! Siblings!" But then you hear one of them talk about their extensive stamp collection, while the other only discusses their passion for extreme unicycling, and you realize, "Hmm, maybe not so much." That's precisely what we're doing here, but with chemicals. We're looking for the imposter, the one that doesn't quite fit the family resemblance.
So, let's meet some of our potential acid-base pairs. We’ve got the classic dynamic duo, HCl and Cl-. Now, HCl, hydrochloric acid, is a total proton donor, a real show-off. When it throws that proton away, it becomes Cl-, the chloride ion. And guess what? If Cl- gets a proton back, it becomes HCl again! They're like a perfectly matched set, a yin and yang of proton transfer. Definitely a real conjugate pair, no doubt about it!

Then we have the bubbly personality of H2O, water. Water is a real chameleon, you know? It can be an acid and a base. When it acts as an acid, it donates a proton and becomes OH-, the hydroxide ion. So, H2O and OH-? Yep, that's a valid acid-base conjugate pair. But here’s the fun part: water can also be a base! It can accept a proton. When it does that, it becomes H3O+, the hydronium ion. So, H3O+ and H2O? Bingo! Another solid conjugate pair. Water is basically the cool aunt who can do it all!
Now, let's spice things up a little. Consider the mighty NH3, ammonia. Ammonia is a bit of a proton magnet, a real sponge for those little hydrogen heroes. When it grabs a proton, it transforms into NH4+, the ammonium ion. So, NH3 and NH4+? Absolutely! They're a perfectly formed conjugate pair, like two peas in a pod that just happen to carry a charge.

But what about when things get a little...weird? What if we throw in a pair that looks like it should be related, but just isn't? Imagine seeing someone who looks exactly like your best friend's cousin, but then you find out they’re actually a distant acquaintance from a summer camp they attended only once. The resemblance is there, the superficial connection, but the deep, meaningful, proton-swapping relationship is missing.
So, we’re on a quest. We're sifting through the options, looking for the one that’s not an acid-base conjugate pair. It’s the outlier, the odd one out, the one that makes you scratch your head and say, "Wait a minute... something's not right here!" It’s the chemical equivalent of a mismatched sock in a perfectly folded drawer. And when you find it, oh, the satisfaction! It's like solving a mini mystery, a little victory for your curious mind.

Think of it as a puzzle. You've got all these pieces, and they almost fit. But there's one piece that’s just a bit too big, or a bit too small, or has the wrong shape entirely. That’s the one we’re hunting for. It’s the challenge that makes the game fun, the moment of realization that makes your brain do a little happy dance. So, keep your eyes peeled, your chemistry brains switched on, and get ready to spot that imposter pair!
The key to spotting a conjugate pair is a difference of exactly one proton (H+). If two species differ by more than that, or by something completely different, they’re probably not a pair!
So, as you ponder the fascinating relationships between these chemical entities, remember to have fun with it. Chemistry isn't just about memorizing formulas; it's about understanding the intricate dance of molecules, the ebb and flow of protons, and the delightful connections that make up our chemical universe. And when you correctly identify the pair that's not an acid-base conjugate pair, give yourself a pat on the back. You've just aced a crucial step in understanding the electrifying world of acid-base chemistry!
