Which Of The Following Is A Secondary Alkyl Halide
Have you ever wondered about the invisible building blocks that make up everything around us? Chemistry, at its heart, is like a giant puzzle, and understanding how different molecules are put together helps us unlock the secrets of the universe. One little piece of that puzzle, which might sound a bit technical but is surprisingly interesting, is the concept of a secondary alkyl halide. It's not just a term for a chemistry textbook; it’s a fundamental idea that helps us understand how certain substances behave and interact.
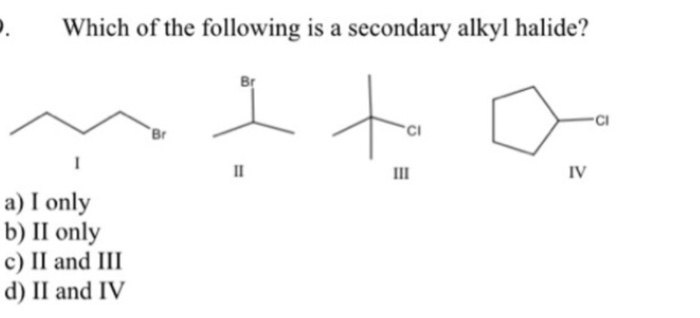
So, what exactly is a secondary alkyl halide, and why should you care? In the simplest terms, an alkyl halide is a molecule where a halogen atom (like chlorine, bromine, or iodine) is attached to a chain of carbon atoms (an "alkyl" group). The "secondary" part tells us where that halogen is attached. Imagine a chain of carbon atoms like a train. A primary alkyl halide has the halogen attached to the very first or last car of the train. A tertiary has it attached to a car with three other cars connected to it. A secondary alkyl halide is like the halogen being on one of the middle cars, specifically one that's connected to exactly two other carbon atoms. This seemingly small detail has a big impact on how the molecule reacts and what it can be used for.
The purpose of understanding these classifications, including secondary alkyl halides, lies in their reactivity. This is the key benefit! Knowing whether a molecule is primary, secondary, or tertiary helps chemists predict how it will behave in chemical reactions. This predictability is incredibly valuable. For instance, secondary alkyl halides often participate in specific types of reactions, like substitution and elimination, which are crucial for building more complex molecules. Think of it like knowing how a certain type of Lego brick will connect to others – it allows you to design and construct more intricate structures.
Where might you encounter the principles behind secondary alkyl halides in everyday life or education? While you won't be pointing out a secondary alkyl halide in your morning coffee, the underlying chemistry is at play in many areas. Many pharmaceuticals are synthesized using reactions involving alkyl halides. The development of new medicines relies heavily on understanding how to precisely manipulate molecules, and alkyl halides are common starting materials or intermediates. In educational settings, learning about alkyl halides is a cornerstone of organic chemistry, building a foundation for more advanced studies in medicine, materials science, and environmental science. Even in the development of new plastics and polymers, the controlled reactions involving these types of molecules play a role.
If you're feeling a spark of curiosity and want to explore this a bit further without needing a full chemistry lab, you can do a few simple things. Look up visualizations of organic molecules online. Many websites and apps offer interactive 3D models where you can see carbon chains and halogen atoms. You can then practice identifying which carbon atom the halogen is attached to and count its neighboring carbon atoms. It's like playing a chemical spot-the-difference game! You can also search for common examples of reactions involving secondary alkyl halides, like the SN2 reaction (which tends to favor primary but can involve secondary) or elimination reactions. Seeing these reactions play out visually can make the concept much more concrete and, dare we say, fun.
