Which Of The Following Is A Bronsted Lowry Base
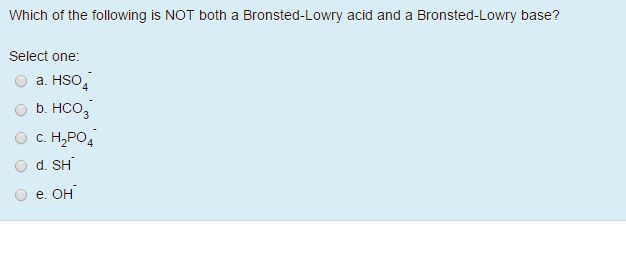
Hey there, science adventurers and curious minds! Get ready to dive headfirst into the wonderfully wacky world of acids and bases. Today, we're going on a quest to uncover a very special kind of base, one that plays by a particular set of rules. It's not just any base; it's a Bronsted-Lowry base, and trust me, it's way cooler than it sounds!
Imagine you're at a super-exclusive party, the kind where everyone has a secret handshake and a special role. In the world of chemistry, our acids and bases are having their own epic party. And our star of the show today, the Bronsted-Lowry base, has a very specific job at this party.
Think of it like this: some people at the party are great at giving away things. They’re the life of the party, handing out hugs or maybe even their last cookie. These are our acids!
But then, there are the folks who are super excited to receive things. They're the ones with open arms, ready to grab that hug or snag that cookie with a gleeful grin. These, my friends, are our Bronsted-Lowry bases!
Their main gig, their ultimate purpose in the chemical realm, is to be a proton acceptor. Yes, you heard that right – a proton acceptor! It's like they're collecting little hydrogen ions, these tiny positively charged particles, with all the enthusiasm of a kid collecting shiny marbles.
So, if you see a molecule or an ion that's just dying to grab a proton, that's your cue! You've likely found yourself a bona fide Bronsted-Lowry base. It's all about that eagerness, that "give it here!" attitude when it comes to protons.
Let's get a little more concrete, shall we? Imagine a molecule like ammonia. It's represented by the formula NH₃. This little guy is practically begging for a proton.
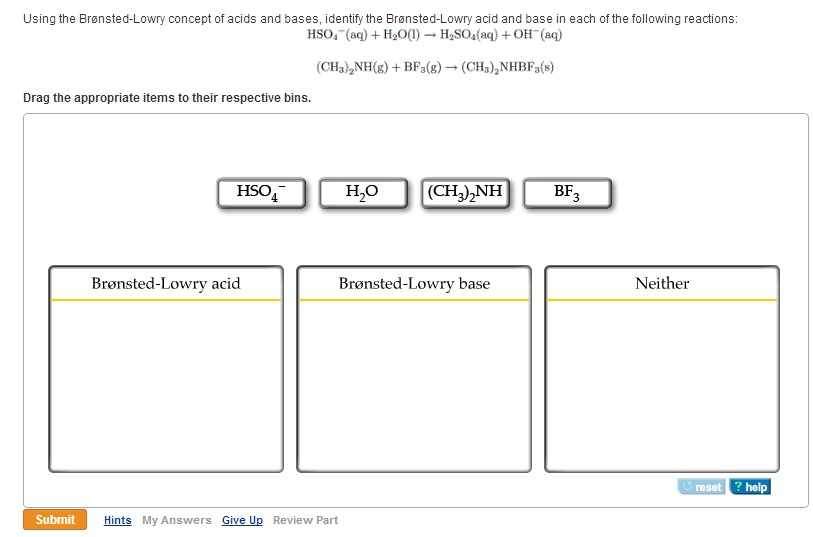
When ammonia encounters an acid, which, remember, is a proton donor, it's like a match made in chemical heaven. The acid is like, "Here, take this proton!" and ammonia swoops in, snatching it up with glee. Boom! You've got yourself a Bronsted-Lowry base in action.
Think of it like a game of "catch the proton." The acid throws it, and the Bronsted-Lowry base is the MVP of catching. They are the ultimate proton collectors, the champions of receiving that tiny but mighty particle.
It’s not just about being alkaline, though that's often a side effect. It’s specifically about that act of accepting a proton. Some substances might feel a bit slippery or taste bitter, like typical bases, but the Bronsted-Lowry definition hones in on this proton-grabbing superpower.
So, if you're presented with a list of chemical goodies and asked, "Which one of these is a Bronsted-Lowry base?", keep an eye out for those eager proton-acceptors. They are the ones with the electron pairs just waiting to bond with that incoming proton.
Consider hydroxide ions, OH⁻. These are also fantastic Bronsted-Lowry bases. They have a negative charge, which makes them super attractive to positive protons. It’s like a magnet for protons!
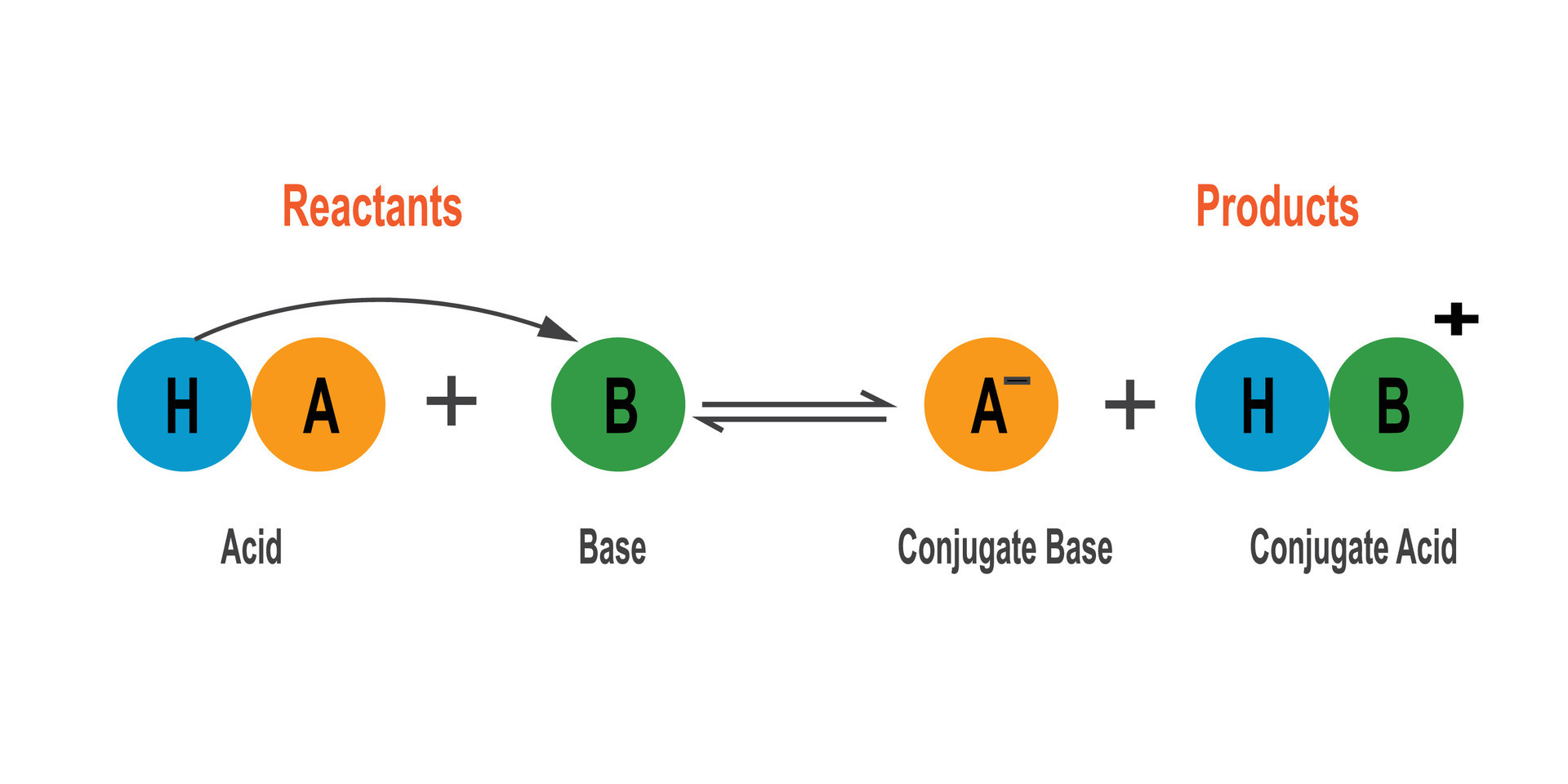
When hydroxide gets its hands on a proton (H⁺), it transforms into water (H₂O). Pretty neat, right? It’s a transformation fueled by its fundamental desire to accept a proton.
It's important to remember that not all bases are Bronsted-Lowry bases, and not all Bronsted-Lowry bases are what we traditionally think of as "basic" in everyday terms. It's a specific chemical behavior, a particular dance they do with protons.
Think about water itself, H₂O. Under certain circumstances, water can act as both an acid and a base! It can donate a proton, acting as an acid, but it can also accept one, acting as a Bronsted-Lowry base. It's a chemical chameleon, a master of disguise!
When water acts as a Bronsted-Lowry base, it accepts a proton and becomes a hydronium ion, H₃O⁺. This happens when it's hanging out with a stronger acid. Water is just being polite, you know, accepting the proton that’s being enthusiastically offered.
So, when you’re faced with a chemical scenario, ask yourself: "Who is willing to be the proton receiver here?" The answer to that question will likely point you directly to your Bronsted-Lowry base. It's like a chemical scavenger hunt, and the prize is understanding this awesome concept.
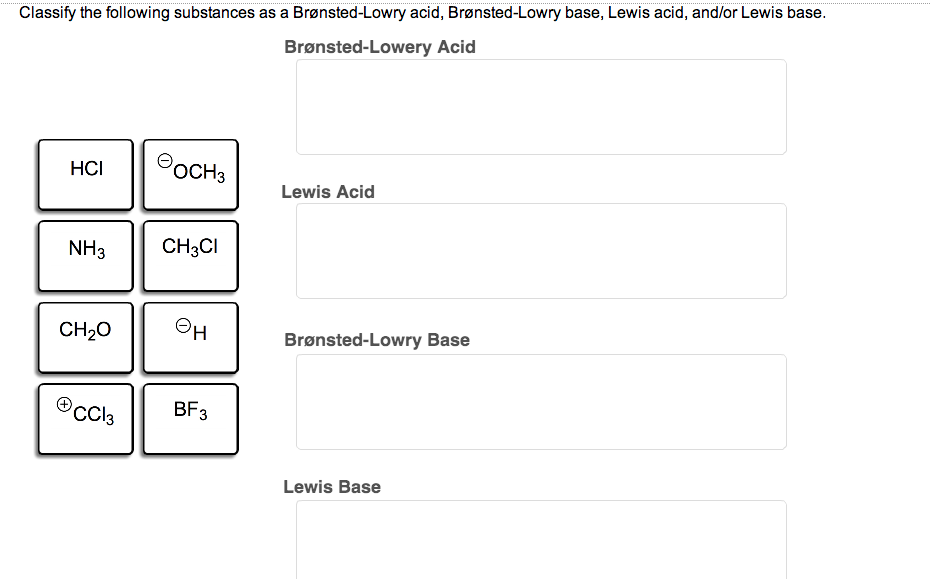
It's all about that transfer of protons. Acids are the givers, and Bronsted-Lowry bases are the enthusiastic receivers. They complete the proton-transfer reaction, making the chemical world go 'round.
Let's say you have hydrochloric acid (HCl), a classic acid. It's practically bursting with protons to give away. When it meets our friend ammonia (NH₃), the magic happens.
HCl donates its proton to NH₃. The NH₃, being a magnificent Bronsted-Lowry base, happily accepts it, forming ammonium (NH₄⁺). Meanwhile, HCl is left with a chloride ion (Cl⁻).
See? It's a beautiful, proton-exchanging ballet. The acid performs a graceful donation, and the Bronsted-Lowry base executes a perfect, eager reception. It’s a perfectly choreographed chemical performance!
So, the next time you see a molecule that seems a bit electron-rich, with available spots for protons, give it a nod. You might be looking at a superhero in disguise, a valiant Bronsted-Lowry base ready to accept its noble proton charge.
Remember the key: proton acceptor. That's the golden ticket to identifying a Bronsted-Lowry base. They are the proton hoarders of the chemical universe, in the most delightful way possible.
It’s not about being a "mean" or "nice" molecule; it’s about a fundamental chemical property. Some molecules are just built to accept those positive protons, and they do it with gusto!
So, if you are presented with a choice, and you see a substance that has a lone pair of electrons ready to form a new bond with a proton, or an existing negative charge that can attract a proton, you've likely found your champion. That’s your Bronsted-Lowry base, shining bright!
It's a concept that unlocks so much more of the chemical world. Understanding these roles – the proton givers and the proton receivers – is like getting a secret decoder ring for chemical reactions. And our Bronsted-Lowry bases are essential players in that grand decoding.
So, go forth and identify those proton-snatching wonders! Embrace the fun of chemistry, and remember that the world of acids and bases is full of fascinating characters, each with their own unique and vital role. And our Bronsted-Lowry bases are, without a doubt, some of the most exciting characters in the entire chemical play!
