Which Of The Following Is An Isotope Of Lithium Labster
Ever wondered how scientists peek into the microscopic world, unraveling the secrets of atoms and molecules without a super-powered microscope? Well, it turns out there's a seriously cool way to do just that, and it involves playing around with the building blocks of matter itself! We're talking about isotopes, and they're not just for fancy lab coats and complex equations. They're everywhere, from the food we eat to the medical scans that keep us healthy. Today, we're diving into the fascinating world of lithium isotopes, and specifically, how a tool called Labster helps us explore them in a way that's both fun and incredibly informative.
Why Isotopes Are Such a Big Deal (and So Much Fun!)
So, what's the buzz about isotopes? Think of them as siblings from the same atomic family. They’re the same element, meaning they have the same number of protons in their core, but they have a different number of neutrons. This subtle difference can lead to some pretty significant changes in their properties, making them incredibly useful for all sorts of applications. For instance, some isotopes are used in medicine to diagnose diseases or even treat certain cancers. Others are used to date ancient artifacts, helping us understand history like never before. And then there are isotopes that play a crucial role in understanding nuclear energy and environmental science. The possibilities are endless, and that’s what makes them so exciting to explore!
Now, imagine being able to experiment with these fascinating particles without ever having to step foot in a real-life laboratory. That's where Labster comes in. Labster is an online platform that offers incredibly realistic virtual laboratory simulations. They’ve created a world where you can conduct experiments, learn scientific concepts, and explore complex topics in a safe, engaging, and incredibly interactive way. It's like having a virtual science playground at your fingertips!
Unlocking the Secrets of Lithium Isotopes with Labster
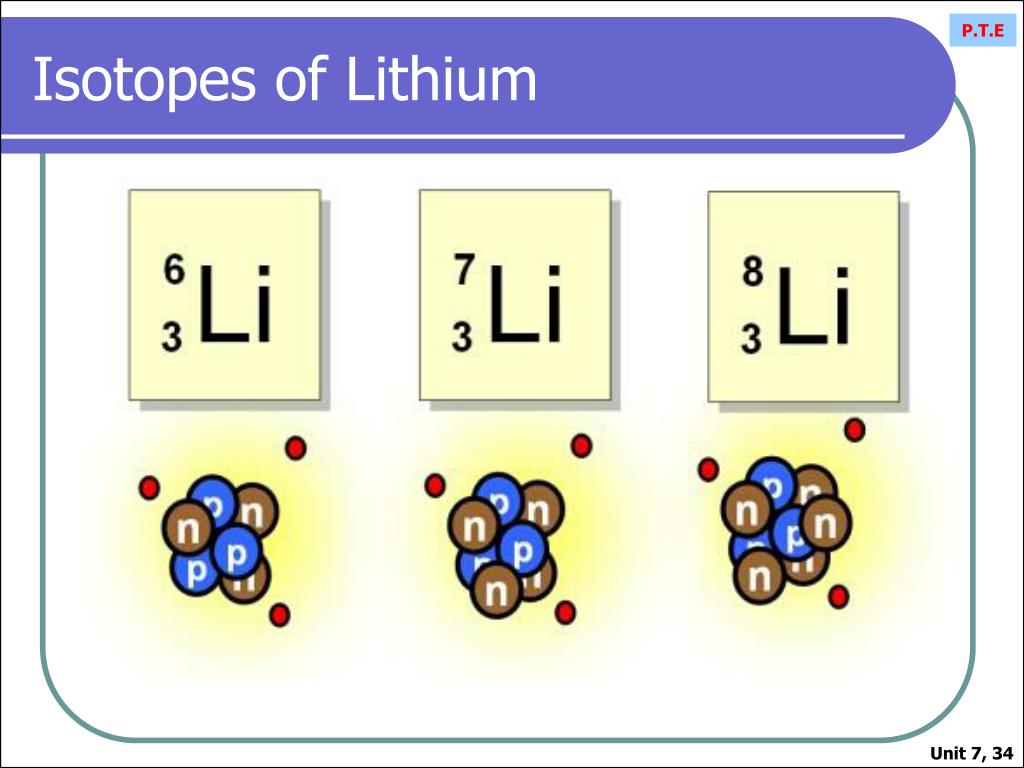
Lithium, that light and reactive alkali metal, has its own cast of isotopic siblings. The most common ones are Lithium-6 and Lithium-7. While they both share the same number of protons (three, to be exact!), Lithium-6 has three neutrons in its nucleus, while Lithium-7 has four. This slight difference in neutron count affects their mass and how they behave in certain reactions. Understanding these differences is key to unlocking their potential.
The Labster simulation focusing on lithium isotopes isn't just about memorizing numbers; it's about hands-on (virtual!) exploration. You get to step into a virtual lab environment and conduct experiments that demonstrate the unique properties of each lithium isotope. You might be tasked with separating them, observing their reactions, or even using them in simulated applications. The beauty of Labster is that it allows you to make mistakes, learn from them, and try again without any real-world consequences. It fosters a deeper understanding because you're not just reading about it; you're doing it.

“Labster makes learning about complex scientific concepts like isotopes accessible and genuinely fun. It’s like being in a real lab, but with unlimited retries!”
The benefits of using a platform like Labster for learning about isotopes are numerous. Firstly, it democratizes science education. Not everyone has access to expensive laboratory equipment or the opportunity to participate in hands-on experiments. Labster breaks down these barriers, making advanced scientific learning available to a much wider audience. Secondly, it enhances engagement. The interactive nature of the simulations captures your attention and makes learning feel less like a chore and more like an adventure. You’re actively participating in the discovery process, which leads to better retention and a more profound understanding.

Moreover, Labster’s simulations are designed with pedagogical principles in mind. They guide you through experiments step-by-step, provide immediate feedback, and offer explanations for the phenomena you observe. This is crucial when dealing with concepts like isotopes, where the nuances can be tricky to grasp. By experiencing these concepts in a virtual environment, you can visualize abstract ideas and build a solid foundation for further scientific study.
So, when you encounter questions about which of the following is an isotope of lithium, and you've had the chance to virtually experiment with them on Labster, you'll be armed with a much more intuitive understanding. You’ll know that it’s not just a theoretical concept, but something you’ve actively explored and understood. It’s this kind of immersive, engaging learning that makes topics like isotopes not just important, but genuinely exciting!
