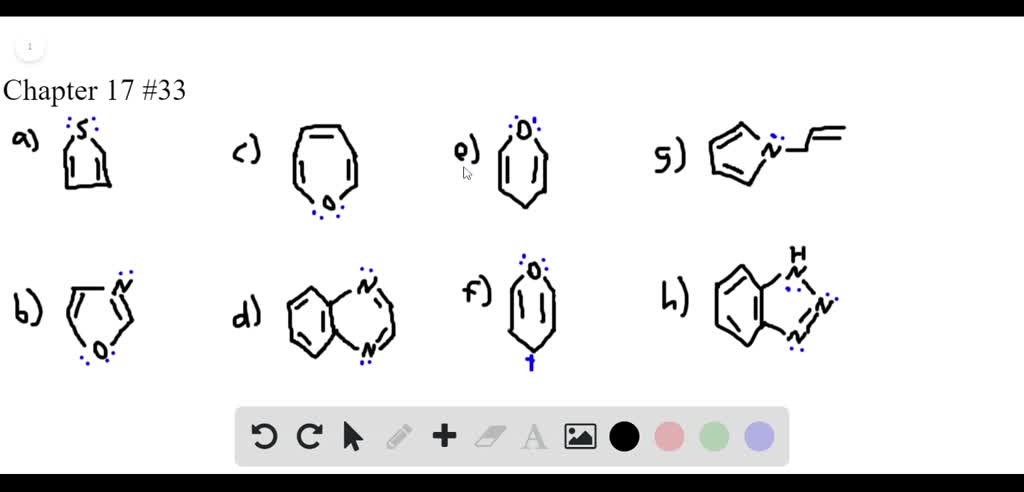
Which Of The Following Heterocycles Are Aromatic
Ever looked at a molecule and wondered what makes some of them so… special? Like, why are certain ring-shaped structures in chemistry so stable and reactive in predictable ways? Well, get ready to dive into the super cool world of aromatic heterocycles! This isn't just for lab coat enthusiasts; understanding these nifty molecules is like unlocking a secret code to how many important substances around us work, from life-saving medicines to the very colors we see. It’s a topic that’s both fascinatingly complex and surprisingly relevant to everyday life, making it a real crowd-pleaser in the chemistry universe.
So, what's the big deal? Think of aromaticity as a kind of "superpower" for molecules. It gives them extra stability and influences how they interact with other chemicals. And when you throw heterocycles into the mix – which are rings containing atoms other than carbon, like nitrogen, oxygen, or sulfur – you get a whole new level of chemical creativity. These aromatic heterocycles are the building blocks for an astonishing array of compounds that are fundamental to biology, medicine, and materials science. They’re the unsung heroes behind countless innovations that improve our lives, making them incredibly useful and, dare we say, fun to explore.
Let's get down to the nitty-gritty: how do we identify these aromatic wonders? It all boils down to a few key rules, often summarized by the wonderfully catchy Hückel's rule. For a ring-shaped molecule to be aromatic, it needs to meet these criteria:
- It must be cyclic: The atoms have to form a closed loop.
- It must be planar: The ring needs to lie flat, like a pancake.
- It must have complete delocalization of pi electrons: This is where things get a bit more exciting! Think of these as mobile electrons that can move freely around the entire ring, like a happy family on a road trip.
- It must have (4n + 2) pi electrons: Here, 'n' is a whole number (0, 1, 2, and so on). This is the magic number of mobile electrons needed to achieve that aromatic stability. So, rings with 2, 6, 10, 14 pi electrons are usually aromatic.
Now, let's put on our detective hats and examine a few common contenders. Imagine presenting a lineup of heterocyclic rings and asking, "Which of these are aromatic?" It’s a classic chemistry quiz, and understanding the answers reveals a lot about chemical behavior.
The stability and unique reactivity conferred by aromaticity make these compounds indispensable in drug design, where precise interactions with biological targets are crucial.
Nucleophilic Aromatic Substitution - Chemistry Steps
Consider the following:
The Usual Suspects: Are They Aromatic?
Let's start with a few very famous examples. One of the most iconic is pyridine. It's a six-membered ring just like benzene, but one of the carbon atoms is replaced by a nitrogen atom. Does it fit the aromatic bill? Let's check:
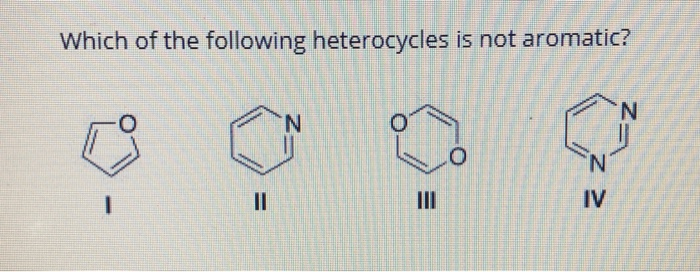
- Cyclic? Yes!
- Planar? Yes, it's a nice flat ring.
- Delocalized pi electrons? Pyridine has alternating double and single bonds within its ring, giving it a system of pi electrons. The nitrogen atom contributes one electron to this pi system, and the carbons contribute one each. This makes for a total of 6 pi electrons (4 from the carbons in the double bonds and 2 from the nitrogen).
- (4n + 2) pi electrons? With 6 pi electrons, and setting n=1, we get 4(1) + 2 = 6. Bingo! So, pyridine is indeed aromatic. This aromatic character makes it a stable, yet reactive molecule, crucial in many organic syntheses and found in vitamins like niacin (Vitamin B3).
Next up, let's look at pyrrole. This is a five-membered ring containing one nitrogen atom. It sounds a bit different, so will it behave the same way? Let's see:
- Cyclic? Absolutely.
- Planar? Yes, it forms a flat ring.
- Delocalized pi electrons? Pyrrole has two double bonds within its ring, contributing 4 pi electrons. The nitrogen atom, however, has a lone pair of electrons. In pyrrole, this lone pair is not busy with bonding but is instead able to participate in the pi system, contributing 2 more electrons. So, we have a total of 4 (from double bonds) + 2 (from nitrogen's lone pair) = 6 pi electrons circulating.
- (4n + 2) pi electrons? Again, with 6 pi electrons (n=1), pyrrole meets the requirement. Therefore, pyrrole is also aromatic. This makes it a fundamental component of many biological molecules, including hemoglobin and chlorophyll!
What about something like furan? This is similar to pyrrole but with an oxygen atom instead of nitrogen in the five-membered ring. Let's investigate:
- Cyclic? Check.
- Planar? Yes.
- Delocalized pi electrons? Furan has two double bonds, giving us 4 pi electrons. The oxygen atom also has two lone pairs. One of these lone pairs is oriented in a way that it can participate in the pi system, much like the nitrogen in pyrrole. This adds another 2 electrons. So, total pi electrons = 4 + 2 = 6.
- (4n + 2) pi electrons? With 6 pi electrons (n=1), furan also qualifies as aromatic. It's a versatile building block in organic chemistry and contributes to the flavor profiles of many foods.
Now, let's consider a slightly different case: thiophene. This is a five-membered ring with a sulfur atom. Sulfur is a bit larger and has different electron configurations than nitrogen or oxygen. Does it still play by the same rules?
- Cyclic? Yes.
- Planar? Yes.
- Delocalized pi electrons? Thiophene has two double bonds (4 pi electrons). Sulfur has two lone pairs. Similar to oxygen and nitrogen in their respective heterocycles, one of sulfur's lone pairs is available to delocalize into the pi system, contributing 2 more electrons. This gives us a total of 6 pi electrons.
- (4n + 2) pi electrons? With 6 pi electrons (n=1), thiophene is declared aromatic. This sulfur-containing ring is a key component in pharmaceuticals and materials science.
These molecules – pyridine, pyrrole, furan, and thiophene – are the rockstars of aromatic heterocycles. Their aromatic nature means they're not just static structures; they possess a special kind of stability and a predictable way of reacting that makes them incredibly valuable. Understanding if a heterocycle is aromatic is a fundamental step in predicting its behavior and harnessing its potential, whether in the lab or in the complex machinery of life itself. It’s this blend of structural elegance and functional importance that makes exploring aromatic heterocycles such a rewarding journey for any curious mind.

