Which Of The Following Is Not A Conjugate Acid-base Pair
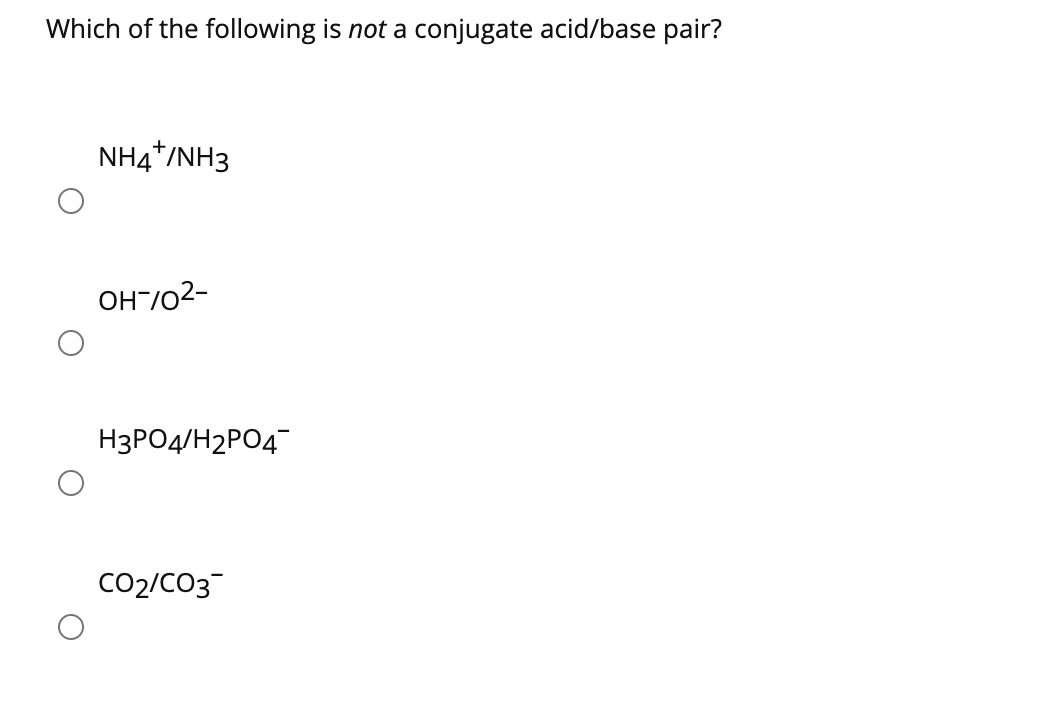
Hey there, science enthusiasts and curious cats! Ever wondered what makes a good acid and its trusty sidekick, the base, hang out together? Today, we're diving into the wonderfully wacky world of conjugate acid-base pairs. Think of it like a superhero duo, always there for each other. But sometimes, in the grand scheme of chemistry, a pairing just doesn't make the cut. And that's where our little game comes in: Which of the following is not a conjugate acid-base pair?
Now, before you start picturing caped crusaders of pH, let's get a little cozy with the concept. An acid, in simple terms, is a proton (that's a hydrogen ion, H+) donor. A base is a proton acceptor. Easy peasy, right? Like a game of chemical tag, passing protons back and forth.
When an acid loses its proton, it becomes its conjugate base. And when a base accepts a proton, it becomes its conjugate acid. They are essentially the same molecule, just with or without that little H+ buddy. They're like twins who occasionally borrow each other's hats.
So, why is this whole conjugate thing so cool? Because it helps us understand how reactions happen, how things dissolve, and why your stomach might rumble after a spicy meal (hello, hydrochloric acid!). It's the secret handshake of the chemical world.
But here's the juicy bit, the part that makes our game exciting. Not every acid and base can form a valid conjugate pair. It's like trying to match socks from different laundry baskets – sometimes it just doesn't fit. And that's what we're here to uncover!
Let's imagine we're presented with a few options. We'll call them "Suspect A," "Suspect B," "Suspect C," and so on. Your mission, should you choose to accept it, is to spot the imposter. The one that doesn't belong.
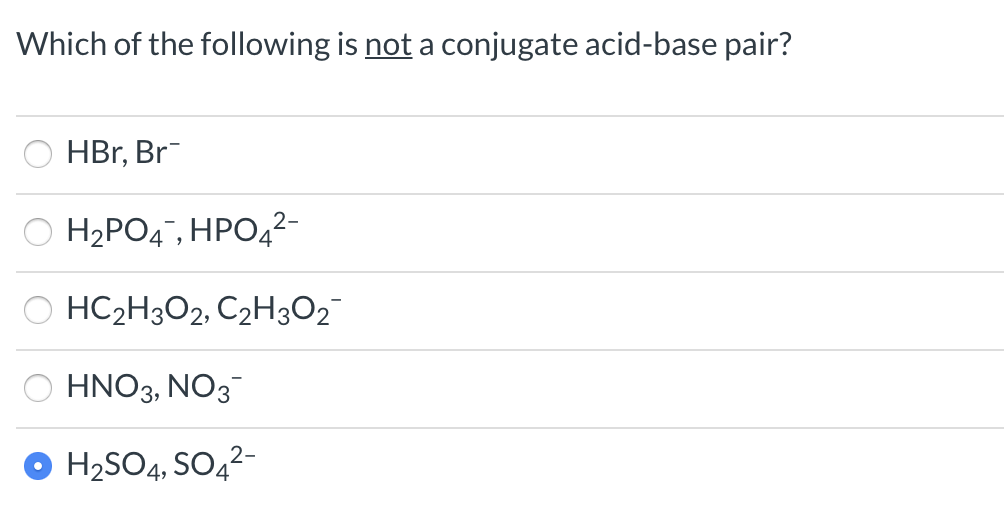
Think of the chemical formulas like the suspects' fingerprints. For a pair to be considered conjugate, their formulas should differ by exactly one hydrogen ion (H+). That's the golden rule. If the difference is more than one proton, or no protons at all, then that pair is definitely not a conjugate pair.
Let's get our hands dirty with some examples, shall we? Imagine you see:
Example 1: H₂SO₄ and HSO₄⁻
Take a look at these two. What's the difference? One hydrogen atom. And guess what? The charge also changes by one (from neutral to -1). This is a classic example of a conjugate acid-base pair! Sulfuric acid (H₂SO₄) is the acid, and when it gives up a proton, it becomes the hydrogen sulfate ion (HSO₄⁻), its conjugate base. Pretty neat, huh?

Example 2: NH₃ and NH₄⁺
Now, let's check out ammonia (NH₃) and the ammonium ion (NH₄⁺). What do you notice? The ammonium ion has one more hydrogen than ammonia. And again, the charge shifts (from neutral to +1). Ammonia acts as a base, grabbing a proton to become the ammonium ion, its conjugate acid. These two are also best buds in the conjugate world.
Example 3: H₂O and OH⁻
Water (H₂O) and the hydroxide ion (OH⁻). See the pattern? Water can act as an acid, losing a proton to become the hydroxide ion. So, H₂O is the acid, and OH⁻ is its conjugate base. It's like water doing a little magic trick!
But what if we threw in something a bit… off? Something that just doesn't fit the mold?
Example 4: HCl and Cl⁻
Hydrochloric acid (HCl) and the chloride ion (Cl⁻). This is another slam dunk! HCl is a strong acid. It readily donates its proton, leaving behind the chloride ion. Bingo! They differ by exactly one H+. Conjugate pair, no doubt.

Example 5: NaOH and Na⁺
Here's where things get interesting. We have sodium hydroxide (NaOH) and the sodium ion (Na⁺). Now, let's think about what happens when NaOH acts as a base. It dissociates, meaning it splits into its ions: Na⁺ and OH⁻. So, while OH⁻ is the conjugate base of water, NaOH as a whole molecule isn't directly forming a conjugate pair with just Na⁺ by donating or accepting a proton.
The key is that difference of one proton. NaOH contains Na⁺ and OH⁻. The OH⁻ part is the actual conjugate base. Na⁺ is just a spectator ion in this context. It doesn't participate in the proton exchange that defines a conjugate pair. It's like the bodyguard who's always there but doesn't actually fight the main battle.
So, if you were presented with a list of options, and one of them was something like NaOH and Na⁺, that would be your prime suspect for not being a conjugate acid-base pair. The other pair would have to differ by exactly one H+.

Isn't that fun? It's like a chemical scavenger hunt, looking for that one perfect match. The beauty of chemistry is in these subtle relationships, these proton-sharing friendships.
Why does this matter? Well, understanding conjugate pairs helps us predict the outcome of acid-base reactions. It's fundamental to buffering systems (like the ones in your blood keeping you from getting too acidic or too basic – super important!), and even in understanding how certain medicines work.
So next time you're faced with a chemistry problem involving acids and bases, remember our little game. Look for that one proton difference. It's your secret weapon to spotting the odd one out!
And hey, if you ever feel like impressing your friends at a party, just casually drop a fact about conjugate acid-base pairs. You might just become the life of the chem-themed soirée! Keep exploring, keep questioning, and most importantly, keep having fun with science!
